Speciality and Orphan Drug Market Size, Share, Trend Report 2026 to 2035
What is Speciality and Orphan Drug Market Size?
Speciality and Orphan Drug Market Size is predicted to reach grow at a 11.6% CAGR during the forecast period for 2026 to 2035.
Speciality and Orphan Drug Market Size, Share & Trends Analysis by Therapeutic Area (Oncology, Neurology & CNS Disorders, Hematology, Metabolic & Genetic Disorders, Lysosomal Storage Disorders, Immunology & Autoimmune Rare Diseases, Cardiovascular Rare Diseases, Infectious Diseases (Rare / Specialty), Rare Pediatric Diseases (Cross-segment), Respiratory Rare Diseases (CF, rare ILDs), Endocrine & Rare Hormonal Disorders), by Drug Modality (Small Molecules, Biologics, Cell & Gene Therapy, Nucleic Acid Therapies, Enzyme Replacement Therapy (ERT), Substrate Reduction Therapy, Peptides & Hormonal Therapies, Reformulated), by Dosage Form (Oral Solids (Tablets, Capsules), Oral Liquids / Suspensions / Solutions, Injectables, Long-Acting / Depot Formulations, Transdermal / Topical, Inhalation / Nasal, Advanced Delivery Systems), Distribution Channel (Speciality Pharmacy, Hospital / Buy-and-Bill, Retail Pharmacy (Limited), Hub Services (Patient Support Programs), Direct-to-Patient Distribution), and Segment Forecasts 2026 to 2035.
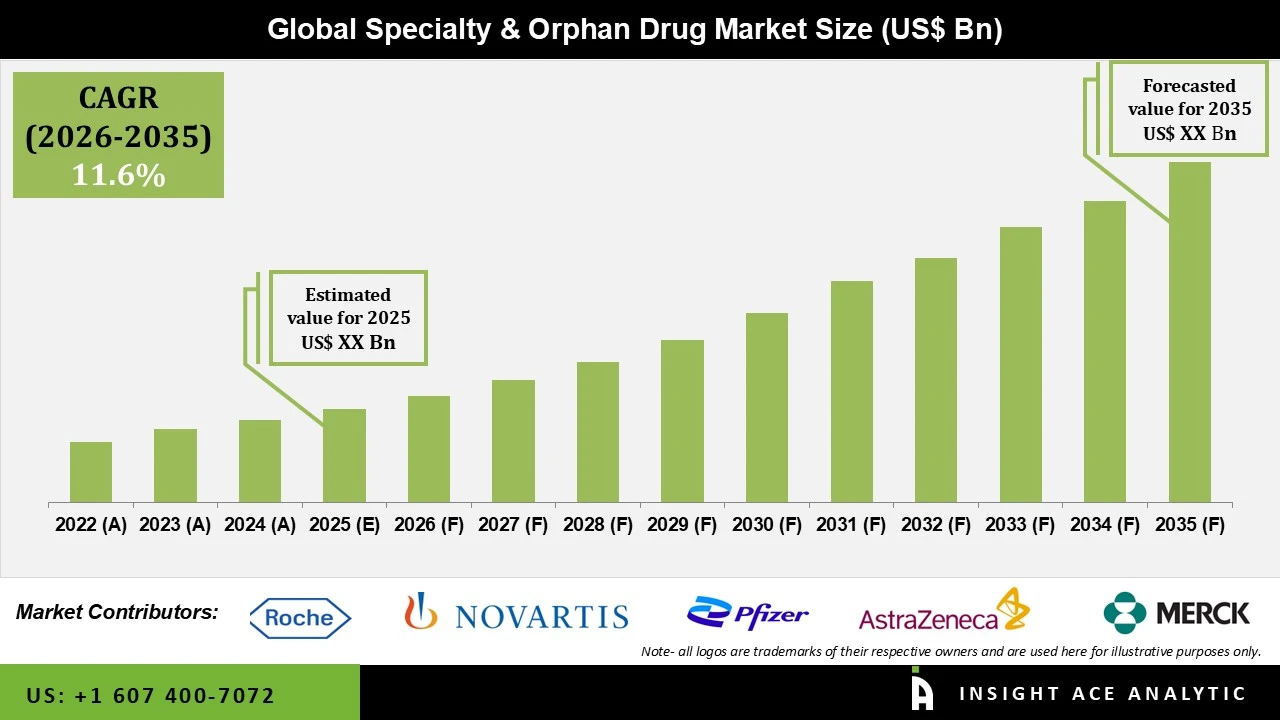
The speciality and orphan drug market shows a rapidly growing segment of the pharmaceutical industry, focusing on treatments for rare, complex, and hard-to-treat conditions. This market covers drugs for rare genetic disorders, oncology, immunology, metabolic diseases, and other speciality therapeutic areas where conventional treatments may be limited or unavailable. Speciality and orphan drugs often involve advanced modalities, including biologics, gene therapies, enzyme replacement therapies, and personalised medicines, designed to address highly specific patient needs.
These drugs provide significant benefits by offering targeted, life-changing therapies for patients with rare or underserved conditions and help improve patient outcomes, quality of life, and overall disease management. In recent years, Pharmaceutical companies have increasingly invested in research and development for these therapies, leveraging innovations in genomics, biotechnology, and precision medicine. The growing focus on rare diseases, combined with supportive government policies such as orphan drug incentives, market exclusivity, and expedited approval pathways, has fueled market growth.
Market growth is further supported by advanced healthcare infrastructure, well-developed distribution channels, and strong patient support programs, which help ensure that high-cost, complex therapies reach the right patients efficiently. North America leads the market due to favourable regulations, increased healthcare spending, robust research and innovation ecosystems, and strong collaborations between industry, academia, and patient advocacy groups. Despite the possibilities, the market faces challenges such as high drug development costs, complex manufacturing processes, pricing and reimbursement issues, and limited patient populations, which can slow adoption. However, ongoing innovation in biologics, gene therapies, and patient-centric programs, combined with growing awareness of rare diseases, continues to drive the Speciality and Orphan Drug Market forward, making it a vital and dynamic segment of modern healthcare.
Competitive Landscape
Which are the Leading Players in the Speciality & Orphan Drug Market?
• Roche
• Novartis
• Pfizer
• Johnson & Johnson
• AstraZeneca
• Merck & Co.
• Sanofi
• Takeda Pharmaceutical
• Bristol Myers Squibb
• Eli Lilly and Company
• Vertex Pharmaceuticals
• BioMarin Pharmaceutical
• Ultragenyx Pharmaceutical
• argenx
• Alnylam Pharmaceuticals
• Ionis Pharmaceuticals
• Sarepta Therapeutics
• Blueprint Medicines
• Incyte
• Azurity Pharmaceuticals
• Horizon Therapeutics
• Chiesi Farmaceutici
• Recordati Rare Diseases
• Amryt Pharma
• Supernus Pharmaceuticals
• Collegium Pharmaceutical
• Assertio Holding
Market Dynamics
Driver
Addressing Rare Diseases and Unmet Medical Needs
The growing focus on rare diseases and unmet medical needs is strongly driving the speciality and orphan drug market. Many patients suffering from rare or complex conditions have historically had limited or no treatment options, creating an urgent demand for innovative therapies. This has encouraged pharmaceutical and biotechnology companies to invest heavily in the development of specialised treatments, including biologics, gene therapies, and personalised medicines, that target specific patient populations. In addition, supportive government policies such as orphan drug incentives, market exclusivity, and faster regulatory approvals make it more feasible for companies to bring these therapies to market. As a result, the market continues to expand, offering life-changing solutions for patients while opening new opportunities for healthcare innovation.
Restrain/Challenge
High cost and complexity of development
The most significant challenges in the Speciality and Orphan Drug Market are the high cost and complexity of developing new therapies. Treatments for rare and complex diseases often rely on advanced technologies such as biologics, gene therapies, and personalised medicine, which require extensive research, specialised manufacturing, and rigorous clinical trials. Developing these therapies is time-consuming and resource-intensive, requiring significant investment in laboratories, equipment, and expert personnel. Because these conditions affect relatively small patient populations, companies face difficulty in recovering costs, making it harder to achieve financial sustainability. This challenge is further amplified by the need to comply with strict regulatory requirements, ensure product quality, and manage complex distribution and storage. Despite these hurdles, companies continue to invest in innovation, as these therapies offer life-changing solutions for patients with rare and unmet medical needs, making them a critical focus in modern healthcare.
Oncology Segment Expected to Drive the Specialty & Orphan Drug Market
Oncology is the leading segment in the Speciality and Orphan Drug Market, driven by the high prevalence of cancer and the increasing need for advanced, targeted therapies. Patients with complex or rare cancers often require specialised treatments that go beyond conventional chemotherapy, such as biologics, immunotherapies, and gene-targeted therapies. Pharmaceutical companies are investing heavily in research and development to bring these innovative therapies to market, supported by regulatory incentives and faster approval pathways. The dominance of oncology is also supported by strong patient awareness, robust clinical trial networks, and specialised healthcare infrastructure, which help ensure timely diagnosis and access to treatment. Additionally, the high unmet medical need and willingness of healthcare systems to adopt innovative therapies make oncology a high-value, fast-growing segment within the broader Speciality and Orphan Drug Market.
Injectables Segment Expected to Drive the Specialty & Orphan Drug Market
Injectables are the leading dosage form in the Specialty and Orphan Drug Market, reflecting the growing demand for advanced therapies that require precise and controlled administration. Many specialty and orphan drugs, including biologics, gene therapies, and enzyme replacement therapies, are best delivered through injections to ensure maximum efficacy and safety. Injectables also allow healthcare providers to manage dosing accurately, which is particularly important for rare or complex conditions where patient response must be closely monitored. The preference for injectables is further supported by specialised healthcare infrastructure, including hospitals, clinics, and trained personnel, as well as patient support programs that help ensure proper administration and adherence. As a result, injectables remain a critical and high-value segment, driving the adoption of specialty therapies and shaping the growth of the market.
Why North America Led the Speciality & Orphan Drug Market?
North America leads the Speciality and Orphan Drug Market due to its strong focus on innovation, supportive regulations, and advanced healthcare infrastructure. Government policies, including orphan drug incentives, offer benefits such as faster approvals, market exclusivity, and financial support, encouraging companies to invest in treatments for rare diseases. The region benefits from world-class research institutions, biotech hubs, and pharmaceutical companies that collaborate to develop biologics, gene therapies, and other advanced therapies. Strong healthcare infrastructure, experienced specialists, and dedicated rare disease centres ensure early diagnosis and efficient treatment delivery.
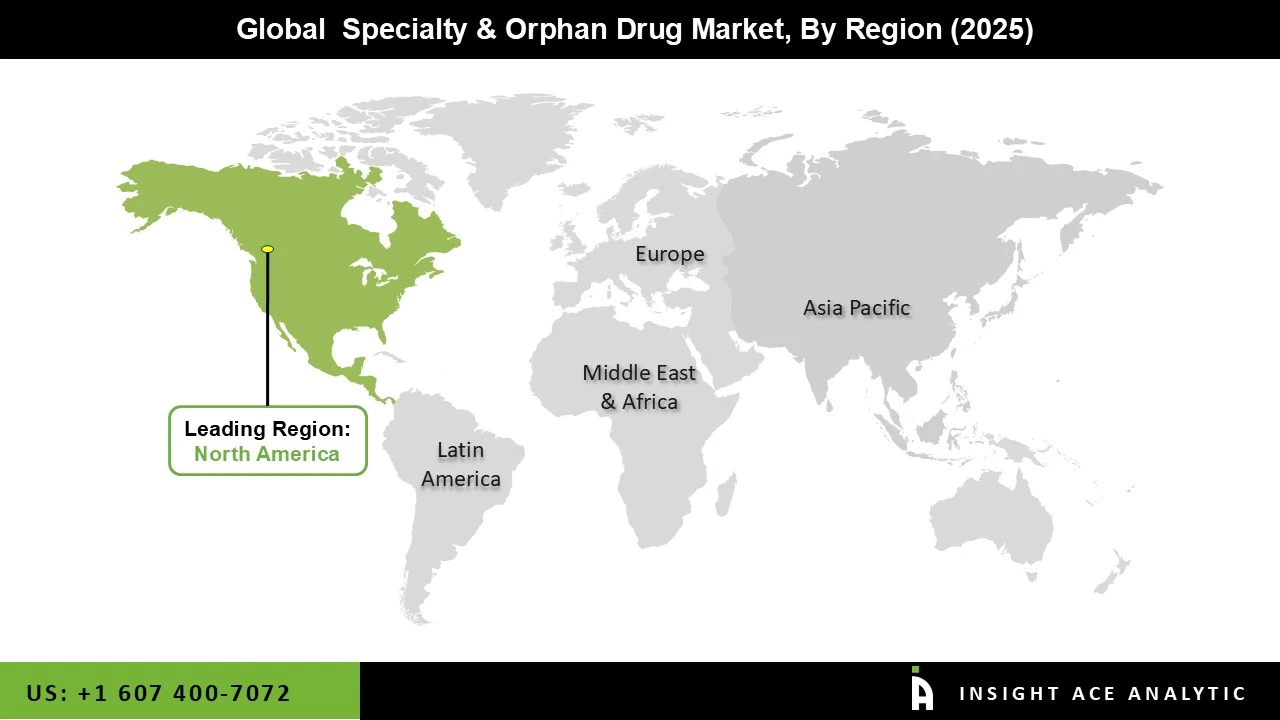
High healthcare spending and favourable reimbursement systems make access to costly speciality drugs easier for patients, while patient awareness and advocacy groups drive demand for innovative therapies. Additionally, well-established distribution networks, including speciality pharmacies and patient support programs, ensure therapies reach patients effectively. Combined, these factors innovation, supportive policies, advanced research, healthcare access, and efficient distribution, have solidified North America’s leadership in the Speciality and Orphan Drug Market, making it a hub for the development and adoption of rare disease treatments.
Key Developments
• In September 2025, Novartis entered into multiple strategic collaborations in CAR-T therapy, radiotherapy, and RNA-based drugs to target rare cancers and genetic disorders.
• In July 2025, Vertex Pharmaceuticals expanded into gene-editing by advancing CRISPR-based therapies for rare diseases, enhancing its specialty drug pipeline.
Speciality & Orphan Drug Market Report Scope:
| Report Attribute | Specifications |
| Growth Rate CAGR | CAGR of 11.6% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2021 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Therapeutic Areas, Drug Modality, Dosage Form, Distribution Channel and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Roche, Novartis, Pfizer, Johnson & Johnson, AstraZeneca, Merck & Co., Sanofi, Takeda Pharmaceutical, Bristol Myers Squibb, Eli Lilly and Company, Vertex Pharmaceuticals, BioMarin Pharmaceutical, Ultragenyx Pharmaceutical, argenx, Alnylam Pharmaceuticals, Ionis Pharmaceuticals, Sarepta Therapeutics, Blueprint Medicines, Incyte, Azurity Pharmaceuticals, Horizon Therapeutics, Chiesi Farmaceutici, Recordati Rare Diseases, Amryt Pharma, Supernus Pharmaceuticals, Collegium Pharmaceutical, Assertio Holding |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Speciality & Orphan Drug Market Segmentation:
Speciality & Orphan Drug Market by Therapeutic Areas -
- Oncology
-
- Rare Cancers
- Hematologic Malignancies
- Neurology & CNS Disorders
-
- SMA
- Epilepsy
- ALS
- Rare Neurogenetic
-
- Sickle Cell Disease
- Hemophilia
- Rare Anaemias
- Metabolic & Genetic Disorders
-
- Lysosomal Storage Disorders
-
-
- Pompe
- Gaucher
- Fabry
-
-
- Mitochondrial Diseases
- PKU & Urea Cycle Disorders
- Immunology & Autoimmune Rare Diseases
-
- Complement Disorders
- Primary Immunodeficiency
- Cardiovascular Rare Diseases
-
- ATTR Amyloidosis
- Pulmonary Arterial Hypertension
- Infectious Diseases (Rare / Speciality)
-
- Resistant infections
- HIV speciality segment
- Rare Pediatric Diseases (Cross-segment)
- Respiratory Rare Diseases (CF, rare ILDs)
- Endocrine & Rare Hormonal Disorders
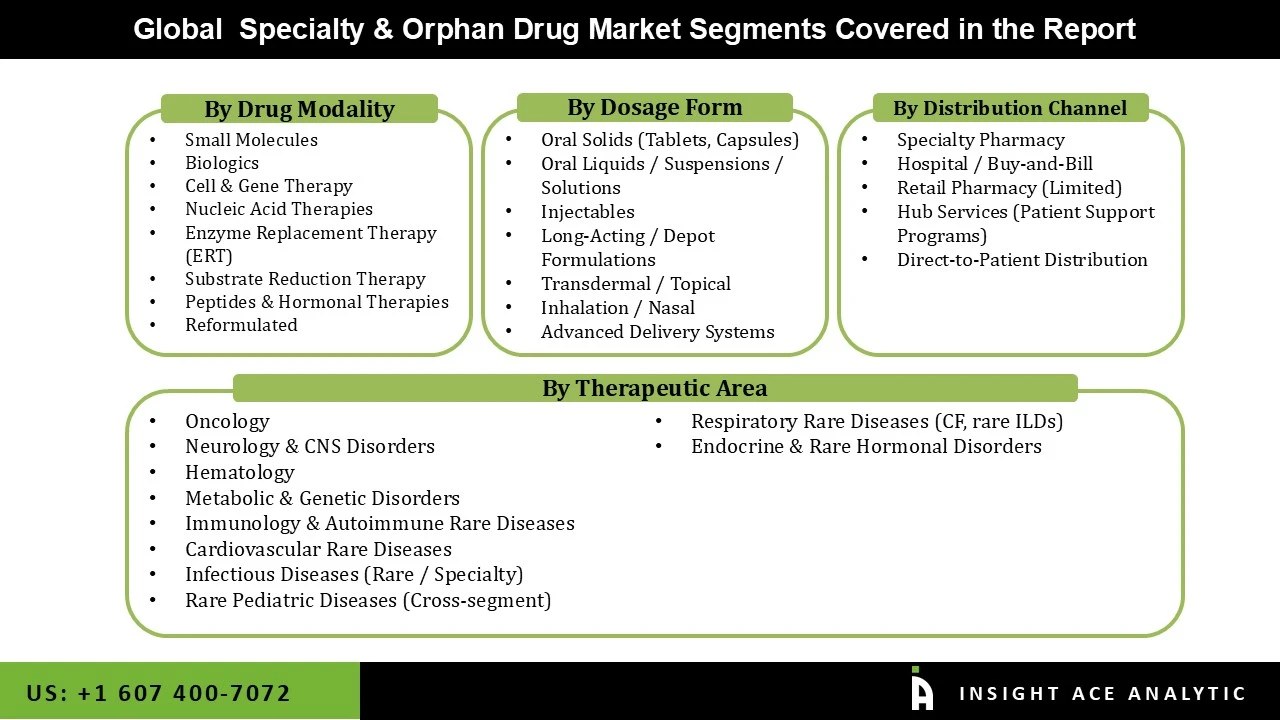
Speciality & Orphan Drug Market by Drug Modality-
- Small Molecules
- Biologics
- Monoclonal Antibodies
- Bispecific Antibodies
- ADCs
- Cell & Gene Therapy
- CAR-T
- AAV Gene Therapy
- CRISPR
- Nucleic Acid Therapies
- ASO
- siRNA
- mRNA
- Enzyme Replacement Therapy (ERT)
- Substrate Reduction Therapy
- Peptides & Hormonal Therapies
- Reformulated
Speciality & Orphan Drug Market by Dosage Form -
- Oral Solids (Tablets, Capsules)
- Oral Liquids / Suspensions / Solutions
- Injectables
- IV
- Subcutaneous
- IM
- Long Acting / Depot Formulations
- Transdermal / Topical
- Inhalation / Nasal
- Advanced Delivery Systems
-
- Implants
- Pumps
Speciality & Orphan Drug Market by Distribution Channel–
• Speciality Pharmacy
• Hospital / Buy-and-Bill
• Retail Pharmacy (Limited)
• Hub Services (Patient Support Programs)
• Direct-to-Patient Distribution
Speciality & Orphan Drug Market by Region-
North America-
• The US
• Canada
Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Speciality and Orphan Drug Market Size is predicted to reach grow at a 11.6% CAGR during the forecast period for 2026 to 2035.
Roche, Novartis, Pfizer, Johnson & Johnson, AstraZeneca, Merck & Co., Sanofi, Takeda Pharmaceutical, Bristol Myers Squibb, Eli Lilly and Company, Vertex Pharmaceuticals, BioMarin Pharmaceutical, Ultragenyx Pharmaceutical, argenx, Alnylam Pharmaceuticals, Ionis Pharmaceuticals, Sarepta Therapeutics, Blueprint Medicines, Incyte, Azurity Pharmaceuticals, Horizon Therapeutics, Chiesi Farmaceutici, Recordati Rare Diseases, Amryt Pharma, Supernus Pharmaceuticals, Collegium Pharmaceutical, Assertio Holding
Speciality and Orphan Drug Market is segmented into Therapeutic Areas, Drug Modality, Dosage Form, Distribution Channel and By Region.
North America region is leading the Speciality and Orphan Drug Market.