Next Generation Obesity Drug Pipeline Market Size, Share, Revenue Report 2026 to 2035
What is Next generation Obesity Drug Pipeline Market?
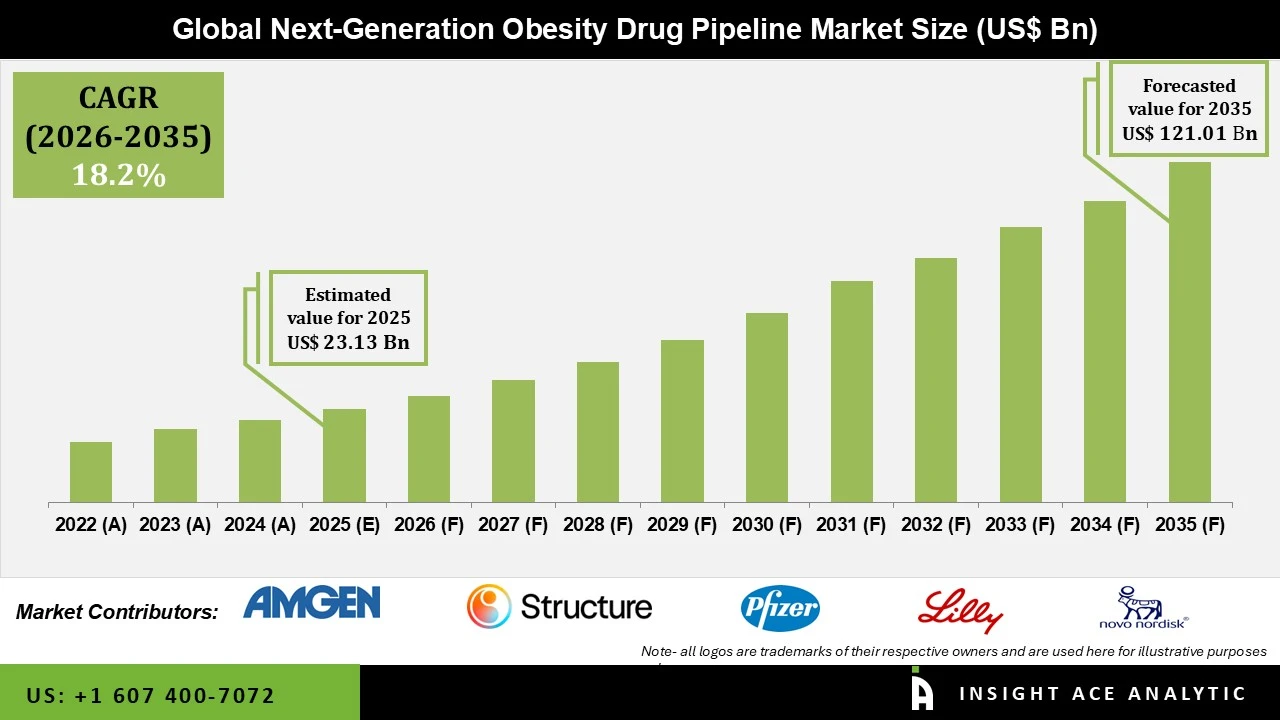
Next generation Obesity Drug Pipeline Market Size is valued at USD 23.13 Bn in 2025 and is predicted to reach USD 121.01 Bn by the year 2035 at a 18.2% CAGR during the forecast period for 2026 to 2035.
Next generation Obesity Drug Pipeline Market Size, Share & Trends Analysis Distribution by Route of Administration (Subcutaneous Injectable, Oral Tablet/Capsule, and Long-Acting Implant), Mechanism of Action (GLP-1/GIP Dual Agonist, Amylin Analog, GLP-1/GIP/Glucagon Triple Agonist, Small Molecule Oral, and Combination Therapy), Patient Segment (Obesity with T2D Comorbidity, Class III Obesity (BMI >40), Pediatric Obesity, and Post-Bariatric Maintenance), and Segment Forecasts, 2026 to 2035
The next generation obesity drug pipeline is a new class of cutting-edge treatments that go beyond first-generation GLP-1 receptor agonists to enhance weight control. By focusing on several pathways related to energy expenditure, glucose metabolism, and appetite management, these new medications aim to improve efficacy, tolerability, and metabolic advantages. In addition to new mechanisms such as amylin analogs, oral peptide treatments, and muscle-preserving weight reduction medications, the pipeline comprises dual and triple agonists like GLP-1/GIP, GLP-1/glucagon, and multi-receptor combinations. Growing desire for more individualized and efficient treatments, rising investment in metabolic medication innovation, and rising obesity incidence worldwide are the main factors propelling the next-generation obesity drug pipeline market's expansion.
The next generation obesity drug pipeline market is being greatly accelerated by the growing prevalence of obesity and associated metabolic problems worldwide, as healthcare systems place a greater emphasis on long-term weight control strategies. By offering greater efficacy, better safety profiles, and long-lasting weight loss results, these cutting-edge medicines are being created to overcome the drawbacks of previous treatments. Furthermore, the need for novel pharmaceutical treatments is being accelerated by patients' and doctors' rising recognition of obesity as a chronic illness rather than a lifestyle issue. The demand for multifunctional medicines that can treat several metabolic pathways at once is being further reinforced by the rising prevalence of comorbidities such as type 2 diabetes, cardiovascular conditions, and non-alcoholic fatty liver disease.
In addition, it is anticipated that ongoing developments in drug delivery technology, precision medicine methodologies, and a greater emphasis on patient-centric treatments will boost the next-generation obesity drug pipeline market. Faster development and commercialization are also being facilitated by regulatory support for innovative treatments and accelerated approval processes for ground-breaking medications. Additionally, there is a growing trend toward customized treatments based on each patient's unique metabolic profile, which is promoting advancements in biomarker-based medication development. In order to speed up clinical studies and broaden therapeutic portfolios, the next-generation obesity drug pipeline market is also seeing an increase in partnerships between biotechnology companies and major pharmaceutical corporations.
Competitive Landscape
Which are the Leading Players in Next-generation Obesity Drug Pipeline Market?
• Pfizer
• Eli Lilly and Company
• Amgen
• Novo Nordisk
• Carmot Therapeutics
• Structure Therapeutics
• Metsera
• Zealand Pharma
Market Dynamics
Driver
Growing Rates of Metabolic Diseases and Obesity
One of the main factors behind the next-generation obesity drug pipeline market is the rapidly rising global burden of obesity. Obesity rates have sharply increased in both established and emerging economies due to sedentary lifestyles, poor nutritional practices, and urbanization. Additionally, this expanding patient population is very vulnerable to related comorbidities such as non-alcoholic fatty liver disease, type 2 diabetes, and cardiovascular diseases, which put a significant burden on healthcare systems. Therefore, longer-term, more effective therapy options that go beyond lifestyle modifications are desperately needed. Furthermore, pharmaceutical companies are increasingly concentrating on next-generation solutions that enhance overall metabolic health in addition to promoting significant weight loss, which is fueling the next-generation obesity drug pipeline market expansion.
Restrain/Challenge
High Price and Restricted Availability of Cutting-edge Treatments
The enormous expense of developing and commercializing these cutting-edge treatments is one of the main obstacles to the next-generation obesity drug pipeline market. The intricacy of creating multi-target medications, carrying out extensive clinical trials, and guaranteeing long-term safety considerably raises total R&D costs. A significant portion of the population, especially in low- and middle-income nations, cannot afford these expenses because they are frequently included in the ultimate cost of the medications. Further limiting patients' access to these treatments are issues with reimbursement and insurance coverage. In addition, this financial obstacle makes it difficult for the next generation of obesity medications to gain extensive market penetration, which could hinder the next-generation obesity drug pipeline market overall expansion.
Oral Tablet/Capsule Segment is Expected to Drive the Next-generation Obesity Drug Pipeline Market
The Oral Tablet/Capsule category held the largest share in the Next-generation Obesity Drug Pipeline market in 2025 driven by the increasing need for treatment solutions that are more patient-friendly and convenient. Oral formulations provide easier administration and better compliance, especially for long-term weight management, in contrast to injectable medicines, which may be linked to discomfort, needle aversion, and lower long-term adherence. The development of efficient oral versions of treatments that were previously only available through injections is made possible by advancements in drug delivery technologies, such as peptide stabilization, absorption enhancers, and protective coatings. This change is making obesity therapies more accessible to a wider range of patients, including those who are reluctant to start injectable regimens.
GLP-1/GIP Dual Agonist Segment is Growing at the Highest Rate in the Next-generation Obesity Drug Pipeline Market
In 2025, the GLP-1/GIP Dual Agonist category dominated the Next-generation Obesity Drug Pipeline market due to the fact that it is more effective than conventional single-hormone treatments and has wider metabolic advantages. More successfully than previous treatments, these dual agonists increase insulin secretion, decrease hunger, and improve energy balance by concurrently targeting both GLP-1 and GIP receptors. These treatments are very appealing to individuals with type 2 diabetes and obesity since clinical results show much better weight loss and improved glycemic control. This mechanism has been proven by the significant clinical and financial success of medications such as tirzepatide, which has prompted pharmaceutical companies to add more dual-acting compounds to their pipelines.
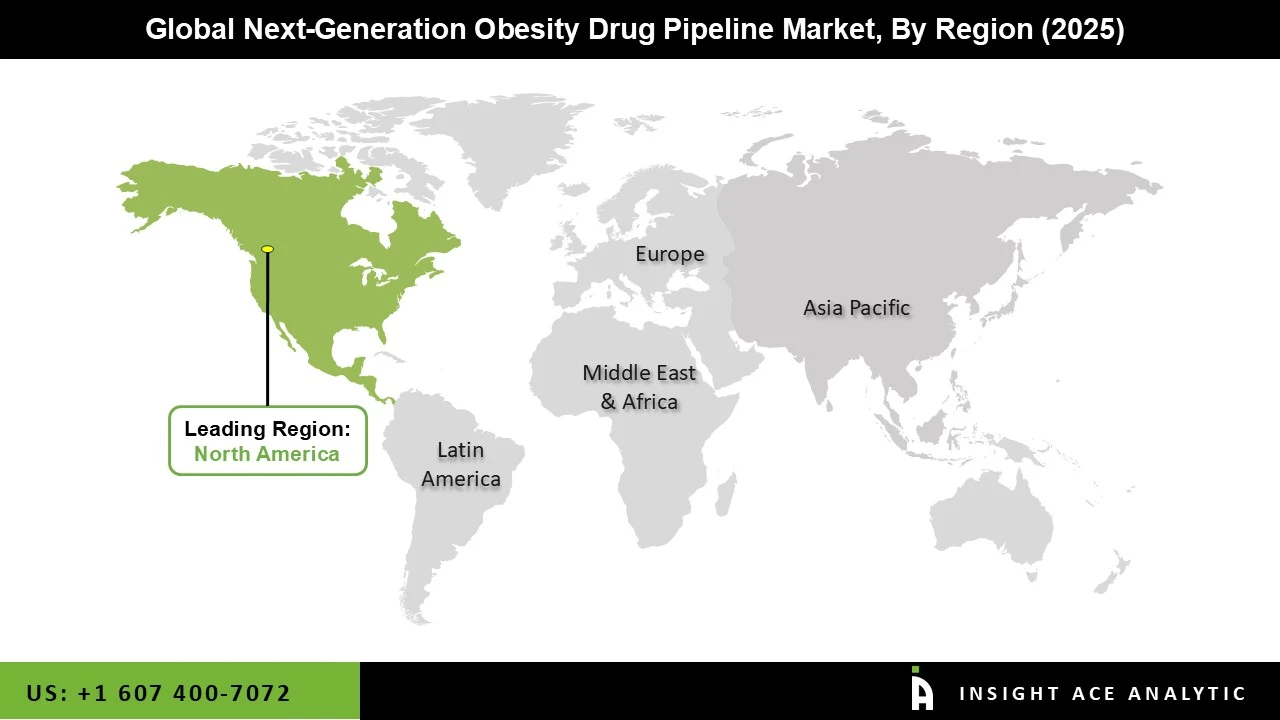
Why North America Led the Next-generation Obesity Drug Pipeline Market?
The Next-generation Obesity Drug Pipeline market was dominated by North America region in 2025 fueled by a robust healthcare system and a high rate of obesity. Due to its sizable patient population with obesity and associated disorders such as type 2 diabetes and cardiovascular diseases, the United States in particular holds a significant portion of the market. The need for improved therapeutics is being driven by rising awareness of obesity as a chronic illness and an increasing readiness to use pharmaceutical treatments. Furthermore, the region's innovation and clinical developments are being accelerated by the presence of top pharmaceutical and biotechnology businesses as well as significant investments in R&D. Additionally, organizations such as the U.S. Food and Drug Administration are facilitating the quick commercialization of next-generation obesity drugs through favorable regulatory frameworks and expedited approval procedures.
Key Development:
February 2026: The Phase 3 TRIUMPH-1 trial results for orforglipron showed a mean body weight reduction of 14.7% at 36 weeks in adults with obesity without diabetes, according to Eli Lilly and Company. This was the highest efficacy ever reported for a once-daily oral non-peptide GLP-1 receptor agonist, and it paved the way for an NDA submission to the FDA in mid-2026.
Next-generation Obesity Drug Pipeline Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 23.13 Bn |
| Revenue forecast in 2035 | USD 121.01 Bn |
| Growth Rate CAGR | CAGR of 18.2% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Route of Administration, Mechanism of Action, Patient Segment, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Pfizer, Eli Lilly and Company, Amgen, Novo Nordisk, Carmot Therapeutics, Structure Therapeutics, Metsera, and Zealand Pharma. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Next-generation Obesity Drug Pipeline Market by Route of Administration-
• Subcutaneous Injectable
• Oral Tablet/Capsule
• Long-Acting Implant
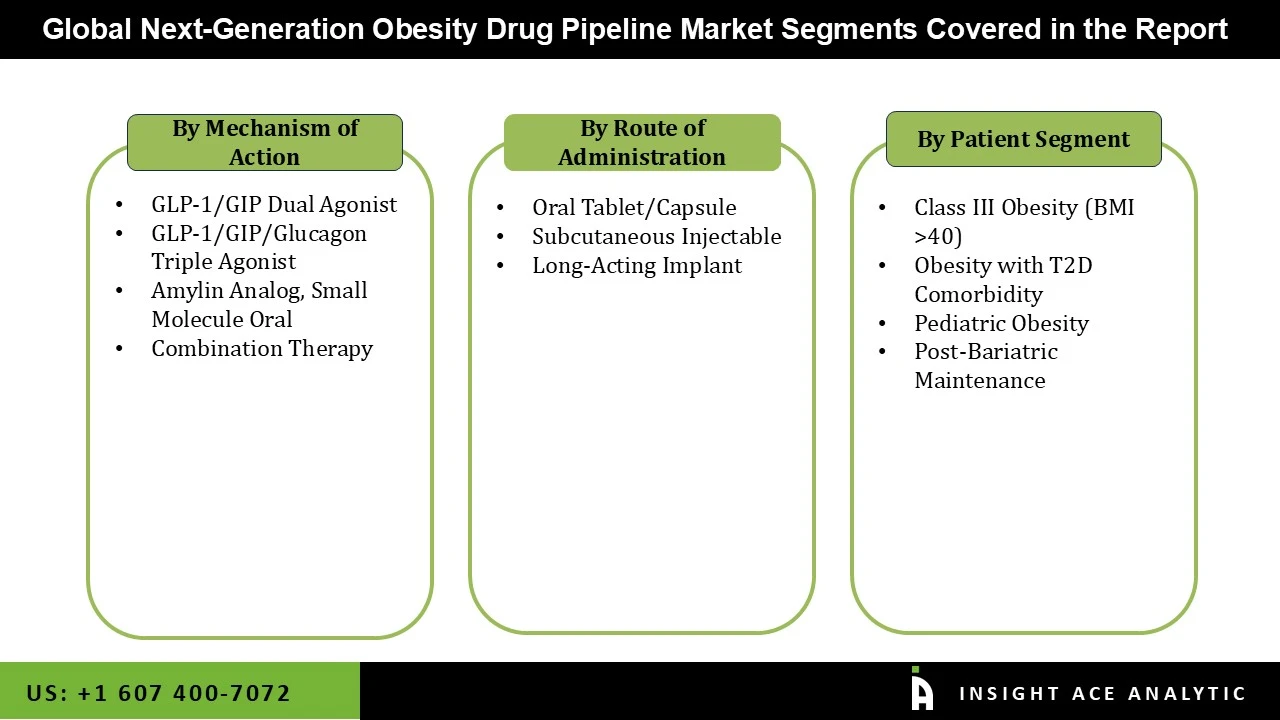
Next-generation Obesity Drug Pipeline Market by Mechanism of Action-
• GLP-1/GIP Dual Agonist
• Amylin Analog
• GLP-1/GIP/Glucagon Triple Agonist
• Small Molecule Oral
• Combination Therapy
Next-generation Obesity Drug Pipeline Market by Patient Segment-
• Obesity with T2D Comorbidity
• Class III Obesity (BMI >40)
• Pediatric Obesity
• Post-Bariatric Maintenance
Next-generation Obesity Drug Pipeline Market By Region-
North America-
• The US
• Canada
Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Next generation Obesity Drug Pipeline Market Size is valued at USD 23.13 Bn in 2025 and is predicted to reach USD 121.01 Bn by the year 2035
Next generation Obesity Drug Pipeline Market Size is predicted to grow at a 18.2% CAGR during the forecast period for 2026 to 2035.
Pfizer, Eli Lilly and Company, Amgen, Novo Nordisk, Carmot Therapeutics, Structure Therapeutics, Metsera, and Zealand Pharma.
Next generation Obesity Drug Pipeline Market is segmented into Route of Administration, Mechanism of Action, Patient Segment, and By Region
North America region is leading the Next generation Obesity Drug Pipeline Market.