DNA Read Write and Edit Market Size, Trend, Forecast Report 2026 to 2035
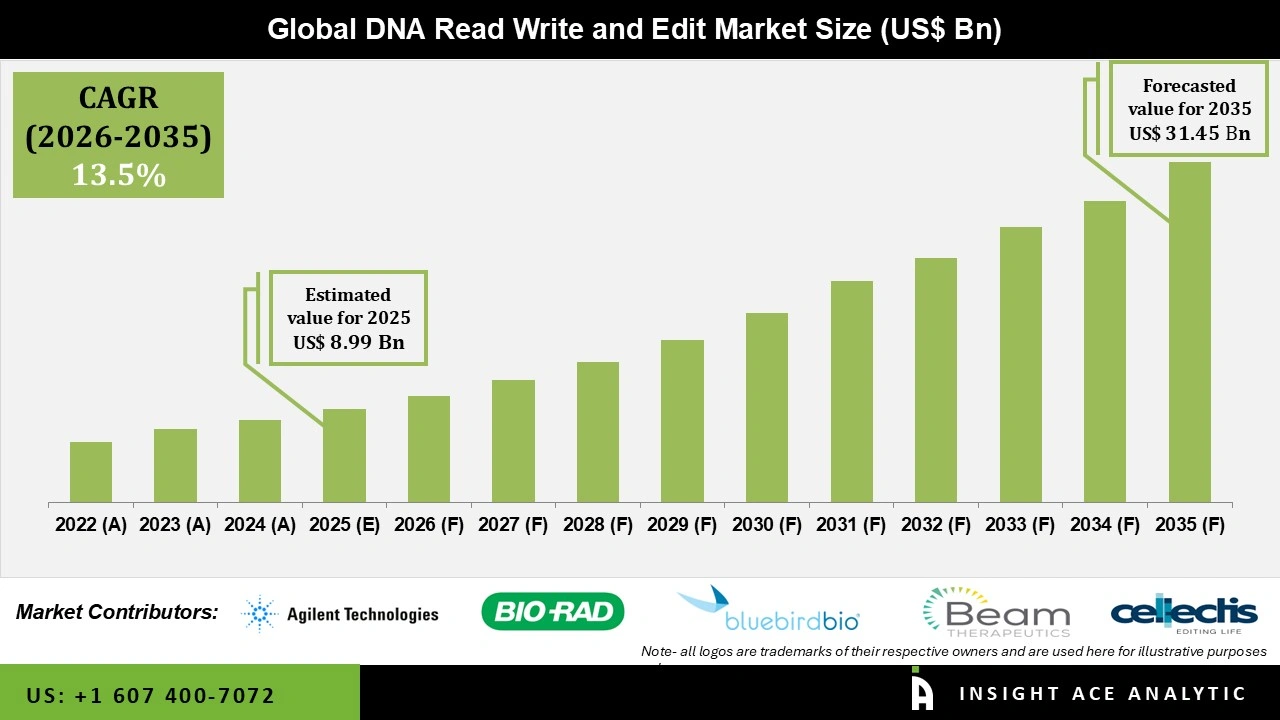
Global DNA Read Write and Edit Market Size is valued at USD 8.99 Bn in 2025 and is predicted to reach USD 31.45 Bn by the year 2035 at a 13.5% CAGR during the forecast period for 2026 to 2035.
DNA Read Write and Edit Market Size, Share & Trends Analysis Distribution by Product Type (Kits and Reagents, Animal Models, Cell Line Engineering, and Services), Delivery Mode (Microinjection, Electroporation, Viral Vectors, and Non-Viral Vectors), Targeted Organism (Humans, Plants, and Animals), Application (Gene Therapy, Agricultural, Diagnostics, Synthetic Biology, and Biotechnology), Technology (CRISPR-Cas Systems, TALEN’s, Base Editors, and ZFN’s), End-user (Pharmaceutical and Biotechnology Companies, Clinical Research Organizations, Academic and Research Institutes, Others), and Segment Forecasts, 2026 to 2035.
The tools that enable scientists to examine, create, and alter genetic material are referred to as DNA read, write, and edit. Researchers can better interpret genetic information and find mutations associated with diseases by using sequencing methods to determine the precise order of nucleotides (A, T, C, and G) in a DNA molecule. The capacity to chemically synthesize or create new DNA sequences in labs is known as "DNA writing," and it makes it possible to create synthetic genes or complete genomes for use in biotechnology and research. Rapid developments in genetic engineering, rising funding for biotechnology research, and the growing incidence of genetic illnesses that call for novel treatment approaches are all factors contributing to the DNA read write and edit market's expansion.
The DNA read write and edit market is expanding due to the quick development of genomics and synthetic biology, which allow scientists to accurately alter genes for scientific and medical purposes, synthesize DNA sequences, and analyze genetic data. The DNA read write and edit market is expanding more quickly due to rising investments in gene editing tools, including CRISPR-based technologies, as well as the rising need for better diagnostics and personalized therapeutics. Additionally, the biotechnology and pharmaceutical industries are adopting DNA sequencing and gene editing solutions due to the growing prevalence of genetic illnesses and the need for novel treatment options. The DNA read write and edit market is therefore being greatly aided by the growing uses of genomic research in industrial biotechnology, healthcare, and agriculture.
Furthermore, the future of DNA read write and edit market is anticipated to be shaped by ongoing technological advancements in genome editing platforms, automated DNA synthesis, and next-generation sequencing. To increase productivity in genetic research and therapeutic applications, businesses and academic institutions are concentrating on creating genomic tools that are quicker, more precise, and more affordable. Advances in genetic engineering and synthetic biology are also being accelerated by the growing cooperation between government agencies, biotechnology firms, and academic institutions. Moreover, the DNA read write and edit market outlook is being strengthened by the increasing recognition of precision medicine and the incorporation of AI in genomic analysis. Thus, the market for DNA read write and edit is anticipated to increase significantly as demand for cutting-edge genomic technologies keeps rising.
Competitive Landscape
Which are the Leading Players in DNA Read Write and Edit Market?
- Agilent Technologies
- Editas Medicine
- GenScript Biotech
- Beam Therapeutics
- Cellectis
- CRISPR Therapeutics
- Danaher
- Illumina
- Bluebird Bio
- Oxford Nanopore Technologies
- QIAGEN
- Roche
- Sangamo Therapeutics
- Pacific Biosciences
- PerkinElmer (through acquisition of Horizon Discovery)
- Thermo Fisher Scientific
- Intellia Therapeutics
- Bio-Rad Laboratories
- Twist Bioscience
Market Dynamics
Driver
Increased Demand in Precision Medicine
The DNA read write and edit market is mostly driven by the growing need for personalized healthcare and precision medicine. Due to the developments in genomic technologies, medical professionals and researchers may now evaluate a person's genetic profile and create personalized treatments. For diseases like cancer, uncommon genetic abnormalities, and inherited diseases, early diagnosis and the creation of tailored therapeutics are made possible by the identification of disease-causing mutations by DNA sequencing and editing techniques. Research on gene therapy and regenerative medicine has accelerated due to technologies like CRISPR-based gene editing. Furthermore, the increasing use of genomic research by biotechnology and pharmaceutical businesses to create novel medications is propelling investment and growth in DNA read, write, and edit technologies throughout the world's healthcare industry.
Restrain/Challenge
Increasing Regulatory and Ethical Issues
The ethical issues and stringent legal frameworks pertaining to genetic modification are a major barrier to the DNA read, write, and edit business. There are serious ethical concerns about safety, long-term repercussions, and potential abuse when editing human genes, particularly in germline cells that can pass on alterations to future generations. Strict rules and approval procedures for gene editing research and clinical applications are enforced by governments and regulatory organizations in numerous nations. These rules may raise research expenses, impede the development of new products, and postpone the commercialization of innovative technologies. In addition, the broad use of these technologies in clinical practice is limited by worries about unintentional genetic mutations or off-target consequences during DNA editing, which present extra difficulties for researchers and medical professionals.
Gene Therapy Segment is Expected to Drive the DNA Read Write and Edit Market
The Gene Therapy category held the largest share in the DNA Read Write and Edit market in 2025. One of the most promising uses of DNA read, write, and edit technology is gene therapy. Gene therapy has the potential to treat a variety of genetic illnesses, such as hemophilia, cystic fibrosis, and some types of cancer, by enabling the replacement or correction of damaged genes. The need for sophisticated genetic editing technology is being driven by the growing number of clinical trials and regulatory approvals for gene treatments. Furthermore, it is anticipated that continued research and development in this area will result in the identification of novel therapeutic targets and the creation of more potent therapies.
Pharmaceutical and Biotechnology Companies Segment is Growing at the Highest Rate in the DNA Read Write and Edit Market
In 2025, the Pharmaceutical and Biotechnology Companies category dominated the DNA Read Write and Edit market. These businesses have made significant investments in the creation of novel medications and treatments, and cutting-edge genetic editing technologies are essential to their R&D endeavors. These technologies are propelling innovation in the pharmaceutical and biotechnology sectors by making it possible to identify and validate new drug targets, create more accurate disease models, and find novel therapeutic agents. Moreover, the growing need for DNA read, write, and edit technologies in this industry is being further driven by the increased emphasis on precision medicine and the rising funding for biotechnology research.
Why North America Led the DNA Read Write and Edit Market?
The DNA Read Write and Edit market was dominated by North America region in 2025 due to developments in gene-manipulating technologies such as CRISPR and the growing need for customized therapeutic medications, especially in cancer and neurological targeted therapies. Additionally, this area has a strong R&D environment and well-developed infrastructure, which supports the DNA read write and edit market expansion.
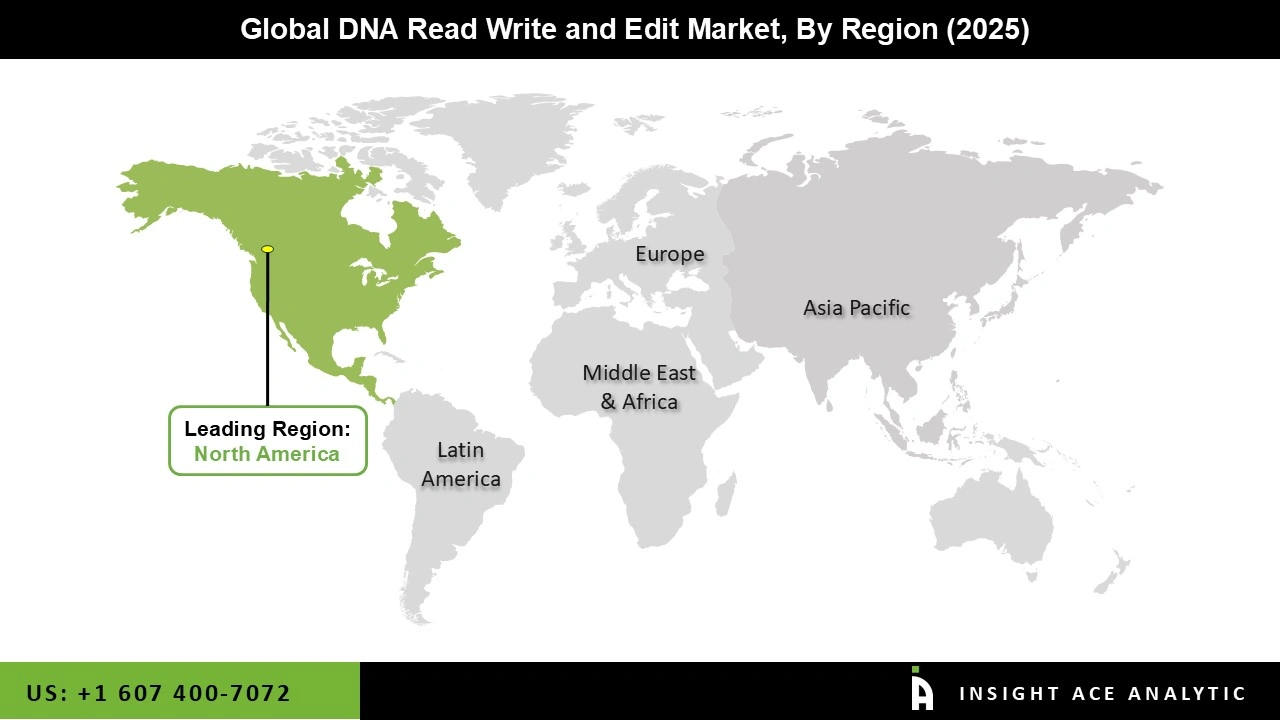
The US is one of the emerging development nations in North America in the relevant market. The US has been experiencing significant growth as a result of contributing factors such as technological advancements like CRISPR/Cas9, the US FDA's approval of new therapies such as Casgevy and Lyfgenia, and the rising demand for synthetic DNA services used in rare diseases.
Key Developments:
• June 2025: Editas Medicine and Synlogic announced a partnership to develop CRISPR-based treatments for metabolic disorders by combining Synlogic's synthetic biology techniques with Editas Medicine's gene editing capabilities.
DNA Read Write and Edit Market Report Scope :
| Report Attribute | Specifications |
| Market size value in 2025 | USD 8.99 Bn |
| Revenue forecast in 2035 | USD 31.45 Bn |
| Growth Rate CAGR | CAGR of 13.5% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026 to 2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Delivery Mode, Targeted Organism, Application, Technology, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Agilent Technologies, Editas Medicine, GenScript Biotech, Beam Therapeutics, Cellectis, CRISPR Therapeutics, Danaher, Illumina, Bluebird Bio, Oxford Nanopore Technologies, QIAGEN, Roche, Sangamo Therapeutics, Pacific Biosciences, PerkinElmer (through acquisition of Horizon Discovery), Thermo Fisher Scientific, Intellia Therapeutics, Bio-Rad Laboratories, and Twist Bioscience. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentations of DNA Read Write and Edit Market:
DNA Read Write and Edit Market by Product Type-
- Kits and Reagents
- Animal Models
- Cell Line Engineering
- Services
DNA Read Write and Edit Market by Delivery Mode-
- Microinjection
- Electroporation
- Viral Vectors
- Non-Viral Vectors
DNA Read Write and Edit Market by Targeted Organism-
- Humans
- Plants
- Animals
DNA Read Write and Edit Market by Application-
- Gene Therapy
- Agricultural
- Diagnostics
- Synthetic Biology
- Biotechnology
DNA Read Write and Edit Market by Technology-
- CRISPR-Cas Systems
- TALEN’s
- Base Editors
- ZFN’s
DNA Read Write and Edit Market by End-user-
- Pharmaceutical and Biotechnology Companies
- Clinical Research Organizations
- Academic and Research Institutes
- Others
DNA Read Write and Edit Market By Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
DNA Read Write and Edit Market Size is valued at USD 8.99 Bn in 2025 and is predicted to reach USD 31.45 Bn by the year 2035
The DNA Read Write and Edit Market is expected to grow at a 13.5% CAGR during the forecast period for 2026 to 2035
Agilent Technologies, Editas Medicine, GenScript Biotech, Beam Therapeutics, Cellectis, CRISPR Therapeutics, Danaher, Illumina, Bluebird Bio, Oxford Nanopore Technologies, QIAGEN, Roche, Sangamo Therapeutics, Pacific Biosciences, PerkinElmer (through acquisition of Horizon Discovery), Thermo Fisher Scientific, Intellia Therapeutics, Bio-Rad Laboratories, Twist Bioscience and Others
DNA Read Write and Edit Market is segmented into Product Type, Delivery Mode, Targeted Organism, Application, Technology, End-user, and Other.
North America region is leading the DNA Read Write and Edit Market.
