Bioadhesive Ophthalmic Treatments Market Size, Trend, Forecast Report 2026 to 2035
What is Bioadhesive Ophthalmic Treatments Market?
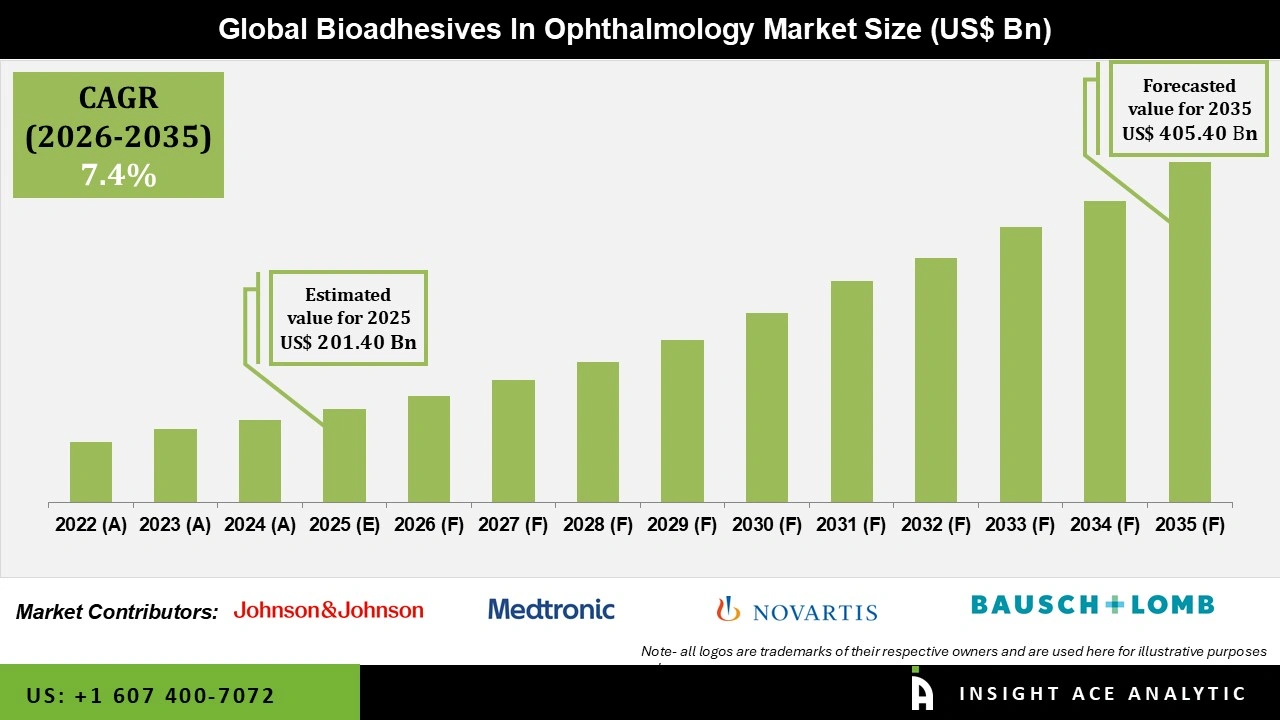
Global Bioadhesive Ophthalmic Treatments Market Size is valued at USD 201.40 Bn in 2025 and is predicted to reach USD 405.40 Bn by the year 2035 at a 7.4% CAGR during the forecast period for 2026 to 2035.
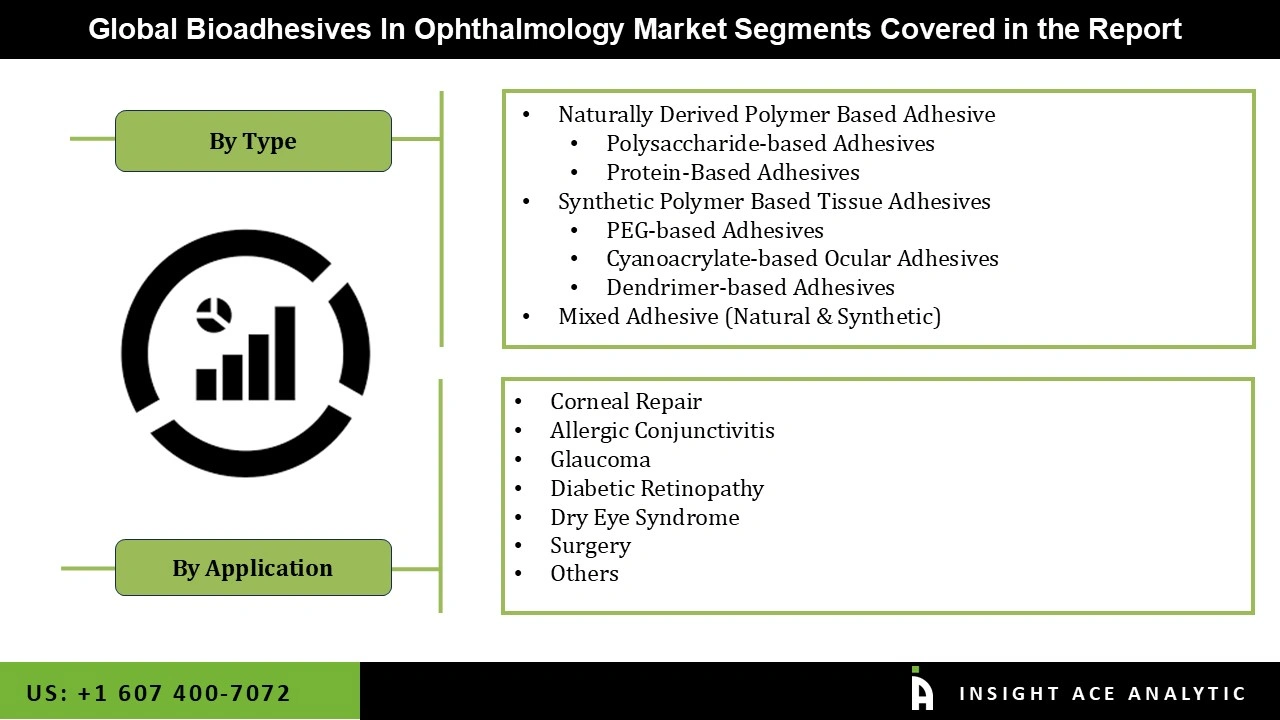
Bioadhesive Ophthalmic Treatments Market Size, Share & Trends Analysis Distribution by Type (Naturally Derived Polymer Based Adhesive (Polysaccharide-based Adhesives, Protein-Based Adhesives), Synthetic Polymer Based Tissue Adhesives (PEG-based Adhesives, Cyanoacrylate-based Ocular Adhesives, Dendrimer-based Adhesives), and Mixed Adhesive (Natural & Synthetic)), Application (Corneal Repair, Glaucoma, Diabetic Retinopathy, Dry Eye Syndrome, Allergic Conjunctivitis, Surgery, and Others), and Segment Forecasts, 2026 to 2035.
Bioadhesive ophthalmic treatments are cutting-edge drug delivery methods intended to increase the efficacy and duration of drug retention when applied to the eye. These formulations contain bio-adhesive polymers that may stick to the ocular surface, enabling medications to stay in contact with eye tissues for longer periods of time than traditional eye drops, which are quickly removed by tears and blinking. This increases patient compliance, decreases the frequency of doses, and improves medication absorption. In the upcoming years, the bio adhesives in ophthalmology treatments market are anticipated to increase significantly due to factors such as the increasing frequency of ocular ailments, the growing need for minimally invasive operations, and the growing use of drug delivery systems.
The bioadhesive ophthalmic treatments market is being driven by the rising incidence of chronic eye conditions such as glaucoma, dry eye syndrome, and post-operative inflammation because these formulations improve therapeutic efficacy and medication retention on the ocular surface. Furthermore, the transition to bio adhesive methods that offer prolonged medication release and less frequent dosage has been expedited by the drawbacks of traditional eye drops. As patients and healthcare professionals prioritize therapies that increase compliance and provide long-lasting relief, this trend is anticipated to continue. Additionally, the use of biocompatible polymers such as hyaluronic acid, chitosan, and carbomers, together with other developments in polymer science, is broadening the scope of these treatments' application to a variety of ocular disorders.
In addition, the bioadhesive ophthalmic treatments market is probably going to be influenced by continuous advancements in formulation technologies and an increasing focus on patient-centered care. The bioadhesive ophthalmic treatments market is expanding as a result of increased spending in ophthalmic research and regulatory backing for sophisticated drug delivery systems. Moreover, as precision medicine advances, the industry is seeing a movement toward combination medicines and customized formulations designed for certain eye disorders. The use of bio-adhesive ophthalmic treatments is anticipated to increase gradually as knowledge of the advantages of sustained-release ocular medications grows, especially in developing nations. This growth will be aided by advancements in healthcare infrastructure and easier access to cutting-edge treatments.
Competitive Landscape
Which are the Leading Players in Bioadhesive Ophthalmic Treatments Market?
- Baxter International Inc.
- Aerie Pharmaceuticals Inc.
- AdvanTech Manufacturing Inc.
- Hill-Rom Holdings Inc.
- CromaDex Corporation
- Heraeus Medical GmbH
- I.S. – Fabbrica Italiana Sintetici S.p.A.
- Johnson & Johnson Surgical Industries Pty Limited
Market Dynamics
Driver
Rising Incidence of Age-related and Chronic Eye Problems
One of the main factors driving the growth of bioadhesive ophthalmic treatments market is the rising incidence of age-related and chronic eye problems. Dry eye syndrome, glaucoma, cataracts (after surgery), and retinal abnormalities are among the conditions that are growing more prevalent as a result of environmental pollution, extended screen time, and an aging world population. Since they are rapidly removed from the ocular surface by blinking and tear turnover, traditional ophthalmic solutions frequently fail to produce long-lasting therapeutic effects. This leads to poor medication absorption and the requirement for repeated dosing. By sticking to the eye's mucin layer, bio-adhesive formulations get around these restrictions by extending the duration of drug residence and allowing for regulated, continuous drug release. Additionally, the Bioadhesive Ophthalmic Treatments market is still growing as healthcare systems around the world prioritize effective and patient-friendly treatment choices.
Restrain/Challenge
High Expense of Developing and Formulating Bio Adhesive Ophthalmic Treatments
The difficulty and high expense of developing and formulating bioadhesive ophthalmic treatments are one of the main obstacles preventing their widespread use. In contrast to traditional eye drops, bio adhesive systems necessitate careful polymer and drug delivery mechanism engineering to guarantee maximum adherence, regulated drug release, and minimal discomfort to the delicate ocular surface. In order to achieve this balance, a great deal of research, sophisticated manufacturing processes, and stringent clinical testing are required, all of which greatly raise production costs and time to market. There are additional technological difficulties in preserving product stability, sterility, and consistency throughout time. Furthermore, despite the clinical advantages of these medicines, manufacturers find it challenging to attain widespread adoption due to low awareness and cost concerns in underdeveloped nations.
Naturally Derived Polymer Based Adhesive Segment is Expected to Drive the Bioadhesive Ophthalmic Treatments Market
The naturally derived polymer based adhesive category held the largest share in the Bioadhesive Ophthalmic Treatments market in 2025 driven by the growing need for sustainable, biocompatible, and safe medication delivery methods. Due to their superior mucoadhesive qualities, non-toxicity, and high compatibility with sensitive ocular tissues, natural polymers such as hyaluronic acid, chitosan, alginate, and cellulose derivatives are becoming increasingly popular. Compared to synthetic alternatives, these materials are more comfortable for patients and less likely to cause discomfort since they closely resemble the natural tear film and ocular environment. They are therefore frequently used in formulations for ailments like dry eye syndrome and post-surgical care, when delicate medication delivery and extended hydration are essential.
Dry Eye Syndrome Segment is Growing at the Highest Rate in the Bioadhesive Ophthalmic Treatments Market
In 2025, the dry eye syndrome category dominated the Bioadhesive Ophthalmic Treatments market fueled by factors like excessive screen time, aging populations, environmental pollutants, and changes in lifestyle that contribute to the high and steadily increasing frequency of dry eye disease worldwide. Bio-adhesive formulations that offer prolonged lubrication and sustained drug release are ideal for treating dry eye, which necessitates long-term and frequently daily care. By lowering the frequency of eye drop administration, these treatments greatly increase patient comfort and compliance. Additionally, growing healthcare access and rising ophthalmic care spending in developing nations are bolstering the segment's expansion. It is anticipated that the Bioadhesive Ophthalmic Treatments market will continue to grow as dry eye syndrome becomes more common as a result of contemporary lifestyle choices.
Why North America Led the Bioadhesive Ophthalmic Treatments Market?
The bioadhesive ophthalmic treatments market was dominated by North America region in 2025 driven by the rising incidence of eye problems such glaucoma, dry eye syndrome, and age-related eye disorders. The area gains from early adoption of cutting-edge medication delivery technology, a strong healthcare infrastructure, and sophisticated diagnostic capabilities.
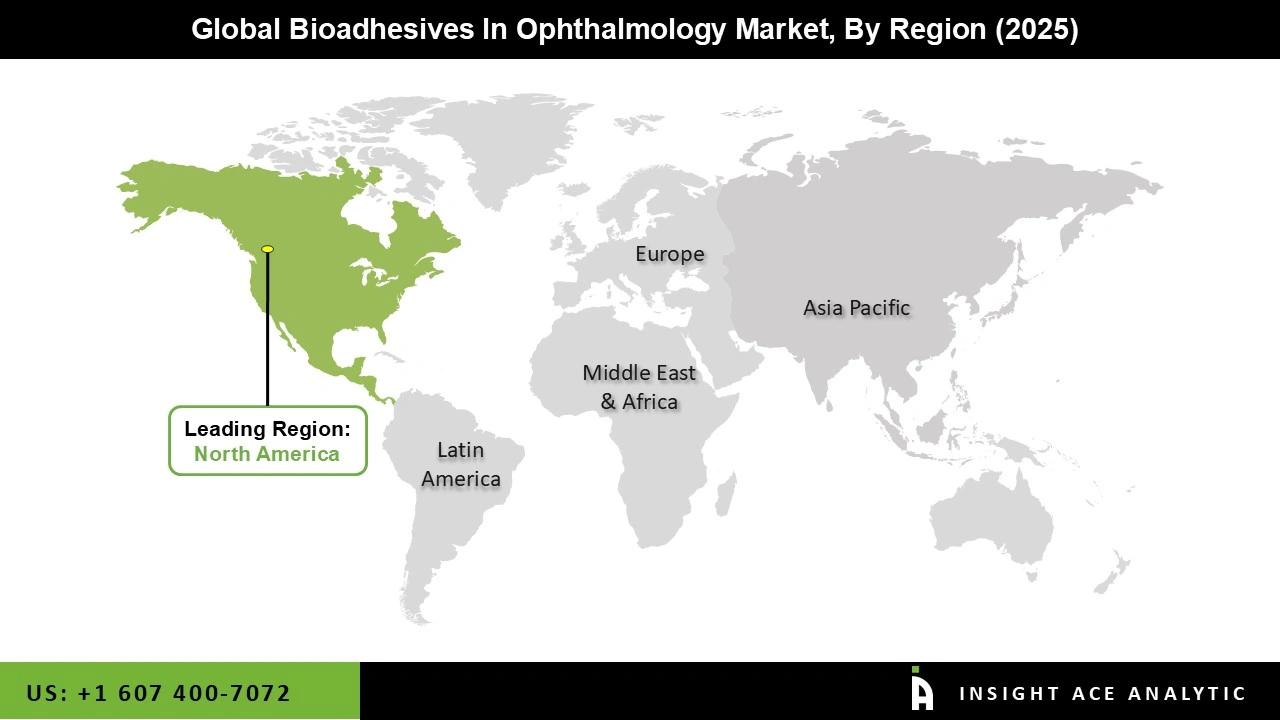
The demand for sustained-release and bio-adhesive formulations is expanding as a result of an increasing number of patients needing long-term ophthalmic treatment due to factors such as increased screen time, an older population, and lifestyle choices. Furthermore, the innovation in ocular drug delivery systems is also being accelerated by the existence of large pharmaceutical and biotechnology businesses, as well as substantial investments in research and development.
Bioadhesive Ophthalmic Treatments Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 201.40 Bn |
| Revenue forecast in 2035 | USD 405.40 Bn |
| Growth Rate CAGR | CAGR of 7.4% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Type, Application, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Baxter International Inc., Aerie Pharmaceuticals Inc., AdvanTech Manufacturing Inc., Hill-Rom Holdings Inc., CromaDex Corporation, Heraeus Medical GmbH, I.S. – Fabbrica Italiana Sintetici S.p.A., and Johnson & Johnson Surgical Industries Pty Limited. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentations of Bioadhesive Ophthalmic Treatments Market:
Bioadhesive Ophthalmic Treatments Market, by Type-
- Naturally Derived Polymer Based Adhesive
- Polysaccharide-based Adhesives
- Protein-Based Adhesives
- Synthetic Polymer Based Tissue Adhesives
- PEG-based Adhesives
- Cyanoacrylate-based Ocular Adhesives
- Dendrimer-based Adhesives
- Mixed Adhesive (Natural & Synthetic)
Bioadhesive Ophthalmic Treatments Market, by Application-
- Corneal Repair
- Glaucoma
- Diabetic Retinopathy
- Dry Eye Syndrome
- Allergic Conjunctivitis
- Surgery
- Others
Bioadhesive Ophthalmic Treatments Market, by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East and Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Bioadhesive Ophthalmic Treatments Market Size is valued at USD 201.40 Bn in 2025 and is predicted to reach USD 405.40 Bn by the year 2035
Bioadhesive Ophthalmic Treatments Market is expected to grow at a 7.4% CAGR during the forecast period for 2026 to 2035
Baxter International Inc., Aerie Pharmaceuticals Inc., AdvanTech Manufacturing Inc., Hill-Rom Holdings Inc., CromaDex Corporation, Heraeus Medical GmbH, I.S. – Fabbrica Italiana Sintetici S.p.A., and Johnson & Johnson Surgical Industries Pty Limited. and Others.
Bioadhesive Ophthalmic Treatments Market is segmented into Type, Application, and Other.
North America region is leading the Bioadhesive Ophthalmic Treatments Market.