Industrial Microbiology Market Size and Revenue Impact Study 2026 to 2035
What is Industrial Microbiology Market Size?
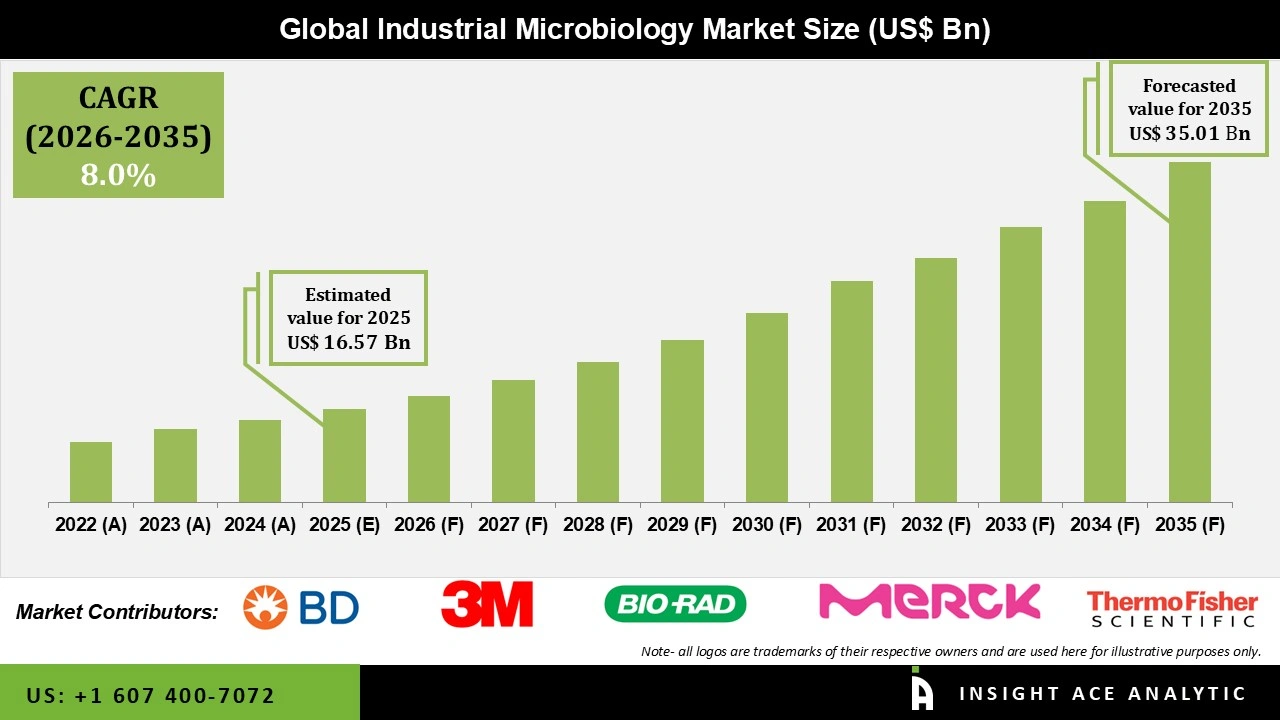
Global Industrial Microbiology Market Size is valued at USD 16.57 Bn in 2025 and is predicted to reach USD 35.01 Bn by the year 2035 at a 8.0% CAGR during the forecast period for 2026 to 2035.
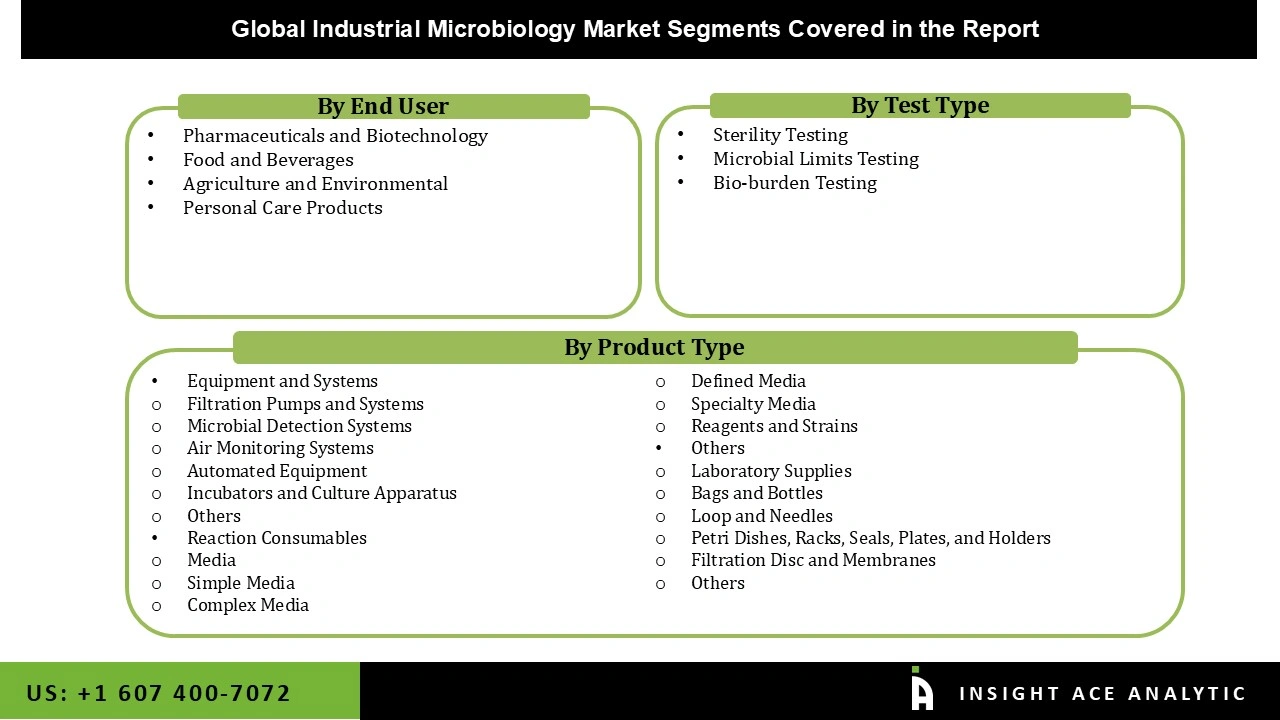
Industrial Microbiology Market Size, Share & Trends Analysis Distribution by Product Type (Reaction Consumables [Media, Defined Media, Complex Media, Simple Media, Specialty Media, Reagents and Strains], Equipment and Systems [Microbial Detection Systems, Filtration Pumps and Systems, Automated Equipment, Incubators and Culture Apparatus, Air Monitoring Systems, Others], and Others [Laboratory Supplies, Filtration Disc and Membranes, Loop and Needles, Bags and Bottles, Petri Dishes, Racks, Seals, Plates, and Holders, Others]), Test Type (Sterility Testing, Bio-burden Testing, and Microbial Limits Testing), End-user (Food and Beverages, Pharmaceuticals and Biotechnology, Personal Care Products, Agriculture and Environmental), By Region and Segment Forecasts, 2026 to 2035
Industrial Microbiology Market Key Takeaways:
|
Industrial microbiology, also known as microbial biotechnology, is the study of manipulating microbes for the benefit of industry, society, and the environment. These include naturally occurring organisms, genetically modified organisms (GMOs), and laboratory-selected mutants, which are utilised to produce large-scale industrial products. Because it uses renewable resources, industrial microbiology helps to replace extremely polluting chemical processes, fossil fuels, and non-biodegradable polymers with biocompatible alternatives. At the moment, it is widely used worldwide for the production of acetone and alcohols. The industrial microbiology market is anticipated to grow during the forecast period due to rising demand for microbiological testing and quality assurance across various sectors.
The use of fermentation in syrup formulations is growing in popularity, along with the rapidly expanding biotechnology and pharmaceutical sectors. The global industrial microbiology market will expand at an exponential rate as a result. The need for sterility testing methods in the pharmaceutical industry is growing as the industry expands and automation becomes increasingly prevalent. Furthermore, because of the increasing demand for consumer goods and the expanding global population, sterility testing for product safety is being used at an unprecedented rate. Additionally, the food and beverage industry's growing adoption of these testing methods is anticipated to support the expansion of the global industrial microbiology market over the projected period.
In the coming years, demand is anticipated to increase due to greater emphasis on industrial safety, food health and cleanliness. Even though there is a chance that antimicrobial drug resistance may increase, particularly in emerging nations such as China and India, the use of description antibiotics is predicted to continue encouraging the use of industrial microbiology. Moreover, the growing focus on process automation over conventional techniques is the main driver of the worldwide market's expansion since it would drastically reduce production costs and promote market growth. Furthermore, the majority of the total mass of pharmaceuticals consists of excipients. The global industrial microbiology market is seeing numerous profitable growth opportunities driven by the growing popularity of industrial applications such as excipients.
Competitive Landscape
Which are the Leading Players in Industrial Microbiology Market?
- Danaher Corporation
- Thermo Fisher Scientific Inc.
- BioMerieuxx SA
- Novamed
- Becton, Dickinson, and Company
- Asiagel Corporation
- QIAGEN
- Sartorius AG
- Bio-Rad Laboratories Inc.
- 3M
- Eppendorf AG
- Merck KGaA
- Others
Market Dynamics
Driver
Growing the Production of Pharmaceuticals
The industrial microbiology market is expected to develop in the future due to rising pharmaceutical production. The aging of the population, changes in regulations, and health crises that increase demand for pharmaceuticals and novel therapies are some of the reasons for the rise in pharmaceutical output. Microbial fermentation techniques are used in industrial microbiology to use microorganisms like bacteria, yeast, and fungi to mass-produce a range of medications, such as vaccines, insulin, and antibiotics such as penicillin. For instance, in April 2025, the Luxembourg-based government agency Eurostat reported that EU exports of pharmaceutical and medical goods reached $316.38 billion (€313.4 billion) in 2024, a 13.5% rise from 2023. Meanwhile, imports which totaled $112.7 billion (€119.7 billion)—only saw a slight 0.5% gain. Thus, rising pharmaceutical output will propel the industrial microbiology market's expansion.
Restrain/Challenge
High R&D Cost and Lack of Skilled Professionals
The industrial microbiology market's expansion is expected to be impeded by the high cost of research and development. Research & development activities demand a lump sum expenditure, which frequently prevents potential firms from entering the market, especially in middle- and lower-income nations. Furthermore, disputes over the use of genetically modified organisms in food sources could limit the growth of the global industrial microbiology market. Additionally, the industrial microbiology market is growing more slowly due to a growing shortage of qualified workers. Notwithstanding technological developments, many labs and production sites still have trouble hiring skilled microbiologists who know how to use complex equipment and decipher complex data. This lack of qualified personnel lowers operating efficiency and prevents the adoption of cutting-edge technologies.
Reaction Consumables Segment is Expected to Drive the Industrial Microbiology Market
The reaction consumables category held the largest share in the Industrial Microbiology market in 2025 because microbiological testing is becoming more and more necessary across a range of sectors. In order to support quality control procedures, reaction consumables—such as media, reagents, and test kits—are essential for microbial detection and analysis. Additionally, the segment for these consumables is probably going to be driven by the growing emphasis on product safety and regulatory compliance in industries including food, beverages, and medicines. Furthermore, it is projected that developments in microbiology methods that call for specific reagents will further expand the industrial microbiology market. The expansion of reaction consumables is also anticipated to be aided by the increasing use of automation in testing procedures.
Pharmaceuticals and Biotechnology Segment is Growing at the Highest Rate in the Industrial Microbiology Market
In 2025, the pharmaceuticals and biotechnology category dominated the Industrial Microbiology market because of the increased emphasis on quality control and microbiological testing in drug development and production. The demand for microbiological testing in the pharmaceutical, vaccine, and biologics sectors is expected to increase due to their increased production. The demand for industrial microbiology solutions is further increased by regulatory agencies' stringent requirements for contamination monitoring and microbiological limits testing in pharmaceutical manufacture. Additionally, the growth in the industrial microbiology market is anticipated to be driven by developments in biopharmaceutical research as well as the growing complexity of biologic medication formulations. The growth of this segment is further supported by the growing global pharmaceutical sector and increased investments in biotechnology research.
Why North America Led the Industrial Microbiology Market?
The Industrial Microbiology market was dominated by North America region in 2025, particularly the United States, driven by a concentration on biotechnology and medicines, robust healthcare infrastructure, and technological developments. With many pharmaceutical businesses, biotechnology corporations, and research institutions using industrial microbiology to improve manufacturing processes and create new products, the United States is a major player in this sector.
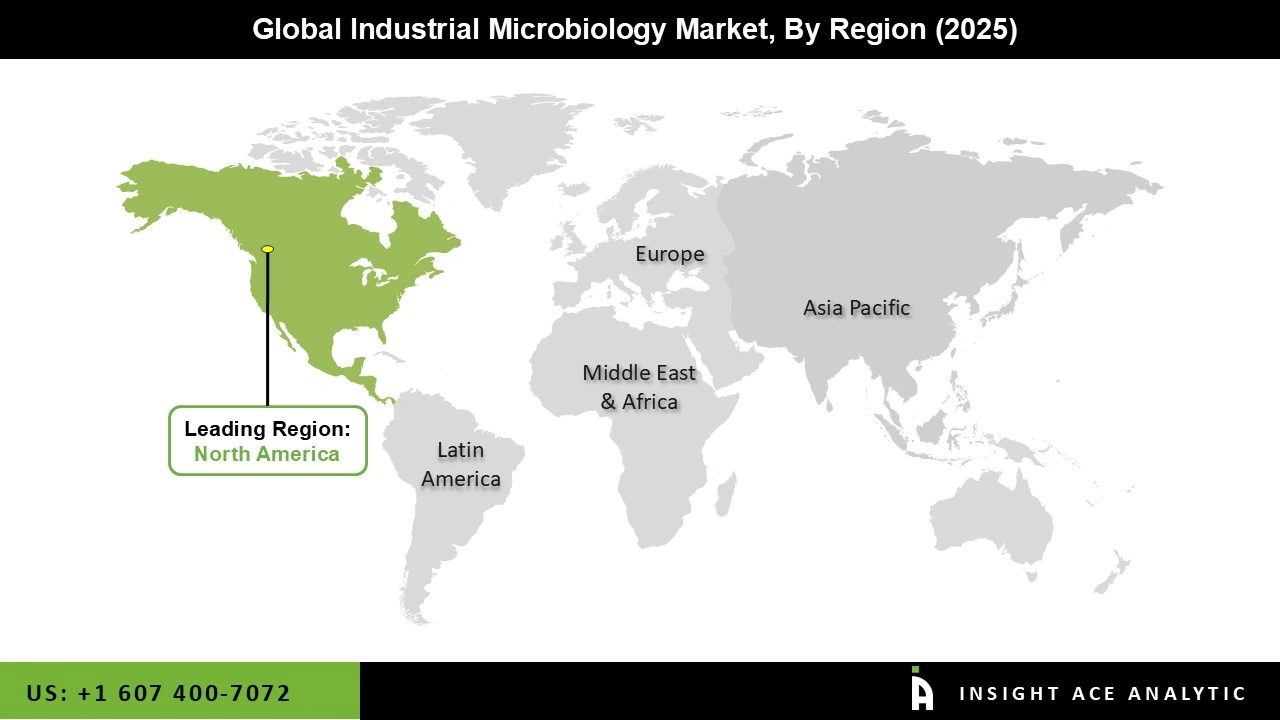
The industrial microbiology market is expanding quickly as a result of the region's investments in R&D, especially in the biotechnology and pharmaceutical industries. Furthermore, the strong regulatory framework in North America guarantees that businesses follow strict guidelines for product sustainability, quality, and safety, which in turn fuels the market for microbiological solutions. Additionally, the market in North America is being driven by the growing emphasis on biopharmaceuticals, personalized treatment, and sustainable food production. With substantial investments in both technology and research, the region is therefore anticipated to maintain its position as the market leader for industrial microbiology.
Key Development:
• January 2025: The first unit under the American Society for Microbiology's (ASM) new strategic roadmap, the ASM Health Scientific Unit, was revealed. An essential part of ASM's strategy for using microbial science to address important health challenges is ASM Health. ASM Health's structure and leadership will enable the health-related microbial sciences community to influence the field's future, guaranteeing societal impact and encouraging international cooperation.
• December 2024: Acies Bio, a leader in microbial biotechnology, and LG Chem, a pioneer in chemical production, partnered to create sustainable solutions for commercial biomanufacturing. The collaboration makes use of Acies Bio's OneCarbonBioTM microbial platform for novel biotechnology-based product techniques. The objective is to create industrial chemical production technology that is commercially scalable.
• May 2024: Merck KGaA established a new M LabTM Collaboration Center in Shanghai with the goal of assisting Chinese biologics producers by providing advanced microbiology training and technical services for aseptic processing facilities.
• June 2023: For both academic and commercial uses, Merck India announced the opening of a Microbiology Application Training Lab (MAT Lab) in Bangalore. The Lab encourages biopharma and pharmaceutical companies to work with Merck to build microbial analysis skills.
Industrial Microbiology Market Report Scope :
| Report Attribute | Specifications |
| Market size value in 2025 | USD 16.57 Bn |
| Revenue forecast in 2035 | USD 35.01 Bn |
| Growth Rate CAGR | CAGR of 8.0% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Test Type, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Danaher Corporation, Thermo Fisher Scientific Inc., BioMerieuxx SA, Novamed, Becton, Dickinson, and Company, Asiagel Corporation, QIAGEN, Sartorius AG, Bio-Rad Laboratories Inc., 3M, Eppendorf AG, Merck KGaA, and Others |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of Industrial Microbiology Market :
Industrial Microbiology Market by Product Type-
- Reaction Consumables
- Media
- Defined Media
- Complex Media
- Simple Media
- Specialty Media
- Reagents and Strains
- Equipment and Systems
- Microbial Detection Systems
- Filtration Pumps and Systems
- Automated Equipment
- Incubators and Culture Apparatus
- Air Monitoring Systems
- Others
- Others
- Laboratory Supplies
- Filtration Disc and Membranes
- Loop and Needles
- Bags and Bottles
- Petri Dishes, Racks, Seals, Plates, and Holders
- Others
Industrial Microbiology Market by Test Type-
- Sterility Testing
- Bio-burden Testing
- Microbial Limits Testing
Industrial Microbiology Market by End-user-
- Food and Beverages
- Pharmaceuticals and Biotechnology
- Personal Care Products
- Agriculture and Environmental
Industrial Microbiology Market By Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Industrial Microbiology Market Size is valued at USD 16.57 Bn in 2025 and is predicted to reach USD 35.01 Bn by the year 2035
Industrial Microbiology Market is expected to grow at a 8.0% CAGR during the forecast period for 2026 to 2035
Danaher Corporation, Thermo Fisher Scientific Inc., BioMerieuxx SA, Novamed, Becton, Dickinson, and Company, Asiagel Corporation, QIAGEN, Sartorius AG, Bio-Rad Laboratories Inc., 3M, Eppendorf AG, Merck KGaA, and Others
Industrial Microbiology Market is segmented into y Product Type (Reaction Consumables [Media, Defined Media, Complex Media, Simple Media, Specialty Media, Reagents and Strains], Equipment and Systems [Microbial Detection Systems, Filtration Pumps and Systems, Automated Equipment, Incubators and Culture Apparatus, Air Monitoring Systems, Others], and Others [Laboratory Supplies, Filtration Disc and Membranes, Loop and Needles, Bags and Bottles, Petri Dishes, Racks, Seals, Plates, and Holders, Others]), Test Type (Sterility Testing, Bio-burden Testing, and Microbial Limits Testing), End-user (Food and Beverages, Pharmaceuticals and Biotechnology, Personal Care Products, Agriculture and Environmental), and Other.
North America region is leading the Industrial Microbiology Market.