Pharmacometric Modeling and Simulation Market Size, Trend, Forecast Report 2026 to 2035
What is Pharmacometric Modeling and Simulation Market?
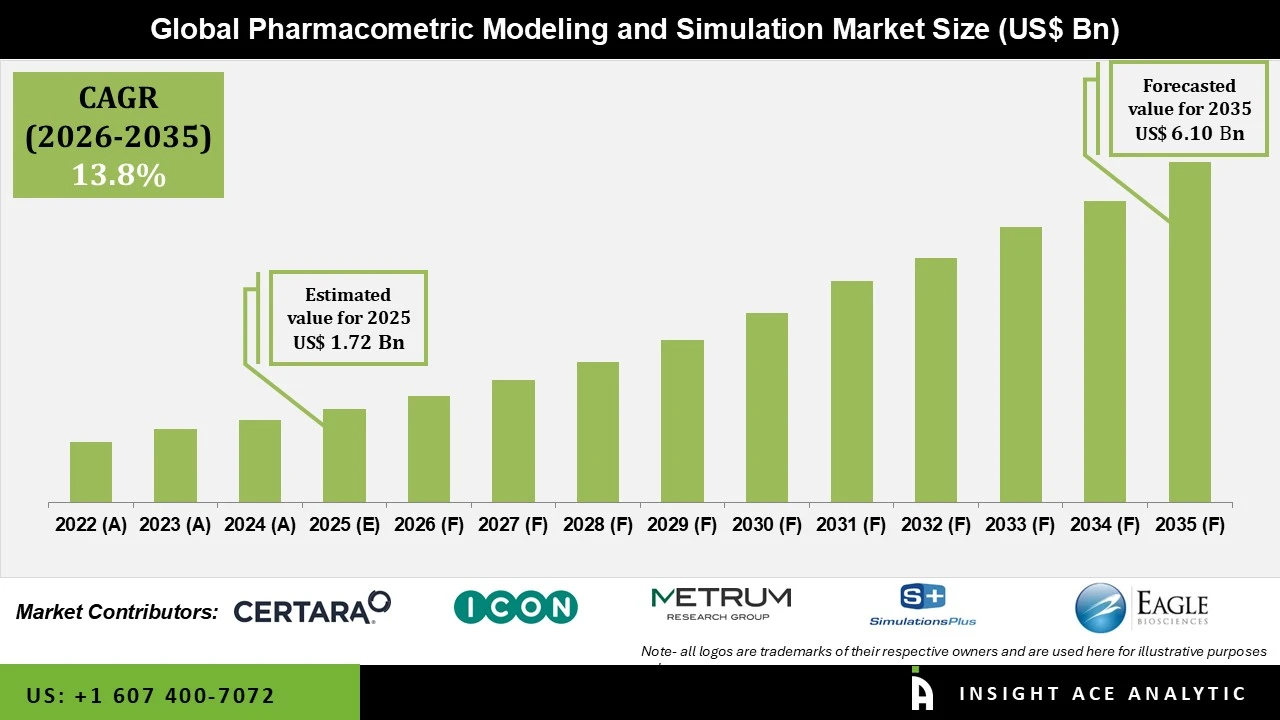
Pharmacometric Modeling and Simulation Market Size is valued at USD 1.72 Bn in 2025 and is predicted to reach USD 6.10 Bn by the year 2035 at a 13.8% CAGR during the forecast period for 2026 to 2035.
Pharmacometric Modeling and Simulation Market Size, Share & Trends Analysis Distribution by Product Type (Software and Services), Application (Clinical Trials, Drug Development, Regulatory Submissions, and Others), End-user (Pharmaceutical Companies, Contract Research Organizations, Biotechnology Companies, Academic and Research Institutes, and Others), and Segment Forecasts, 2026 to 2035
Pharmacometric modeling and simulation (M&S) is a quantitative scientific method used in drug research that integrates biology, statistics, and mathematics to study and forecast how medications will behave in the human body. It entails creating models that explain how a medication is absorbed, transported, metabolized, and removed, in addition to how it causes both positive and negative effects. Pharmacometric models help researchers optimize drug efficacy and safety by simulating various dose regimens, patient groups, and trial situations using clinical and preclinical data. The demand for more affordable clinical trials, the increased complexity of drug development, and the regulatory emphasis on model-informed drug development (MIDD) are the main factors driving the pharmacometric modeling and simulation market's expansion.
The pharmacometric modeling and simulation market is also being accelerated by the growing emphasis on precision dosing and customized medicine, which assist customize treatments for certain patient populations. As regulatory bodies increasingly support model-informed drug development to improve decision-making and lower late-stage failures, the trend is anticipated to continue. Furthermore, in particular groups where standard clinical trials may be difficult or limited, such as children, geriatrics, and patients with comorbidities, pharmacometric modeling is essential. Additionally, developments in machine learning, artificial intelligence, and computational technologies are influencing the field of pharmacometric modeling and simulation by increasing model predictability and accuracy. Its applications are being extended beyond traditional drug development into fields such as illness progression modeling and health economics due to the integration of real-world data and advanced analytics.
In addition, there is an increasing trend toward the creation of regulatory frameworks and standardized platforms that support model-based approaches, encouraging the pharmacometric modeling and simulation market growth. Pharmacometric modeling and simulation are anticipated to play an increasingly important role in influencing the future of drug discovery and development as awareness of their advantages—such as shorter development times, higher success rates, and enhanced therapeutic outcomes—grows. Moreover, the expanding trend of open innovation and collaborative research is opening up new avenues for cross-sector collaborations, allowing businesses to take advantage of complementary resources and expertise to spur the pharmacometric modeling and simulation market expansion. However, a major obstacle to the pharmacometric modeling and simulation market's expansion is the lack of qualified experts with knowledge in modeling, simulation, and quantitative pharmacology.
Competitive Landscape
Which are the Leading Players in Pharmacometric Modeling and Simulation Market?
• Certara Inc.
• Metrum Research Group
• Pharmetheus AB
• Projections Research Inc.
• Eagle Biosciences Inc.
• ICON plc
• Quantitative Solutions (a Certara company)
• Nuventra Pharma Sciences
• Lixoft (a Simulations Plus company)
• IntiQuan
• Pharsight (a Certara company)
• Simulations Plus Inc.
• Cognigen Corporation
• Bayer AG
• Novadiscovery
• QSP Solutions
• Menarini Group
• Evotec SE
• Model Answers Pty Ltd
• PK/PD Associates
Market Dynamics
Driver
Increasing Adoption of Model-informed Drug Development (MIDD)
One of the main factors greatly boosting the growth of the pharmacometric modeling and simulation market is the increasing use of model-informed drug development (MIDD). Pharmaceutical companies are increasingly using pharmacometric methodologies to make data-driven decisions early in the development lifecycle as drug research grows more intricate, expensive, and time-consuming. In order to forecast drug behavior and improve clinical trial design, these models make it possible to integrate a variety of datasets, including preclinical, clinical, and real-world data. Researchers can avoid needless patient exposure to ineffective therapies, uncover the best dosing regimens, and lower the chance of trial failures by simulating various situations. Additionally, regulatory bodies are starting to acknowledge the importance of M&S in assisting with medication approval applications, which is driving the pharmacometric modeling and simulation market's expansion.
Restrain/Challenge
Lack of Qualified Experts and the Intrinsic Difficulty of Model Construction
The scarcity of qualified experts and the intrinsic difficulty of model construction are two of the main obstacles to the pharmacometric modeling and simulation market growth. Building and maintaining competent teams is difficult for many firms because pharmacometric analysis needs knowledge of several disciplines, including pharmacokinetics, pharmacodynamics, biostatistics, and sophisticated computational techniques. Furthermore, robust model creation and validation require high-quality, well-structured datasets, which are not always easily available. Model correctness and dependability are further complicated by inconsistent data standards and gaps in real-world data. Smaller businesses may be discouraged from fully implementing pharmacometric strategies due to this uncertainty, which would limit the pharmacometric modeling and simulation market penetration.
Drug Development Segment is Expected to Drive the Pharmacometric Modeling and Simulation Market
The drug development category held the largest share in the pharmacometric modeling and simulation market in 2025. The most important use of pharmacometric modeling and simulation is in drug development, where it is essential for maximizing dosage plans, forecasting therapeutic results, and reducing side effects. Through the integration of preclinical and clinical study data, these models help researchers make well-informed decisions across the whole drug development process. This shortens the time to market for novel treatments and lowers the risk of late-stage failures. The importance of pharmacometric studies in creating patient-centered clinical trials and determining the best treatment regimens is further highlighted by the increased focus on personalized medicine and targeted treatments. On the other hand, pharmacometric modeling and simulation are also widely used in clinical trails. These techniques are being utilized more and more to anticipate patient responses based on genetic and demographic characteristics, evaluate the effects of different dosage regimens, and simulate different trial designs.
Pharmaceutical Companies Segment is Growing at the Highest Rate in the Pharmacometric Modeling and Simulation Market
In 2025, the pharmaceutical companies category dominated the pharmacometric modeling and simulation market. These companies use pharmacometric methods to improve the likelihood of regulatory success, lower attrition rates, and optimize drug development pipelines. In order to guarantee ideal dosage, safety, and efficacy, the increasing complexity of drug candidates—especially biologics and targeted therapies—requires sophisticated modeling skills. Pharmaceutical businesses are increasingly investing in internal pharmacometric expertise and collaborating with specialist service providers to maintain a competitive edge as competition heats up and regulatory requirements change. However, due to their emphasis on cutting-edge treatments and individualized care, biotechnology companies are also becoming important users of pharmacometric modeling and simulation. Effective and predictive modeling tools are crucial for these businesses to succeed because they frequently have limited resources and short development schedules.
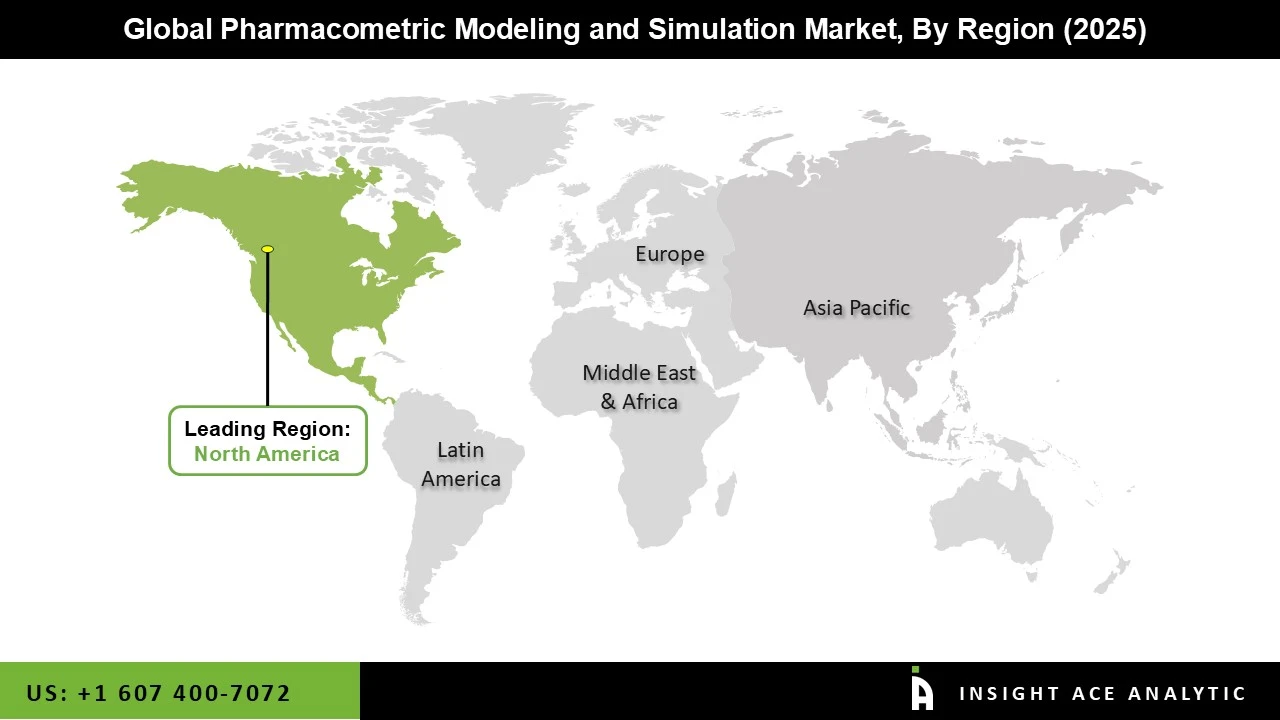
Why North America Led the Pharmacometric Modeling and Simulation Market?
The pharmacometric modeling and simulation market was dominated by North America region in 2025 because of its well-established regulatory framework, robust presence of major industry participants, and highly developed pharmaceutical and biotechnology ecosystem. Significant investments in research and development, which are essential to pharmacometric applications, help the area. The use of simulation tools in regulatory submissions is encouraged by regulatory bodies like the U.S. Food and Drug Administration, which actively supports model-informed medication development. Additionally, pharmacometric models are becoming more accurate and scalable due to the increasing use of cutting-edge technology like artificial intelligence and real-world data analytics. The region's continued growth and leadership in this market are further supported by the existence of qualified people, solid academic-industry partnerships, and a growing emphasis on customized medicine.
Pharmacometric Modeling and Simulation Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 1.72 Bn |
| Revenue forecast in 2035 | USD 6.10 Bn |
| Growth Rate CAGR | CAGR of 13.8% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Application, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Certara Inc., Metrum Research Group, Pharmetheus AB, Projections Research Inc., Eagle Biosciences Inc., ICON plc, Quantitative Solutions (a Certara company), Nuventra Pharma Sciences, Lixoft (a Simulations Plus company), IntiQuan, Pharsight (a Certara company), Simulations Plus Inc., Cognigen Corporation, Bayer AG, Novadiscovery, QSP Solutions, Menarini Group, Evotec SE, Model Answers Pty Ltd, and PK/PD Associates. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Pharmacometric Modeling and Simulation Market by Product Type-
• Software
• Services
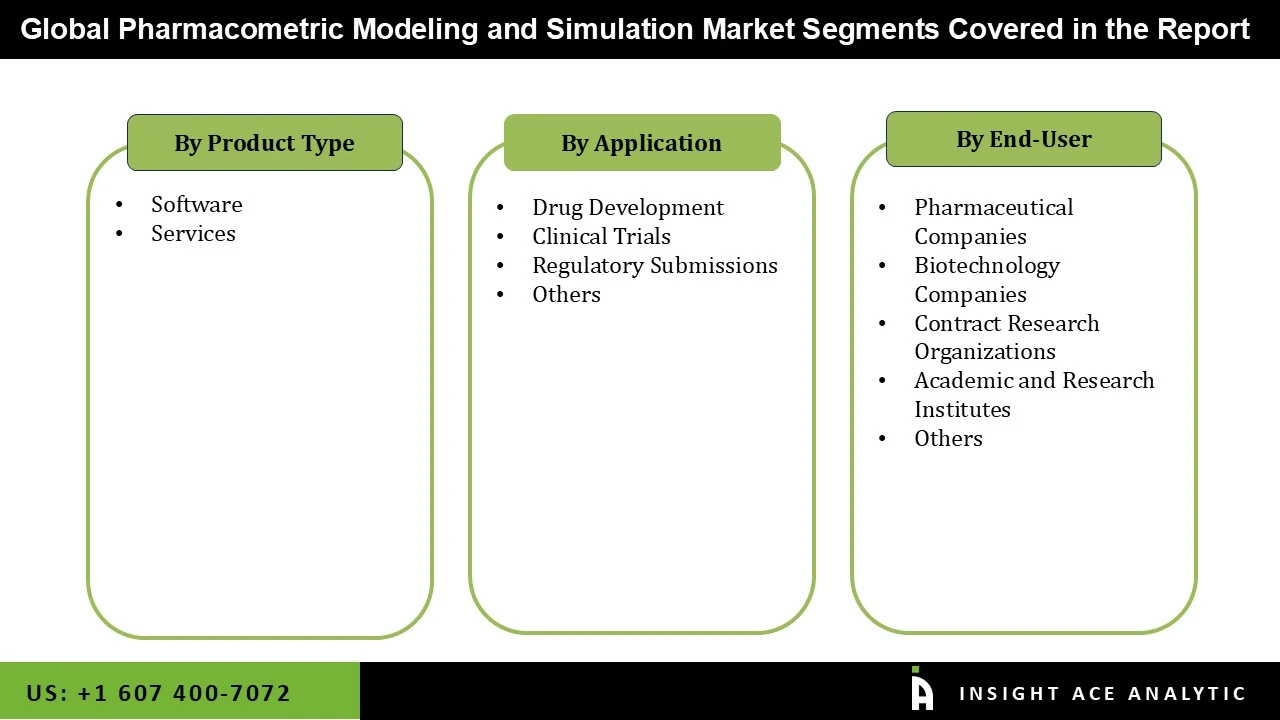
Pharmacometric Modeling and Simulation Market by Application-
• Clinical Trials
• Drug Development
• Regulatory Submissions
• Others
Pharmacometric Modeling and Simulation Market by End-user-
• Pharmaceutical Companies
• Contract Research Organizations
• Biotechnology Companies
• Academic and Research Institutes
• Others
Pharmacometric Modeling and Simulation Market By Region-
North America-
• The US
• Canada
Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Pharmacometric Modeling and Simulation Market Size is valued at USD 1.72 Bn in 2025 and is predicted to reach USD 6.10 Bn by the year 2035
Pharmacometric Modeling and Simulation Market is expected to grow at a 13.8% CAGR during the forecast period for 2026 to 2035.
Certara Inc., Metrum Research Group, Pharmetheus AB, Projections Research Inc., Eagle Biosciences Inc., ICON plc, Quantitative Solutions (a Certara company), Nuventra Pharma Sciences, Lixoft (a Simulations Plus company), IntiQuan, Pharsight (a Certara company), Simulations Plus Inc., Cognigen Corporation, Bayer AG, Novadiscovery, QSP Solutions, Menarini Group, Evotec SE, Model Answers Pty Ltd, and PK/PD Associates.
Pharmacometric Modeling and Simulation Market is segmented into Product Type, Application, End-user, and By Region.
North America region is leading the Pharmacometric Modeling and Simulation Market.