Semaglutide Biosimilar and Generic Market Size, Revenue, Trend Report 2026 to 2035
What is Semaglutide Biosimilar and Generic Market Size?
Global Semaglutide Biosimilar and Generic Market is projected to grow rapidly with a CAGR of 22.1% during the forecast period for 2026 to 2035.
Semaglutide Biosimilar and Generic Market Size, Share and Trends Analysis Distribution by Product Type (Semaglutide Injectable Generic (Subcutaneous), Semaglutide Oral Tablet Generic, Semaglutide Biosimilar (Biologic Pathway), Semaglutide Peptide API (Merchant Supply), Co-Formulated and Fixed-Dose Combination Generics), By Dosage Form (Disposable Multi-Dose Prefilled Pen, Single-Use Prefilled Autoinjector, Multi-Dose Vials, Single-Dose Vials, Prefilled Cartridges for Reusable Pens, Oral Tablets (Immediate Release)), By Dose Strength (Subcutaneous Injection, Oral Tablet) and Segment Forecasts, 2026 to 2035.
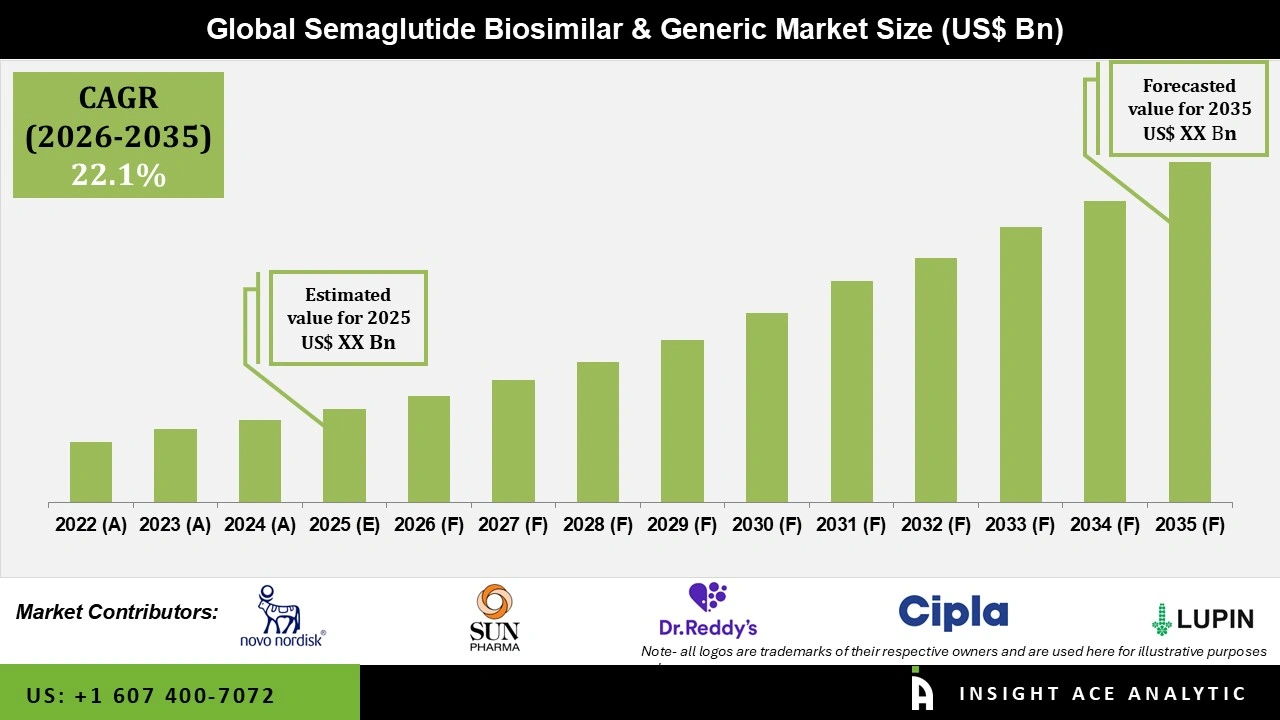
Semaglutide biosimilar and generic versions are positioned as more affordable alternatives to branded therapies such as Ozempic, Wegovy, and Rybelsus, which belong to the class of GLP-1 receptor agonists. These therapies mimic incretin hormones, helping regulate blood glucose levels by enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and reducing appetite—making them highly effective for both glycemic control and weight management. Biosimilars are designed to closely match the reference biologic in terms of safety, efficacy, and quality, following rigorous regulatory pathways and comparability studies. In contrast, the concept of generics in semaglutide is more complex due to its peptide-based nature, but advancements in peptide synthesis and manufacturing are enabling the development of cost-effective alternatives. As patents for originator products approach expiry in key markets, increased competition from biosimilars and follow-on versions is expected to significantly improve patient access and affordability.
The semaglutide biosimilars and generics market comprises all the processes relating to the production and marketing of these treatments as drugs to be used by patients. The drug is mainly utilized in the treatment of type two diabetes and obesity, along with the minimization of the risk of any cardiovascular problems associated with them. Its application extends to the hospital environment, specialized clinics, and even in patients' homes, through means of administration that include injectors and pills.
Competitive Landscape
Which are the Leading Players in the Semaglutide Biosimilar and Generic Market?
Originator / Reference Listed Drug
- Novo Nordisk A/S (Ozempic, Wegovy, Rybelsus)
Indian Generic and Peptide Leaders
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Lupin Limited
- Biocon Limited
- Zydus Lifesciences
- Aurobindo Pharma
- Natco Pharma
- Glenmark Pharmaceuticals
- Torrent Pharmaceuticals
- Alkem Laboratories
- Mankind Pharma
- Hetero Drugs
- Alembic Pharmaceuticals
- Emcure Pharmaceuticals
Chinese Generic and Biosimilar Players
- Jiangsu Hengrui Pharmaceuticals
- Hansoh Pharmaceutical Group
- Sciwind Biosciences
- Chia Tai Tianqing (CTTQ) Pharmaceutical
- Huadong Medicine
- The United Laboratories International
- Shanghai Fosun Pharmaceutical
- Shanghai Benemae Pharmaceutical
- Innovent Biologics
- Changchun High and New Technology (GenSci)
- Livzon Pharmaceutical Group
Global Generic and Biosimilar Majors
- Teva Pharmaceutical Industries
- Sandoz Group AG
- Viatris Inc.
- Hikma Pharmaceuticals
- Fresenius Kabi
- Celltrion Inc.
- Samsung Bioepis
- Dr. Reddy's Biologics Division
Latin American, Middle Eastern and Emerging-Market Players
- EMS Pharma (Brazil)
- Eurofarma Laboratórios (Brazil)
- Libbs Farmacêutica (Brazil)
- Laboratorios Bagó (Argentina)
- Grupo Insud / Chemo (Argentina)
- Abdi Ibrahim (Turkey)
- Deva Holding (Turkey)
- Polfa Tarchomin (Poland)
- Aspen Pharmacare (South Africa)
- Julphar (UAE)
- Peptide API and Contract Manufacturers
- Bachem Holding AG
- PolyPeptide Group
- Corden Pharma International
- Lonza Group
- Piramal Pharma Solutions
- Ambiopharm Inc.
- Hangzhou Jiuyuan Gene Engineering
- Chengdu Shengnuo Biotech
- Auro Peptides (Aurobindo)
- Senn Chemicals (Bachem)
- ChinaPeptides
Compounding Pharmacy and Outsourcing Facilities
- Empower Pharmacy
- Hallandale Pharmacy
- Olympia Compounding Pharmacy
- Red Rock Pharmacy
- Strive Pharmacy
- Belmar Pharma Solutions
Market Dynamics
Driver
Patent Expiry and Demand for Affordable Alternatives
The upcoming expiration of patents for branded semaglutide products such as Ozempic and Wegovy drives the demand in the semaglutide biosimilar and generics market. As patents expire, this creates opportunities for various companies to manufacture cheaper biosimilars, resulting in intense competition, thus significantly lowering the cost of treatment. This trend is especially significant in price-sensitive or developing nations due to the expensive nature of branded treatments. Therefore, the rising need for affordable solutions is fueling the demand for semaglutide biosimilars and penetrating the market.
Restrain/Challenge
Complex Manufacturing and Regulatory Challenges
One limitation that affects the entry of biosimilar and generic semaglutide into the market is its intricate nature in terms of development and regulatory process. Semaglutide is a biologic medication derived from peptides, falling under the GLP-1 receptor agonist class. This necessitates sophisticated manufacturing processes and quality controls. The creation of biosimilar drugs requires rigorous testing, analysis, and clinical trials to prove their similarity in terms of safety and efficacy to the original brand, such as Ozempic. The strict regulatory requirements in different jurisdictions add to the expense and time involved in product development.
Semaglutide Injectable Generic (Subcutaneous) Segment is Expected to Drive the Semaglutide Biosimilar and Generic Market
The segment which is leading the market is Semaglutide Injectable Generic (Subcutaneous). This can be attributed to the successful implementation and high efficacy that comes with injectables like Ozempic and Wegovy in dealing with Type 2 Diabetes and Obesity. The main benefits associated with this mode of treatment include increased availability of drugs, consistent results from the therapy, and the preference by most medical practitioners. Additionally, there are more injectable biosimilars and generics being developed by drug manufacturers because of the ease in their regulatory pathway and delivery system through prefilled pens.
Disposable Multi-Dose Prefilled Pen Segment is Growing at the Highest Rate in the Semaglutide Biosimilar and Generic Market
The disposable multi-dose prefilled pen sub-segment will fuel the biosimilars market growth due to its high compatibility with existing treatment processes and convenience. Being an integral part of popular drugs such as Ozempic and Wegovy, disposable multi-dose prefilled pens provide ease of administration, accuracy of dosing, and improved compliance for diseases including Type 2 Diabetes and obesity. The convenient nature of the pen device decreases the dependence on healthcare professionals while preventing any dosing mistake, thus providing maximum preference from the side of end-users. Another reason why producers are focusing on the development of pens includes their familiarity with the technology used for manufacturing these devices.
Why North America Led the Semaglutide Biosimilar and Generic Market?
North America has dominated the market for semaglutide biosimilars and generics due to a high number of cases of Type 2 Diabetes and Obesity coupled with higher levels of awareness and adoption of new treatments. The extensive use of branded semaglutide drugs like Ozempic and Wegovy has provided a platform for the acceptance of biosimilars in the future.
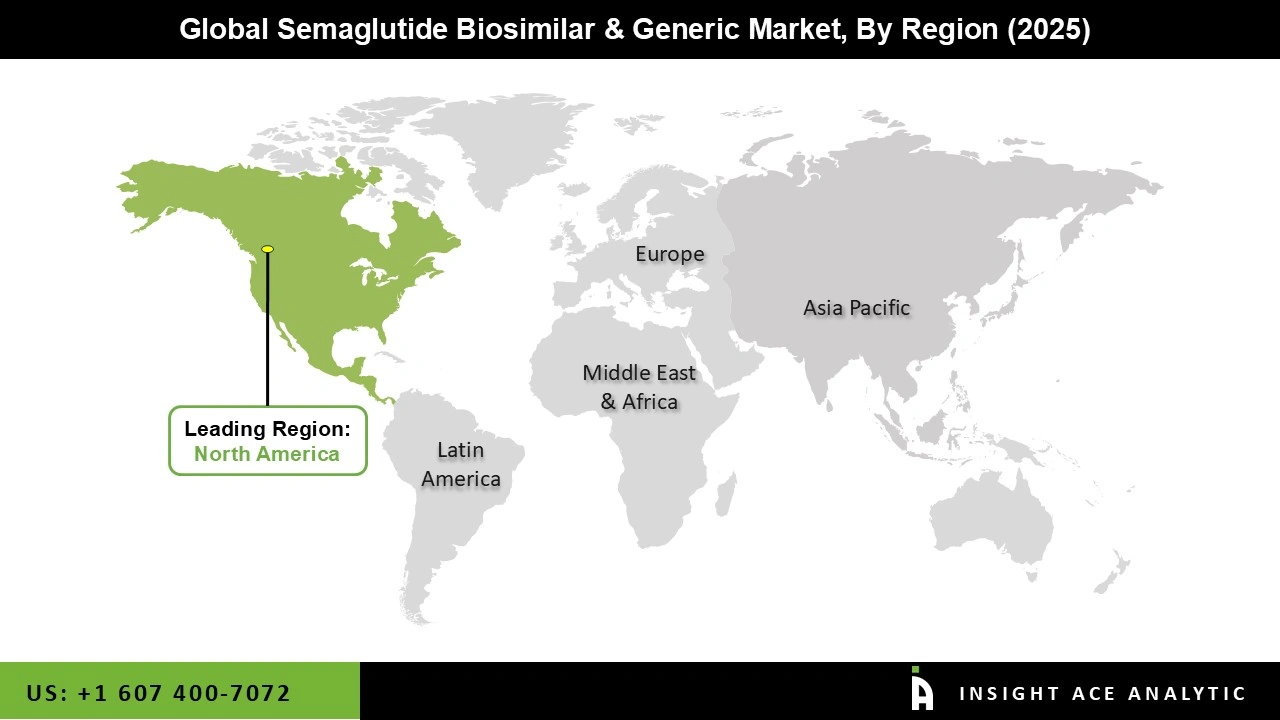
Furthermore, the presence of a robust healthcare system, favorable reimbursement schemes, and leading pharmaceutical companies is expected to boost growth in the market. Growing demand for low-cost solutions and patents expiration will contribute to this dominance in the future.
Key Development :
• In Dec 2025, Novo Nordisk announced that the FDA approved the Wegovy® pill (once-daily oral semaglutide 25 mg) to reduce excess body weight and maintain weight reduction long term, and to reduce the risk of major adverse cardiovascular events the first oral GLP-1 receptor agonist therapy approved for weight management.
Semaglutide Biosimilar and Generic Market Report Scope:
| Report Attribute | Specifications |
| Growth Rate CAGR | CAGR of 22.1% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Dosage Form, Dose Strength, Manufacturing Route, Indication, Regulatory Pathway, Distribution Channel, End User / Prescriber Setting, Pricing and Access, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East and Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Sandoz Group AG, Viatris Inc., Hikma Pharmaceuticals, Fresenius Kabi, Celltrion Inc., Samsung Bioepis, Dr. Reddy's Biologics Division, EMS Pharma (Brazil), Eurofarma Laboratórios (Brazil), Libbs Farmacêutica (Brazil), Laboratorios Bagó (Argentina), Grupo Insud / Chemo (Argentina), Abdi Ibrahim (Turkey), Deva Holding (Turkey), Polfa Tarchomin (Poland), Aspen Pharmacare (South Africa), Julphar (UAE), Peptide API and Contract Manufacturers, Bachem Holding AG, PolyPeptide Group, Corden Pharma International, Lonza Group, Piramal Pharma Solutions, Ambiopharm Inc., Hangzhou Jiuyuan Gene Engineering, Chengdu Shengnuo Biotech, Auro Peptides (Aurobindo), Senn Chemicals (Bachem), ChinaPeptides, Empower Pharmacy, Hallandale Pharmacy, Olympia Compounding Pharmacy, Red Rock Pharmacy, Strive Pharmacy, Belmar Pharma Solutions |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentations of Semaglutide Biosimilar and Generic Market:
Semaglutide Biosimilar and Generic Market, by Product Type -
- Semaglutide Injectable Generic (Subcutaneous)
- Semaglutide Oral Tablet Generic
- Semaglutide Biosimilar (Biologic Pathway)
- Semaglutide Peptide API (Merchant Supply)
- Co-Formulated and Fixed-Dose Combination Generics
Semaglutide Biosimilar and Generic Market, by Dosage Form -
- Disposable Multi-Dose Prefilled Pen
- Single-Use Prefilled Autoinjector
- Multi-Dose Vials
- Single-Dose Vials
- Prefilled Cartridges for Reusable Pens
- Oral Tablets (Immediate Release)
Semaglutide Biosimilar and Generic Market, by Dose Strength -
- Subcutaneous Injection
- 0.25 mg (Initiation)
- 0.5 mg
- 1.0 mg
- 1.7 mg
- 2.0 mg
- 2.4 mg (Obesity Indication)
- Oral Tablet
- 3 mg
- 7 mg
- 14 mg
- 25 mg (High-Dose Obesity)
Semaglutide Biosimilar and Generic Market, by Manufacturing Route -
- Recombinant Fermentation (Yeast rDNA-Based)
- Solid-Phase Peptide Synthesis (SPPS)
- Liquid-Phase Peptide Synthesis (LPPS)
- Hybrid SPPS + LPPS
- Continuous Flow Peptide Synthesis
- Enzymatic and Chemo-Enzymatic Synthesis
Semaglutide Biosimilar and Generic Market, by Indication -
- Type 2 Diabetes Mellitus
- Chronic Weight Management / Obesity
- Cardiovascular Risk Reduction
- Chronic Kidney Disease
- MASH / Metabolic Dysfunction-Associated Steatohepatitis
- Obstructive Sleep Apnea (Off-Label / Label Expansion)
- Peripheral Artery Disease (Label Expansion)
Semaglutide Biosimilar and Generic Market, by Regulatory Pathway -
- Generic Approval (ANDA / Equivalent)
- Biosimilar Approval (351(k) / EMA Biosimilar)
- Hybrid / New Drug Application (NDA / Art. 10(3))
- Compounded Preparation (503A Pharmacy)
- Compounded Preparation (503B Outsourcing Facility)
- Emerging Market Bioequivalence Pathway
Semaglutide Biosimilar and Generic Market, by Distribution Channel -
- Retail Pharmacy
- Hospital and Institutional Pharmacy
- Specialty Pharmacy
- Manufacturer-Direct Digital Pharmacy
- Telehealth and DTP Platforms
- Mail-Order and Online Pharmacy
- Government and Public Tender
- Compounding Pharmacy Channel
Semaglutide Biosimilar and Generic Market, by End User / Prescriber Setting -
- Endocrinology Practices
- Primary Care and Family Medicine
- Obesity Medicine and Weight Management Clinics
- Diabetology and Diabetes Specialty Centers
- Hospitals and IDNs
- Telehealth Prescribers
- Homecare / Self-Administration
Semaglutide Biosimilar and Generic Market, by Pricing and Access -
- Branded-Originator Reference Price
- Generic Cash-Pay Transparent Pricing
- Biosimilar Tendered / Public Payer Pricing
- Private Insurance Formulary
- Government Negotiated (BALANCE / TrumpRx / CMS)
- Emerging-Market Out-of-Pocket
- Compounded Preparation Pricing
Semaglutide Biosimilar and Generic Market, by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East and Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Semaglutide Biosimilar and Generic Market is projected to grow rapidly with a CAGR of 22.1% during the forecast period for 2026 to 2035.
Sandoz Group AG, Viatris Inc., Hikma Pharmaceuticals, Fresenius Kabi, Celltrion Inc., Samsung Bioepis, Dr. Reddy\'s Biologics Division, EMS Pharma (Brazil), Eurofarma Laboratórios (Brazil), Libbs Farmacêutica (Brazil), Laboratorios Bagó (Argentina), Grupo Insud / Chemo (Argentina), Abdi Ibrahim (Turkey), Deva Holding (Turkey), Polfa Tarchomin (Poland), Aspen Pharmacare (South Africa), Julphar (UAE), Peptide API and Contract Manufacturers, Bachem Holding AG, PolyPeptide Group, Corden Pharma International, Lonza Group, Piramal Pharma Solutions, Ambiopharm Inc., Hangzhou Jiuyuan Gene Engineering, Chengdu Shengnuo Biotech, Auro Peptides (Aurobindo), Senn Chemicals (Bachem), ChinaPeptides, Empower Pharmacy, Hallandale Pharmacy, Olympia Compounding Pharmacy, Red Rock Pharmacy, Strive Pharmacy, Belmar Pharma Solutions and Others.
Semaglutide Biosimilar and Generic Market is segmented into Product Type, Dosage Form, Dose Strength, Manufacturing Route, Indication, Regulatory Pathway, Distribution Channel, End User / Prescriber Setting, Pricing and Access, and Other.
North America region is leading the Semaglutide Biosimilar and Generic Market.
