eQMS for Pharma and Medtech Market Size, Trend, Revenue Report 2026 to 2035
What is eQMS for Pharma and Medtech Market?
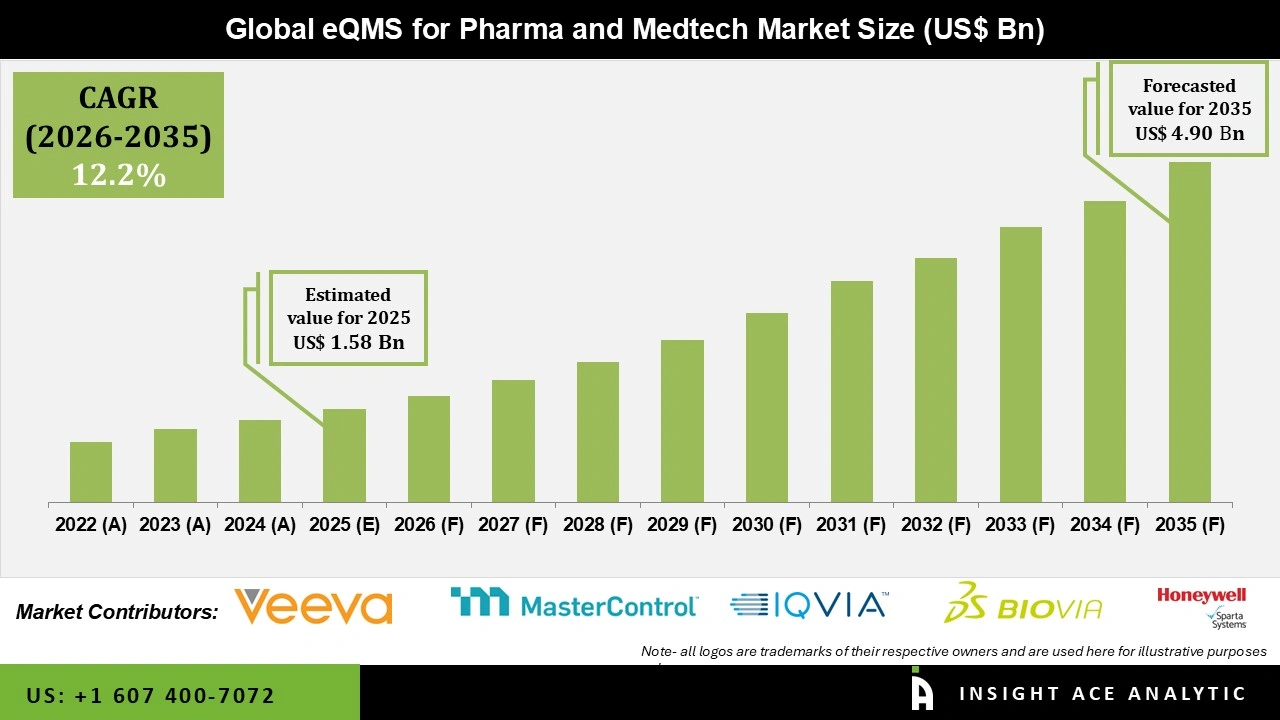
Global eQMS for Pharma and Medtech Market Size is valued at USD 1.58 Bn in 2025 and is predicted to reach USD 4.90 Bn by the year 2035 at a 12.2% CAGR during the forecast period for 2026 to 2035.
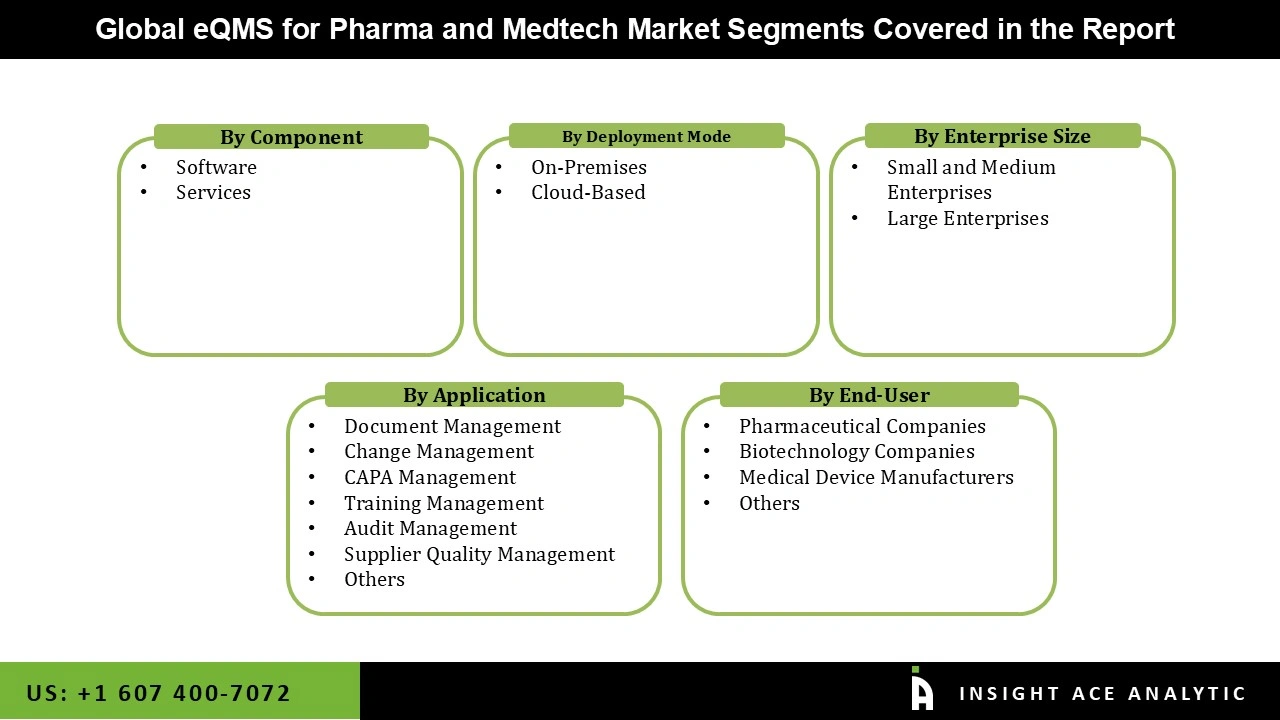
eQMS for Pharma and Medtech Market Size, Share & Trends Analysis Distribution by Component (Software and Services), Enterprise Size (Small and Medium Enterprises, Large Enterprises), Deployment Mode (Cloud-Based and On-Premises), Application (Document Management, Training Management, CAPA Management, Change Management, Supplier Quality Management, Audit Management, and Others), End-user (Pharmaceutical Companies, Medical Device Manufacturers, Biotechnology Companies, and Others), and Segment Forecasts, 2026 to 2035.
An electronic Quality Management System (eQMS) is a digital platform used in the pharmaceutical and medical technology (MedTech) industries to supervise, monitor, and document all quality-related processes in compliance with stringent legal standards. It replaces paper-based approaches with centralized, automated workflows for critical quality tasks such as document control, deviation management, CAPA (Corrective and Preventive Actions), change control, audit management, training management, and complaint handling. The primary factors propelling the eQMS for pharma and medtech market's growth are stricter regulations, increasing complexity in pharmaceutical and medical device production, and the pressing need for digital transformation in quality management systems across the life sciences industry.
The companies are being forced to switch from old paper-based systems to automated, centralized digital platforms due to factors such as growing product complexity, internationalization of supply chains, and more scrutiny over data integrity. Furthermore, the need for strong quality systems that can guarantee traceability, expedite CAPA procedures, and improve audit preparedness is expanding due to the increased focus on patient safety and risk management. The growing adoption of eQMS solutions is partly a result of the growth of the pharmaceutical and MedTech industries, especially in developing nations, as businesses seek to increase operational effectiveness while upholding compliance. Additionally, ongoing technology developments, including cloud-based deployment, analytics powered by artificial intelligence, and integration with corporate systems, are anticipated to boost the eQMS for pharma and medtech market growth.
In order to improve transparency and real-time monitoring, regulatory agencies are progressively promoting digital transformation, which is bolstering the eQMS for pharma and medtech market expansion. Additionally, there is a discernible shift toward scalable and configurable eQMS systems that are suited to particular organizational requirements, suggesting that providers are concentrating on adaptable and user-friendly solutions. The eQMS for pharma and medtech market is anticipated to increase steadily in the upcoming years as more people become aware of the advantages of digital quality management, including decreased human error, quicker decision-making, and enhanced compliance. Moreover, a major factor in the eQMS for pharma and medtech market's expansion is the increasing complexity of supply chains for pharmaceuticals and medical devices.
Competitive Landscape
Which are the Leading Players in eQMS for Pharma and Medtech Market?
- Siemens (Polarion)
- Oracle
- Veeva Systems
- IQVIA
- Qualio
- Greenlight Guru
- Sparta Systems (Honeywell)
- Dassault Systèmes (BIOVIA, Medidata)
- SAP
- Plexus (formerly TrackWise Digital)
- QUMAS
- Ennov
- ValGenesis
- ComplianceQuest
- ETQ
- Dot Compliance
- Intellect
- AssurX
- MasterControl
- Arena Solutions (PTC)
Market Dynamics
Driver
Growing Regulatory Pressure and Compliance Requirements
One of the main factors driving the eQMS for pharma and medtech market is the growing regulatory pressure and compliance requirements. Strict control over documentation, traceability, and quality processes is required by standards like ISO 13485 and regulatory bodies such as the U.S. Food and Drug Administration. Businesses must put in place digital systems that guarantee accuracy, openness, and audit readiness as inspections increase in frequency and data integrity standards rise. eQMS systems lower the risk of human error and non-compliance by enabling centralized data administration, automated workflows, and real-time monitoring. The eQMS for pharma and medtech market is being further accelerated by the increasing need to maintain consistent product quality and avoid regulatory penalties.
Restrain/Challenge
High Cost and Complexity of System Validation
The high cost and complexity of system validation and deployment are major barriers to eQMS for pharma and medtech market growth. The implementation of an eQMS necessitates a significant cost outlay for infrastructure, staff training, software modification, and purchasing. It can be time-consuming and resource-intensive for firms to validate the system in compliance with regulatory requirements set by organizations such as the U.S. Food and Drug Administration. Additionally, there may be technical difficulties when connecting eQMS with current enterprise systems like ERP or LIMS, which could cause ongoing operations to be disrupted. These obstacles have the potential to impede decision-making and reduce the pace at which eQMS solutions are adopted overall.
Software Segment is Expected to Drive the eQMS for Pharma and Medtech Market
The software category held the largest share in the eQMS for Pharma and Medtech market in 2025. As businesses look to automate quality processes, guarantee regulatory compliance, and boost operational efficiencies, the need for comprehensive, adaptable, and user-friendly eQMS platforms is growing. By incorporating cloud-native architectures, artificial intelligence, and advanced analytics into their software products, vendors are constantly innovating. As a result, there is a strong pipeline of feature-rich products that meet the various demands of biotech, medtech, and pharmaceutical companies. Additionally, it is anticipated that the software sector will continue to dominate the market for the duration of the projection period as the industry shifts toward digital transformation.
Document Management Segment is Growing at the Highest Rate in the eQMS for Pharma and Medtech Market
In 2025, the Document Management category dominated the eQMS for Pharma and Medtech market. For operational effectiveness and regulatory compliance, it is crucial to be able to centralize, secure, and manage access to important documents. Automated version control, electronic signatures, and enterprise resource planning (ERP) system integration are the main areas of innovation in this field. Robust document management skills are crucial for preserving data integrity and enabling effective audits as document complexity rises and regulatory requirements change. Furthermore, the use of cloud-based eQMS platforms, remote audits, and international cooperation between R&D and production facilities is increasing the significance of secure document sharing and real-time access.
Why North America Led the eQMS for Pharma and Medtech Market?
The eQMS for Pharma and Medtech market was dominated by North America region in 2025. The existence of significant pharmaceutical and medical device businesses, a developed regulatory framework, and the early embrace of digital health technology all contribute to the region's leadership. Strong investments in R&D and quality control, along with a highly established healthcare ecosystem, are particularly advantageous to the United States.
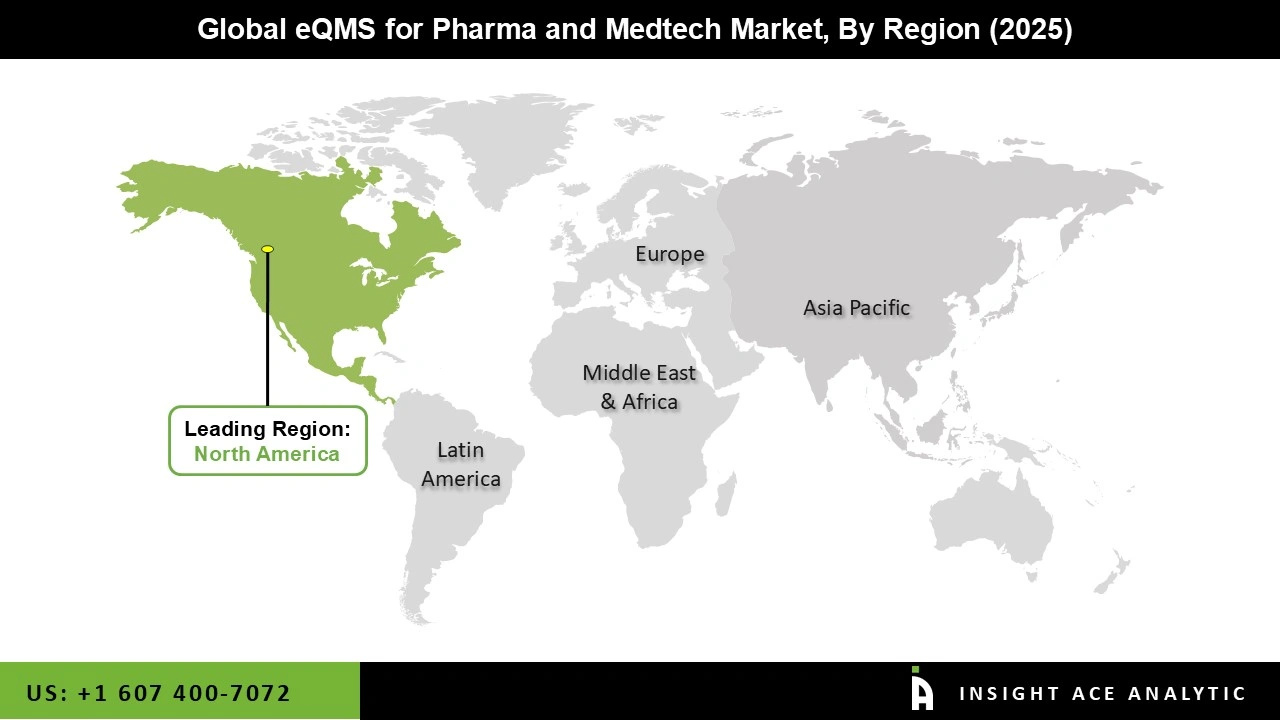
The FDA's strict compliance requirements, along with an innovative and continuous improvement mindset, have propelled the broad use of eQMS platforms. Additionally, the prominent suppliers have solidified their positions in North America by providing customized solutions that cater to the particular requirements of the highly regulated industries in the region.
eQMS for Pharma and Medtech Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 1.58 Bn |
| Revenue forecast in 2035 | USD 4.90 Bn |
| Growth Rate CAGR | CAGR of 12.2% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Component, Enterprise Size, Deployment Mode, Application, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Siemens (Polarion), Oracle, Veeva Systems, IQVIA, Qualio, Greenlight Guru, Sparta Systems (Honeywell), Dassault Systèmes (BIOVIA, Medidata), SAP, Plexus (formerly TrackWise Digital), QUMAS, Ennov, ValGenesis, ComplianceQuest, ETQ, Dot Compliance, Intellect, AssurX, MasterControl, and Arena Solutions (PTC). |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentations of eQMS for Pharma and Medtech Market :
eQMS for Pharma and Medtech Market, by Component-
- Software
- Services
eQMS for Pharma and Medtech Market, by Enterprise Size-
- Small and Medium Enterprises
- Large Enterprises
eQMS for Pharma and Medtech Market, by Deployment Mode-
- Cloud-Based
- On-Premises
eQMS for Pharma and Medtech Market, by Application-
- Document Management
- Training Management
- CAPA Management
- Change Management
- Supplier Quality Management
- Audit Management
- Others
eQMS for Pharma and Medtech Market, by End-user-
- Pharmaceutical Companies
- Medical Device Manufacturers
- Biotechnology Companies
- Others
eQMS for Pharma and Medtech Market, by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East and Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
eQMS for Pharma and Medtech Market Size is valued at USD 1.58 Bn in 2025 and is predicted to reach USD 4.90 Bn by the year 2035
The eQMS for Pharma and Medtech Market is expected to grow at a 12.2% CAGR during the forecast period for 2026 to 2035
Siemens (Polarion), Oracle, Veeva Systems, IQVIA, Qualio, Greenlight Guru, Sparta Systems (Honeywell), Dassault Systèmes (BIOVIA, Medidata), SAP, Plexus (formerly TrackWise Digital), QUMAS, Ennov, ValGenesis, ComplianceQuest, ETQ, Dot Compliance, Intellect, AssurX, MasterControl, and Arena Solutions (PTC). and Others.
eQMS for Pharma and Medtech Market is segmented into Component, Enterprise Size, Deployment Mode, Application, End-user, and Other.
North America region is leading the eQMS for Pharma and Medtech Market.