Nasal Drug Delivery Market Size, Scope, Revenue Report 2026 to 2035
What is Nasal Drug Delivery Market Size?
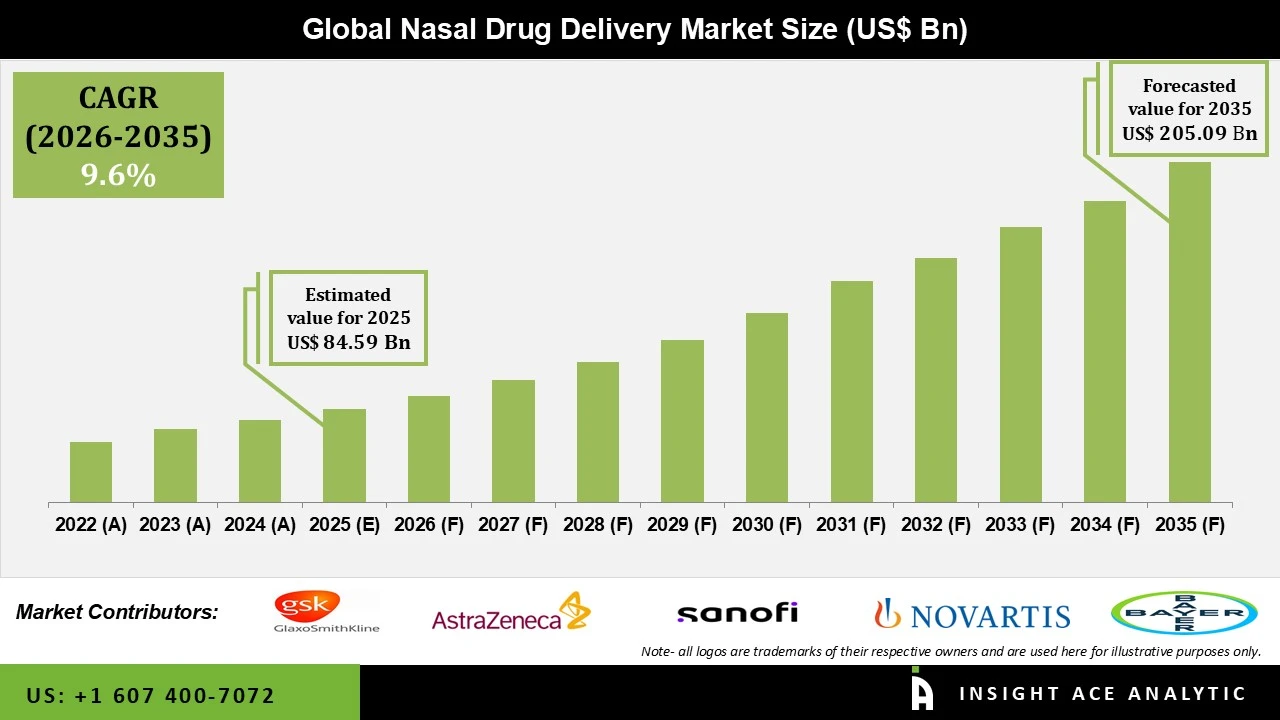
Nasal Drug Delivery Market Size is valued at USD 84.59 Bn in 2025 and is predicted to reach USD 205.09 Bn by the year 2035 at a 9.6% CAGR during the forecast period for 2026 to 2035.
Nasal Drug Delivery Market Size, Share & Trends Analysis Distribution By Product Type (Local Nasal Therapeutics, Systemic Nasal Drug Delivery, Emergency/Rescue Nasal Drugs, CNS/Nose-to-Brain Delivery, Vaccines & Biologics), Dosage Form (Nasal Sprays, Nasal Drops, Nasal Powders, Nasal Gels & Ointments, Others), Delivery System / Device Type (Multi-dose Systems, Unit-dose Systems, Bi-dose Systems), Container Type (Pressurized Containers, Non-pressurized Containers), Therapeutic Application (Allergic & Non-allergic Rhinitis, Nasal Congestion, Sinusitis, Neurological Disorders, Pain & Migraine), Route of Delivery (Local Nasal Delivery, Systemic Delivery, Nose-to-Brain Delivery), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), End User (Hospitals, Homecare Settings, Specialty Clinics), Technology Platform (Mucoadhesive Systems, Permeation Enhancers, Nanoparticle-based Delivery, Controlled-release Systems) and Segment Forecasts, 2026 to 2035
The nasal drug delivery market represents a steadily evolving segment of the pharmaceutical industry focused on delivering medications through the nasal route for both local and systemic effects. This approach has gained strong attention because it offers a non-invasive, fast-acting alternative to traditional oral and injectable methods.
Nasal delivery allows drugs to be absorbed quickly through the nasal mucosa, making it especially useful for conditions that require rapid relief, such as allergies, nasal congestion, migraines, and emergencies, such as severe allergic reactions. In recent years, advancements in formulation technologies, device design, and absorption enhancers have improved drug effectiveness and the patient experience. Companies are also exploring innovative approaches such as nose-to-brain delivery, which enables certain drugs to bypass the blood-brain barrier and directly target the central nervous system.
These solutions are becoming increasingly important as healthcare providers and patients seek more convenient, patient-friendly treatment options. The market is seeing increasing adoption across therapeutic areas, including respiratory disorders, neurological conditions, and pain management. Demand is also supported by the rising prevalence of chronic conditions, increased awareness of self-care treatments, and the widespread availability of over-the-counter nasal products.
At the same time, pharmaceutical companies are investing in research and development to expand the use of nasal delivery for biologics and emergency therapies, further strengthening market growth. However, the market faces several challenges, including limitations in drug formulation, variability in absorption, and stringent regulatory requirements for new delivery systems. Despite this, ongoing innovation in drug-delivery technologies and the increasing acceptance of non-invasive treatments are helping the nasal drug-delivery market continue to grow. Overall, it is becoming an increasingly important part of modern healthcare, offering patients faster, safer, and more convenient treatment options.
Competitive Landscape
Which are the Leading Players in the Nasal Drug Delivery Market?
• GlaxoSmithKline
• AstraZeneca
• Sanofi
• Novartis
• Bayer
• Merck & Co.
• Pfizer
• Johnson & Johnson
• Teva Pharmaceutical Industries
• Bausch Health
• AptarGroup
• Nemera
• Becton, Dickinson and Company
• Consort Medical
• Vectura Group
• OptiNose
• Neurelis
• Impel NeuroPharma
• ARS Pharmaceuticals
• Naveh Pharma
• Other Prominent Player
Market Dynamics
Driver
Rising Demand for Non-Invasive and Convenient Drug Delivery.
The major driver of the nasal drug delivery market is the increasing preference for non-invasive, convenient treatment options. Today, patients are more aware and conscious about their healthcare choices, and they tend to favor methods that are easy to use, painless, and do not require hospital visits. Nasal delivery fits perfectly into this need, as it allows medications to be administered quickly and comfortably, often even at home. This is particularly beneficial for people managing chronic conditions or those who require immediate relief, such as in respiratory issues or pain management. As a result, both patients and healthcare providers are increasingly adopting nasal drug delivery as a practical and user-friendly alternative to traditional treatment methods.
Restrain/Challenge
Variability in Nasal Drug Absorption Limiting Consistent Treatment Outcome
The nasal drug delivery market is the variability in how drugs are absorbed through the nasal cavity. Unlike more controlled delivery methods, nasal absorption can vary from person to person due to factors such as nasal congestion, allergies, and environmental conditions. For example, a blocked or irritated nasal passage can reduce the effectiveness of drug absorption, leading to inconsistent results. This makes it difficult for pharmaceutical companies to ensure precise dosing and reliable outcomes for every patient. As a result, despite its many advantages, maintaining consistent performance remains a significant hurdle for nasal drug delivery systems.
Local Nasal Therapeutics Segment is Expected to Drive the Nasal Drug Delivery Market
The local nasal therapeutics segment is a key driver of the nasal drug delivery market. The treatment of common conditions such as nasal congestion, allergic rhinitis, and sinusitis requires solutions that are fast, effective, and easy to use, and nasal therapeutics meet these needs efficiently. These products deliver medication directly to the affected area, providing quick relief while minimising systemic side effects. Additionally, their widespread availability as over-the-counter products makes them highly accessible to patients, reducing the need for frequent doctor visits. With the rising prevalence of respiratory and allergy-related conditions, the demand for local nasal treatments continues to grow steadily. Pharmaceutical companies are also focusing on improving formulations to enhance effectiveness and patient comfort. Overall, the convenience, affordability, and strong patient preference for these products make the local nasal therapeutics segment a major driver of market growth and adoption.
Nasal Sprays Segment is Expected to Drive the Nasal Drug Delivery Market
The nasal sprays segment plays a leading role in driving the nasal drug delivery market, largely due to its simplicity, effectiveness, and strong patient preference. Nasal sprays are widely used because they are easy to administer, non-invasive, and provide quick relief, making them a convenient option for both everyday use and long-term treatment. Patients can use them without medical supervision, which makes them especially suitable for home care settings. In addition, nasal sprays are commonly available over the counter, further increasing their accessibility and adoption across a broad patient base. They are widely used for conditions such as nasal congestion, allergies, and sinus-related issues, contributing to consistent demand. Pharmaceutical companies are also continuously improving spray technologies to enhance dosing accuracy and patient comfort. Overall, the ease of use, rapid action, and widespread availability of nasal sprays make them a key segment supporting the growth and expansion of the nasal drug delivery market.
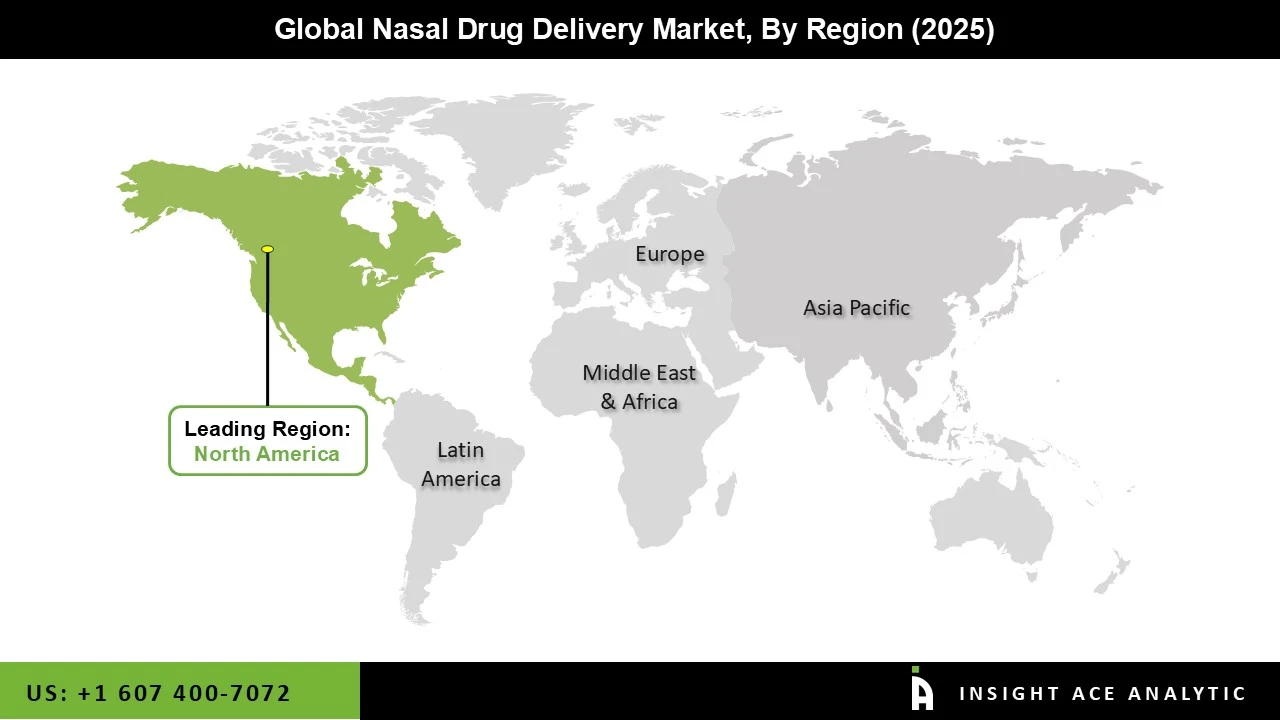
Why North America Led the Nasal Drug Delivery Market?
North America’s leadership in the nasal drug delivery market is driven by a strong combination of advanced healthcare infrastructure, supportive regulations, and high patient demand. The United States, in particular, stands out for its access to cutting-edge treatments, willingness of insurers to cover innovative therapies, and rapid adoption by healthcare professionals. A key advantage is the structured and reliable approval process led by the U.S. Food and Drug Administration, which makes the region a preferred launch market for new nasal drug products. As a result, many of the latest therapies are introduced here first. The region also benefits from a large patient base dealing with conditions like allergies, sinusitis, and respiratory issues, ensuring consistent demand. Additionally, a strong over-the-counter culture encourages consumers to self-manage minor conditions using nasal sprays and similar products. Backed by significant R&D investments from leading pharmaceutical companies, North America continues to be a hub where innovation is developed, validated, and commercialized quickly. Overall, its market leadership is the result of the right balance between infrastructure, regulation, patient needs, and openness to innovation.
Key Developments
• In March 2025, neffy nasal spray approval, The U.S. Food and Drug Administration approved neffy, introducing a needle-free epinephrine nasal spray for children that marked a breakthrough and improved emergency nasal drug delivery.
• In February 2025, Zeteo Biomedical launched dry powder nasal delivery systems, highlighting a shift beyond traditional liquid sprays and demonstrating innovation in nasal delivery of drugs and biologics.
Nasal Drug Delivery Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 84.59 Bn |
| Revenue forecast in 2035 | USD 205.09 Bn |
| Growth Rate CAGR | CAGR of 9.6% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2021 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Dosage Form, Delivery System/Device Type, Container Type, Therapeutic Application, Route of Delivery, Distribution Channel, End User, Technology Platform and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | GlaxoSmithKline, AstraZeneca, Sanofi, Novartis, Bayer, Merck & Co., Pfizer, Johnson & Johnson, Teva Pharmaceutical Industries, Bausch Health, AptarGroup, Nemera, Becton, Dickinson and Company, Consort Medical, Vectura Group, OptiNose, Neurelis, Impel NeuroPharma, ARS Pharmaceuticals, Naveh Pharma, Other Prominent Players |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Nasal Drug Delivery Market Segmentation:
Nasal Drug Delivery Market by Product Type -
• Local Nasal Therapeutics
• Systemic Nasal Drug Delivery
• Emergency/Rescue Nasal Drugs
• CNS/Nose-to-Brain Delivery
• Vaccines & Biologics
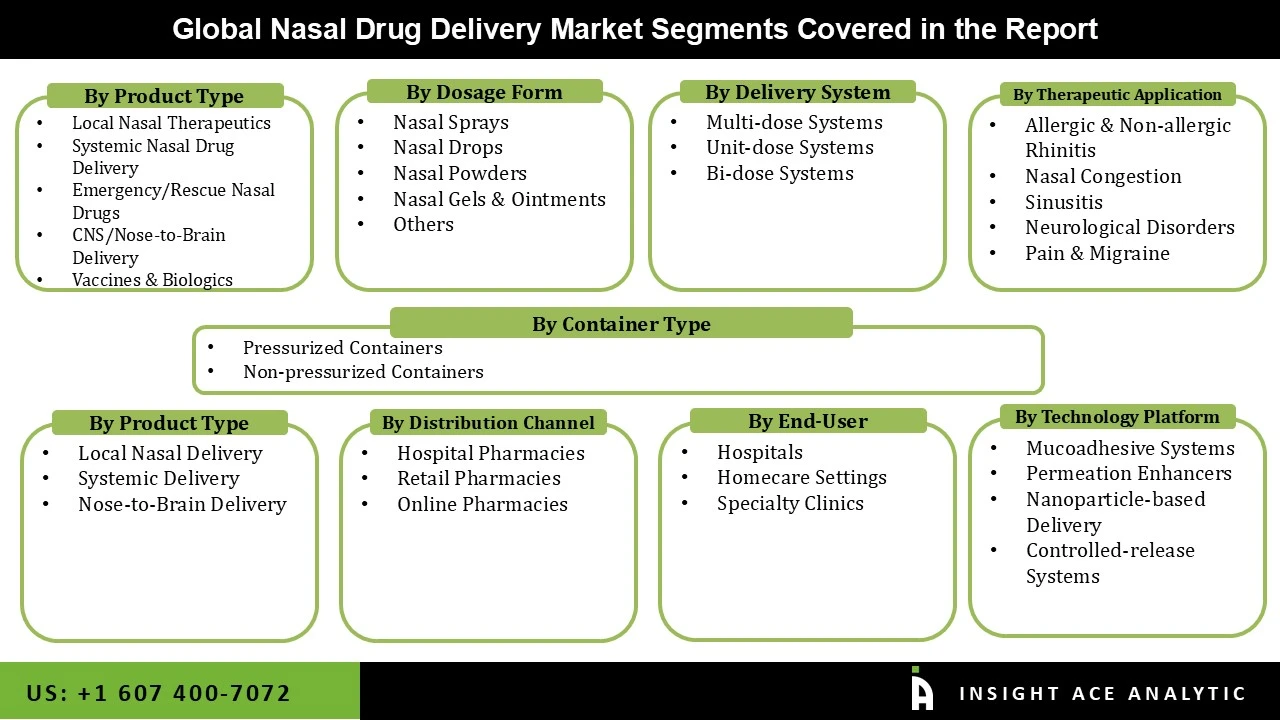
Nasal Drug Delivery Market by Dosage Form-
• Nasal Sprays
• Nasal Drops
• Nasal Powders
• Nasal Gels & Ointments
• Others
Nasal Drug Delivery Market by Delivery System/ Device Type -
• Multi-dose Systems
• Unit-dose Systems
• Bi-dose Systems
Nasal Drug Delivery Market by Container Type -
• Pressurized Containers
• Non-pressurized Containers
Nasal Drug Delivery Market by Deployment Model–
• Cloud-Based Platforms
• On-Premise Solutions
• Hybrid Models
Nasal Drug Delivery Market by Therapeutic Application–
• Allergic & Non-allergic Rhinitis
• Nasal Congestion
• Sinusitis
• Neurological Disorders
• Pain & Migraine
Nasal Drug Delivery Market by Route of Delivery–
• Local Nasal Delivery
• Systemic Delivery
• Nose-to-Brain Delivery
Nasal Drug Delivery Market by Distribution Channel-
• Hospital Pharmacies
• Retail Pharmacies
• Online Pharmacies
Nasal Drug Delivery Market by End Users-
• Hospitals
• Homecare Settings
• Specialty Clinics
Nasal Drug Delivery Market by Technology Platform-
• Mucoadhesive Systems
• Permeation Enhancers
• Nanoparticle-based Delivery
• Controlled-release Systems
Nasal Drug Delivery Market by Region-
North America-
• The US
• Canada
Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Nasal Drug Delivery Market Size is valued at USD 84.59 Bn in 2025 and is predicted to reach USD 205.09 Bn by the year 2035
Nasal Drug Delivery Market is expected to grow at a 9.6% CAGR during the forecast period for 2026 to 2035.
GlaxoSmithKline, AstraZeneca, Sanofi, Novartis, Bayer, Merck & Co., Pfizer, Johnson & Johnson, Teva Pharmaceutical Industries, Bausch Health, AptarGroup, Nemera, Becton, Dickinson and Company, Consort Medical, Vectura Group, OptiNose, Neurelis, Impel NeuroPharma, ARS Pharmaceuticals, Naveh Pharma, Other Prominent Players
Nasal Drug Delivery Market is segmented into Product Type, Dosage Form, Delivery System/Device Type, Container Type, Therapeutic Application, Route of Delivery, Distribution Channel, End User, Technology Platform and By Region
North America region is leading the Nasal Drug Delivery Market.