Exosome Raw Materials and Manufacturing Services Market Size, Share and Trends Report 2026 to 2035
What is Exosome Raw Materials and Manufacturing Services Market Size?
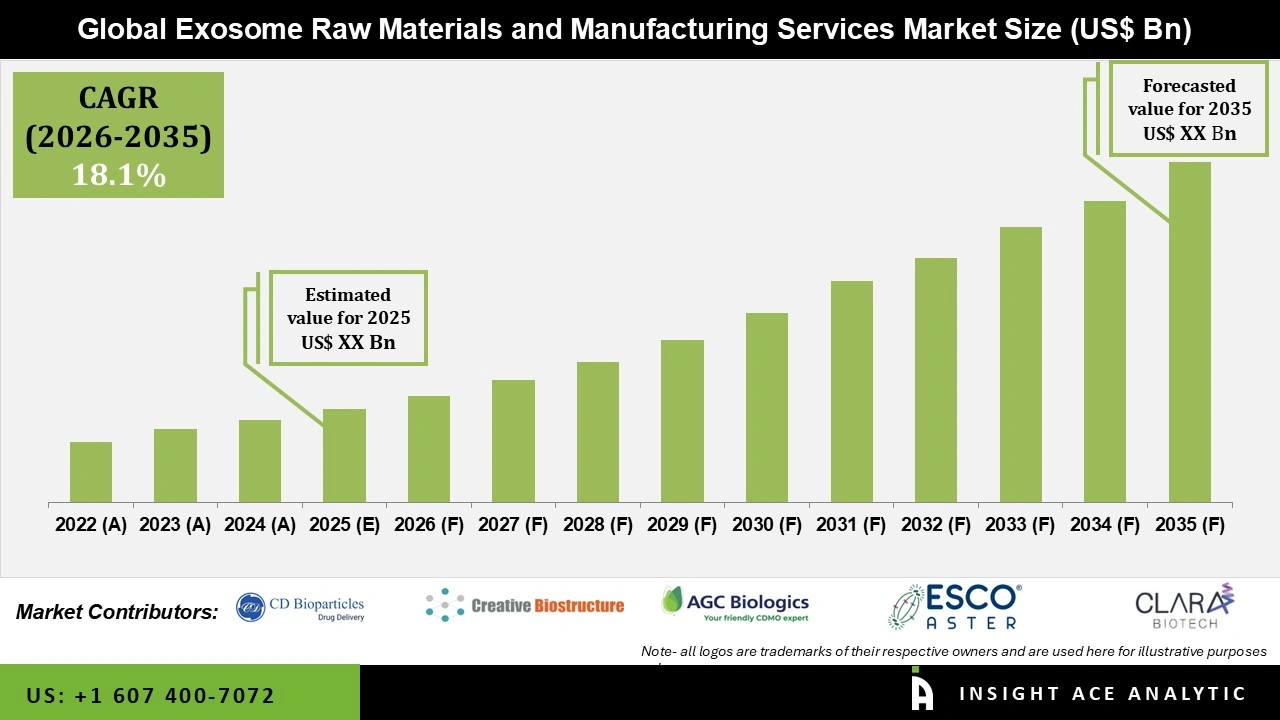
Global Exosome Raw Materials and Manufacturing Services Market Size is predicted to witness a 18.1% CAGR during the forecast period for 2026 to 2035.
Exosome Raw Materials and Manufacturing Services Market, Share & Trends Analysis Report, By Raw Material Type (Cell Sources, Biological Fluids, Plant/Microbial Sources, Synthetic/Engineered Exosomes), By Services Offered (Isolation & Purification, Characterization & Analytics, Engineering & Targeted Delivery, Diagnostic Biomarker Services, GMP Manufacturing (CDMO)), By Application, By End User, By Region, and Segment Forecasts, 2026 to 2035
Exosome Raw Materials and Manufacturing Services Market Key Takeaways:
|
Exosomes are tiny, membrane-enclosed particles released by cells that measure between 30 and 150 nanometers. These vesicles function as cellular messengers, transporting proteins, lipids, and genetic material such as mRNA and miRNA between cells. Due to their natural communication role and versatile cargo capacity, exosomes show considerable promise across multiple therapeutic and diagnostic applications—from cancer immunotherapy and tissue regeneration to disease detection and personalized medicine. To meet the growing demand for high-quality exosomes in research and clinical settings, specialized manufacturers have developed comprehensive production and processing solutions. These services encompass standardized, scalable manufacturing protocols as well as advanced techniques for exosome isolation, purification, quality assessment, and validation. Many of these manufacturers also offer the ability to engineer exosomes by loading them with therapeutic agents such as mRNA, siRNA, proteins, or small-molecule drugs enabling precise, targeted delivery directly to diseased tissues. Contract Development and Manufacturing Organizations (CDMOs) are the primary providers of these specialized exosome production and manufacturing services.
Researchers are increasingly focused on optimizing mesenchymal stem cell (MSC) sources derived from bone marrow, adipose tissue, and umbilical cord to enhance exosome production yields and therapeutic effectiveness. A 2025 comparative study by RoosterBio demonstrated that different MSC sources produce exosomes with notably different characteristics, including variations in particle count, size distribution, and CD73 biomarker expression. These findings provide valuable insights for scaling regenerative medicine applications and have sparked growing market interest in supporting technologies, particularly preconditioning agents and specialized cell culture methods designed to maximize exosome output. To meet this demand, manufacturers are investing in advanced infrastructure, including automated systems and large-scale bioreactors that increase production capacity while maintaining quality. Companies like RoosterBio are simultaneously developing Good Manufacturing Practice (GMP)-compliant MSC platforms specifically designed to support clinical trials and produce therapeutic-grade exosomes. As clinical demand for high-quality exosomes continues to rise, these investments in scalable, standardized cell sources and manufacturing infrastructure have become critical for commercializing exosome-based therapeutics and driving broader market growth.
Competitive Landscape
Which are the Leading Players in Exosome Raw Materials and Manufacturing Services Market?
- CD Bioparticles
- Creative Biostructure
- Direct Biologics
- Elevai
- EriVan Bio
- ExoCoBio
- FUJIFILM Wako Chemicals
- Invitrx Inc.
- Kimera Labs
- Mibelle AG Biochemistry
- Microgentas
- Oasis Diagnostics Corporation
- RoosterBio
- Unicyte AG
- Vitti Labs
- AGC Biologics
- Clara Biotech
- Creative Biolabs
- Esco Aster
- EVerZom
- EXO Biologics (ExoXpert)
- Histocell
- Lonza
- MYCENAX Biotech
- RoosterBio
- SBH Sciences
- WuXi Advanced Therapies
Market Dynamics
Driver
Increase in Therapeutic Applications and Clinical Demand
Expanding clinical applications across immunotherapy, regenerative medicine, and targeted drug delivery are driving substantial demand for exosomes. As researchers validate exosome efficacy in cancer vaccines, tissue repair, and personalized medicine, pharmaceutical companies and clinical developers increasingly require high-quality, therapeutics-grade exosomes for trials and commercialization. This growing pipeline of exosome-based therapeutics creates urgency for reliable manufacturing capacity and standardized production processes. Additionally, exosomes' natural biocompatibility and minimal immunogenicity make them attractive alternatives to synthetic delivery systems, accelerating their adoption in clinical settings. The rise in clinical development programs and regulatory pathways for exosome therapeutics intensifies demand for CDMO services and raw materials, directly propelling market growth and incentivizing investment in manufacturing infrastructure and quality assurance capabilities.
Restrain
Regulatory and Standardization Challenges
The lack of harmonized regulatory frameworks and standardized characterization methods remains a significant restraint on exosome market growth. Regulatory agencies worldwide have not yet established clear guidelines for exosome manufacturing, quality control, and clinical approval, creating uncertainty for manufacturers and developers. Inconsistent standards for particle size distribution, potency assays, purity metrics, and safety testing across regions complicate production scaling and commercialization. Additionally, the complexity of exosome biology including batch-to-batch variability and difficulty in ensuring consistent therapeutic efficacy raises concerns about reproducibility and regulatory acceptance. These uncertainties increase development timelines and costs, delaying clinical applications and deterring investment. Until comprehensive international standards and regulatory pathways are established, companies face significant barriers to bringing exosome therapeutics to market efficiently.
The Isolation and Purification Segment is Expected to Have the Highest Growth Rate During the Forecast Period
The isolation and purification segment is poised for the highest growth rate during the forecast period due to increasing demand for high-quality, therapeutics-grade exosomes. As clinical applications expand, manufacturers require advanced techniques to achieve superior purity and consistency, directly impacting therapeutic efficacy and regulatory compliance. Growing investments in automated purification technologies including ultracentrifugation, size exclusion chromatography, and immunoaffinity methods—enable scalable, reproducible isolation processes. Additionally, the need to remove contaminants and non-therapeutic particles drives adoption of sophisticated purification platforms. CDMOs are expanding their isolation and purification service offerings to meet rising clinical demands, while technological innovations continue improving efficiency and yield. This segment's critical role in ensuring exosome quality and safety positions it as the fastest-growing market component.
The Therapeutics Segment Dominates the Market
The therapeutics segment is experiencing robust growth driven by an expanding pipeline of exosome-based therapies entering clinical development. MSC-derived exosomes are being investigated for serious conditions including acute respiratory distress syndrome (ARDS), stroke recovery, and cancer immunotherapy, all of which require substantial manufacturing capacity. Meeting clinical demands necessitates both large-scale production capabilities and sophisticated purification technologies to ensure consistent quality and safety across batches. Therapeutic applications demand exceptionally pure exosomes to minimize adverse effects and maximize treatment efficacy, driving adoption of advanced isolation techniques such as tangential flow filtration and size-exclusion chromatography. The combination of stringent quality requirements, expensive GMP-compliant manufacturing processes, and premium pricing for clinical-grade exosomes generates significantly higher revenue compared to diagnostics or research applications. This economic advantage, coupled with the critical importance of exosome quality in patient safety, underscores the therapeutics segment's dominant market position and growth potential.
Why North America Led the Exosome Raw Materials and Manufacturing Services Market?
North America maintains a competitive advantage in the exosome market through robust government and private investment in research and development. Federal agencies like the National Institutes of Health (NIH) fund large-scale, multi-institutional research projects focused on extracellular vesicles, while venture capital and major pharmaceutical companies actively invest in exosome-based therapeutics and diagnostic platforms. This supportive ecosystem is reinforced by a favorable regulatory environment that encourages development of GMP-compliant manufacturing processes.
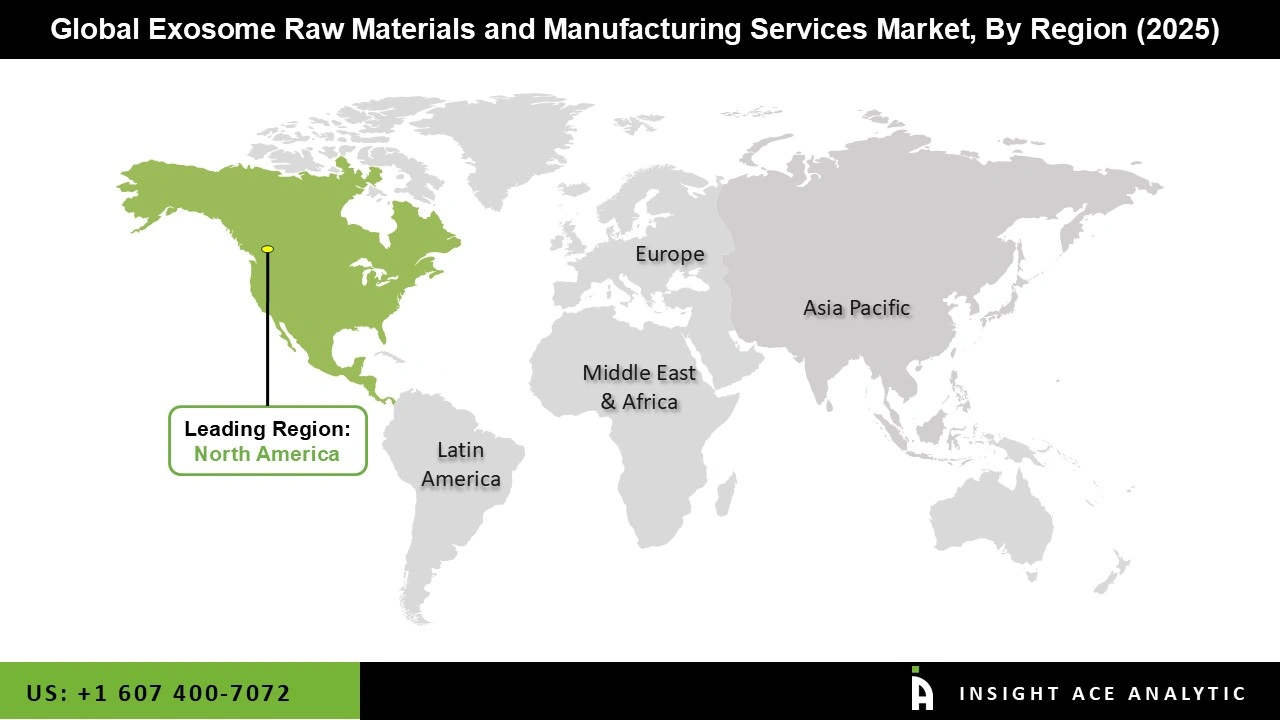
North American companies leverage these advantages to lead in clinical-grade exosome production a critical capability that drives the therapeutics segment's market dominance. Simultaneously, technological innovations including automated production systems and GMP-compliant bioreactors are reducing manufacturing costs and enabling large-scale output without compromising quality. These advancements lower entry barriers for developers, accelerate commercialization timelines, and support both clinical and commercial applications. Collectively, these factors position North America as the global leader in exosome therapeutics development and manufacturing.
Recent Developments:
• In July 2024, Creative Biostructure was thrilled to present the PNExoTM Exosome Series. This cutting-edge range of exosome products made from plants is especially made to aid in the creation of breakthrough skincare formulations and research. Exosomes from plants have a number of special advantages that make them a perfect addition to skincare products. They improve the diffusion and absorption of active substances by blending in perfectly with the lipids in the skin. To improve their stability and skin permeability, plant exosomes are also great carriers of unstable and less permeable compounds, such as retinol, astaxanthin, and β-nicotinamide mononucleotide (NMN).
• In April 2024, EXO Biologics declared that up to EUR 16 million in Series A investment had been successfully secured. The money will help EXO Biologics with its current and upcoming clinical trials. Additionally, it will keep growing its manufacturing business by modernizing its proprietary production platform, ExoPulse(TM), and forming new alliances to provide the platform and GMP clinical-grade exosomes with pan-market access for quicker access to clinical trials.
Exosome Raw Materials and Manufacturing Services Market Report Scope
Segmentation of the Exosome Raw Materials and Manufacturing Services Market :
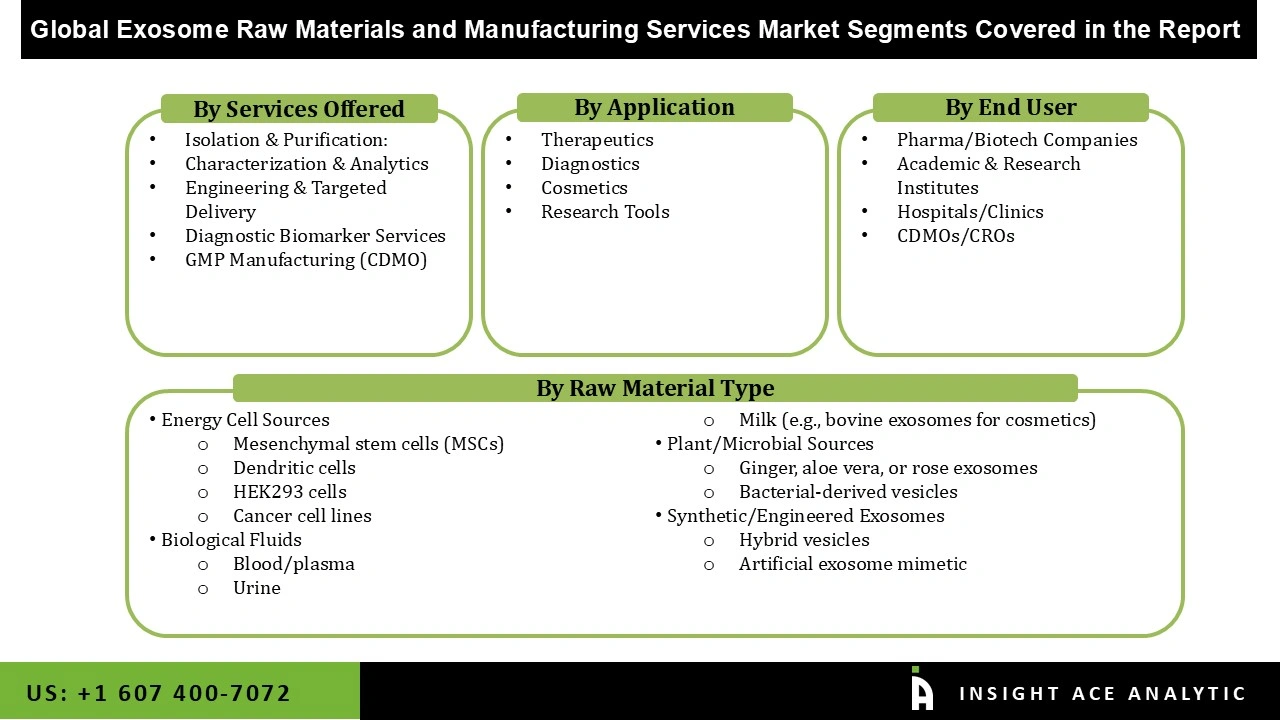
Exosome Raw Materials and Manufacturing Services Market- By Raw Material Type
- Cell Sources
- Mesenchymal stem cells (MSCs)
- Dendritic cells
- HEK293 cells
- Cancer cell lines
- Biological Fluids
- Blood/plasma
- Urine
- Milk (e.g., bovine exosomes for cosmetics)
- Plant/Microbial Sources
- Ginger, aloe vera, or rose exosomes
- Bacterial-derived vesicles
- Synthetic/Engineered Exosomes
- Hybrid vesicles
- Artificial exosome mimetic
Exosome Raw Materials and Manufacturing Services Market – By Services Offered
- Isolation & Purification:
- Characterization & Analytics
- Engineering & Targeted Delivery
- Diagnostic Biomarker Services
- GMP Manufacturing (CDMO)
Exosome Raw Materials and Manufacturing Services Market – By Application
- Therapeutics
- Diagnostics
- Cosmetics
- Research Tools
Exosome Raw Materials and Manufacturing Services Market- By End User
- Pharma/Biotech Companies
- Academic & Research Institutes
- Hospitals/Clinics
- CDMOs/CROs
Exosome Raw Materials and Manufacturing Services Market-by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Exosome Raw Materials and Manufacturing Services Market Size is predicted to witness a 18.1% CAGR during the forecast period for 2026 to 2035.
CD Bioparticles, Creative Biostructure, Direct Biologics, Elevai, EriVan Bio, ExoCoBio, FUJIFILM Wako Chemicals, Invitrx Inc, Kimera Labs, Mibelle AG Biochemistry, Microgentas, Oasis Diagnostics Corporation, RoosterBio, Unicyte AG, Vitti Labs, AGC Biologics, Clara Biotech, Creative Biolabs, Esco Aster, EVerZom, EXO Biologics (ExoXpert), Histocell, Lonza, MYCENAX Biotech, RoosterBio, SBH Sciences, WuXi Advanced Therapies and Others.
Exosome Raw Materials and Manufacturing Services Market is segmented into Raw Material Type, Services Offered, Application and End User.
North America region is leading the Exosome Raw Materials and Manufacturing Services Market.