Cannabidiol API and CDMO Services Market Forecast with Size and Share Analysis 2026 to 2035
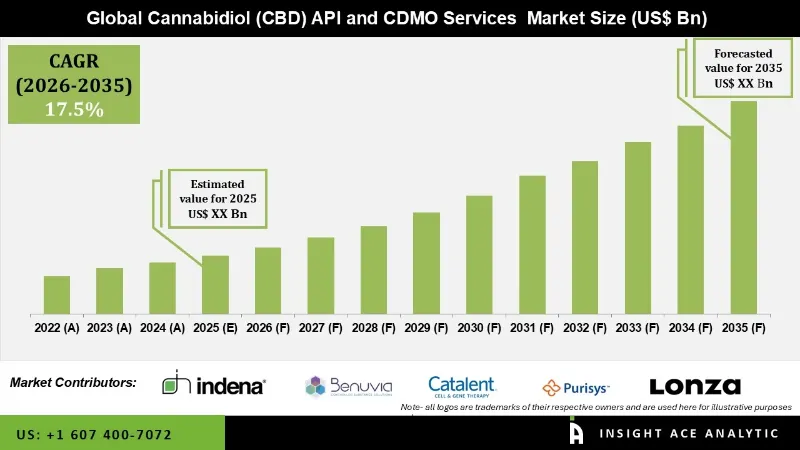
Cannabidiol (CBD) API and CDMO Services Market is estimated to grow at a 17.5% CAGR during the forecast period for 2026 to 2035.
Cannabidiol (CBD) API and CDMO Services Market Size, Share & Trends Analysis Distribution by Type/Source (Hemp-Derived CBD API, Marijuana-Derived CBD API, Synthetic CBD API), By Product Form (CBD Isolate, Full-Spectrum CBD, Broad-Spectrum CBD), By Application/End-Use (Pharmaceuticals, Nutraceuticals, Cosmetics, Food & Beverages, Pet Care), By Services (API Development & Manufacturing, Formulation Development, Clinical Trial & Commercial Manufacturing, Private/White Label Services), and Segment Forecasts, 2026 to 2035
Cannabidiol (CBD) has evolved from a niche cannabinoid into a strategically important pharmaceutical and wellness ingredient, driven by its broad therapeutic potential and non-psychoactive profile. Mostly obtained from hemp or througCosmetics h synthetic and biosynthetic methods, cannabidiol has received greater acceptance in terms of regulations and clinical applications after being approved in prescribed medications, especially in Neurology applications.
The use of cannabidiol has received greater promotion on a worldwide level in North America, Europe, Asia-Pacific, and developing nations due to its increasingly growing clinical research and evolving regulations. The cannabidiol molecule has applications in being analgesic, anxiolytic, neuroprotective, and antiepileptic, making its applications possible in Dermatology, Nutraceuticals, Cosmetics, and Neurology. Non-drug applications of cannabidiol are increasingly developing on a worldwide level; however, the role of pharmaceutical grade cannabidiol in applications demands high-quality, consistent, and traceable cannabidiol produced on strict quality standards.
The cannabidiol Active Pharmaceutical Ingredient (API) & CDMO market has grown as a result of this increasing global adoption as pharma companies look for stable sources of GMP-qualified CBD as well as the necessary know-how for complex CDMO manufacturing processes. Cannabidiol Active Pharmaceutical Ingredient refers to highly purified, standardized CBD that is manufactured to comply with pharmaceutical regulatory standards, such as highly controlled impurities and THC levels. CDMOs serve as crucial partners for pharma companies by providing one-stop support for manufacturing processes such as extraction or synthetic production, development of processes, purification, formulation development, scaleup work, analytical development work, and regulatory assistance to help companies expedite launch timelines in various geographies.
Drivers for this market have been the accelerating clinical confirmation of CBD-based medicines, increasingly complex contract manufacturing work outsourcing by pharma companies, and pressures for worldwide harmonized logistics chains. Among challenges, there has been divergent regulation of cannabinoids and THC limits.
Competitive Landscape
Which are the Leading Players in the Cannabidiol (CBD) API and CDMO Services Market?
• Indena
• Veranova
• Brains Bioceutical (Brains API)
• Vantage Hemp
• Noramco
• CAELO
• Canexis Pharma
• Aesica Pharmaceuticals
• HYTN
• Benuvia
• Axplora (Farmabios)
• Purisys
• Lonza Group
• Thermo Fisher Scientific (Patheon CDMO)
• Catalent
• Recipharm
Market Dynamics
Driver
Increasing legalization of cannabis for medicinal purposes
A prominent factor propelling the market for the cannabidiol API and CDMO has been the increasing legalization of cannabis for medical purposes, with CBD being integrated with regulated medical systems. Given that more countries are also accepting cannabis for medical purposes, especially relating to neurological and chronic diseases, pharmaceutical companies are also investing more in medical drug development involving CBD, leading to a rising demand for pharmaceutical-grade cannabidiol APIs with a high degree of purity. Based on such changes brought about within a regulated medical setting, there is also a transition from wellness production to pharmaceutical production.
Restrain/Challenge
Regulatory Approval Delays
The major restraint to the growth of the cannabidiol API and CDMO market is the regulatory hurdles in the form of cautious and delayed approval of drugs derived from CBD by the FDA. There is only one approved drug, Epidiolex, as yet approved out of those with substantial clinical data; these delays put a damper on global demand for CBD APIs and slow down therapeutic adoption of the same. Such oversight has been strong because of the complex chemical nature of CBD and the possibility of resultant side effects, leading to uncertainty for CDMOs making investments in scale-up since growth in the market depends by and large on quicker regulatory approvals and clearer pathways for new product launches.
Hemp-Derived CBD API Segment is Expected to Drive the Cannabidiol (CBD) API and CDMO Services Market
The Hemp-Derived CBD API segment is anticipated to fuel the market. The main reason for this would be the fact that hemp-derived CBD APIs are legally recognized and approved in most nations with a strict THC level, and they are easily accessible compared to marijuana-derived CBD. Another significant factor would be the increasing requirement for GMP-certified medical-grade CBD products globally, which would make this segment the primary revenue generator in the market.
Full-Spectrum CBD is Growing at the Highest Rate in the Cannabidiol (CBD) API and CDMO Services Market
The Full Spectrum CBD market is expanding at the highest rate amongst the lot. Full Spectrum CBD products include not only CBD but also a combination of natural cannabinoids and various biochemicals in the Cannabis plant, and are known to help in the “Entourage Effect” and offer better results. Full-spectrum CBD products are highly popular in medical products, especially in the field of Neurology for managing epilepsy and chronic neurologic problems and in Dermatology, Nutraceuticals, and Cosmetics products.
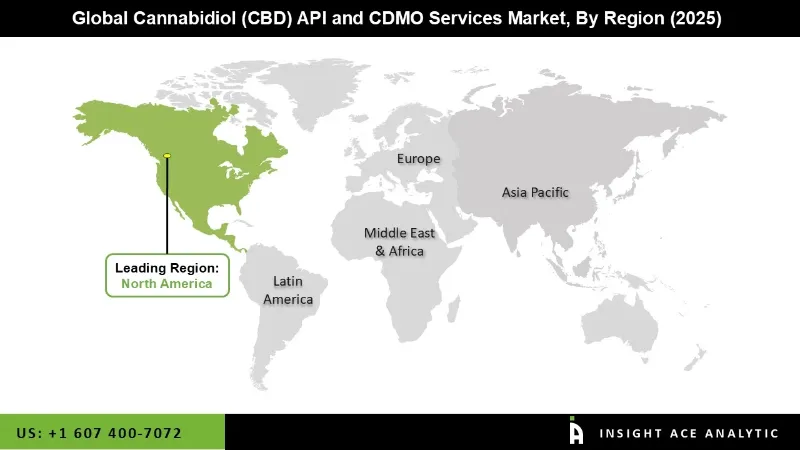
Why North America Led the Cannabidiol (CBD) API and CDMO Services Market?
The North America region dominates the market for CBD API and CDMO. This is due to the well-established regulatory guidelines, especially in the U.S., which have paved ways for CBD derived from hemp as well as FDA guidelines for pharmaceutical CBD approvals, including that of Epidiolex for Epilepsy, creating a success route for CBD in this domain as well. Moreover, a vast rise in the applications of CBD in Medical, Neuroscience, Dermatology, Nutraceuticals, and Cosmetics has continuously driven demand for highly pure CBD API.
Key Development
• In July 2025, HYTN Innovations Inc. announced that it received a Cannabis Drug Licence (CDL) from Health Canada, complementing its existing Drug Establishment Licence (DEL) and federal Cannabis Licences. This approval places the company among a limited group authorized to manufacture cannabis-derived pharmaceutical drugs in Canada.
Cannabidiol (CBD) API and CDMO Services Market Report Scope:
| Report Attribute | Specifications |
| Growth Rate CAGR | CAGR of 17.5% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2024 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Type/Source, Product Form, Application/End-Use, Services, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Indena, Veranova, Brains Bioceutical (Brains API), Vantage Hemp, Noramco, CAELO, Canexis Pharma, Aesica Pharmaceuticals, HYTN, Benuvia, Axplora (Farmabios), Purisys, Lonza Group, Thermo Fisher Scientific (Patheon CDMO), Catalent, Recipharm |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Cannabidiol (CBD) API and CDMO Services Market by Type/Source-
- Hemp-Derived CBD API
- Marijuana-Derived CBD API
- Synthetic CBD API
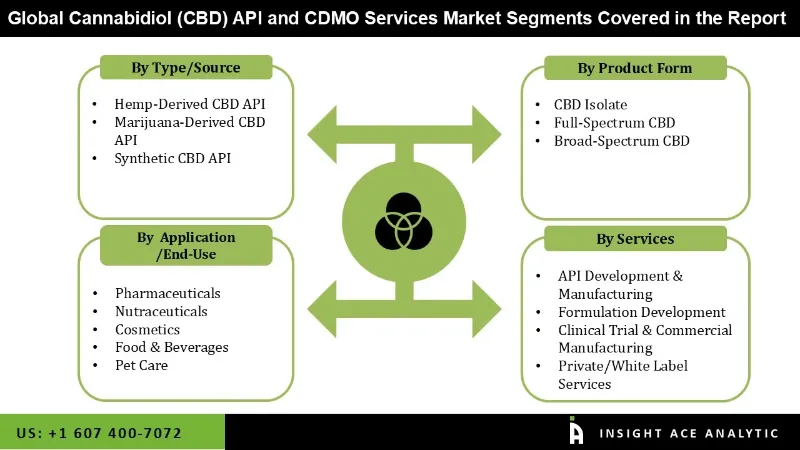
Cannabidiol (CBD) API and CDMO Services Market by Product Form-
- CBD Isolate
- Full-Spectrum CBD
- Broad-Spectrum CBD
Cannabidiol (CBD) API and CDMO Services Market by Application/End-Use-
- Pharmaceuticals
- Nutraceuticals
- Cosmetics
- Food & Beverages
- Pet Care
Cannabidiol (CBD) API and CDMO Services Market by Services -
- API Development & Manufacturing
- Formulation Development
- Clinical Trial & Commercial Manufacturing
- Private/White Label Services
Cannabidiol (CBD) API and CDMO Services Market By Region-
- North America-
• The US
• Canada
- Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
- Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
- Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
- Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Cannabidiol (CBD) API and CDMO Services Market is estimated to grow at a 17.5% CAGR during the forecast period for 2026 to 2035.
Indena, Veranova, Brains Bioceutical (Brains API), Vantage Hemp, Noramco, CAELO, Canexis Pharma, Aesica Pharmaceuticals, HYTN, Benuvia, Axplora (Farmabios), Purisys, Lonza Group, Thermo Fisher Scientific (Patheon CDMO), Catalent, Recipharm
Type/Source, Product Form, Application/End-Use, Services, and By Region are the key segments of the Cannabidiol (CBD) API and CDMO Services Market.
North America region is leading the Cannabidiol (CBD) API and CDMO Services Market.