Neurotech Devices Market Size, Scope and Analysis 2026 to 2035
What is Neurotech Devices Market Size?
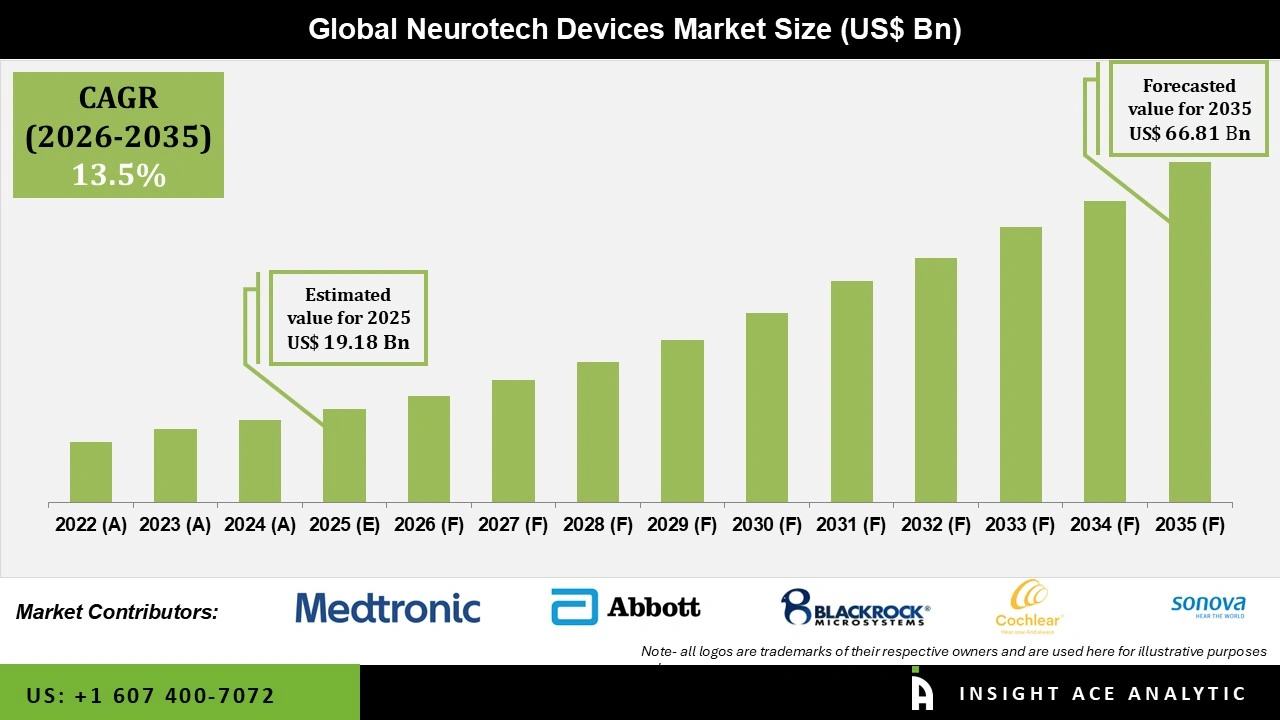
Global Neurotech Devices Market Size is valued at USD 19.18 Bn in 2025 and is predicted to reach USD 66.81 Bn by the year 2035 at a 13.5% CAGR during the forecast period for 2026 to 2035.
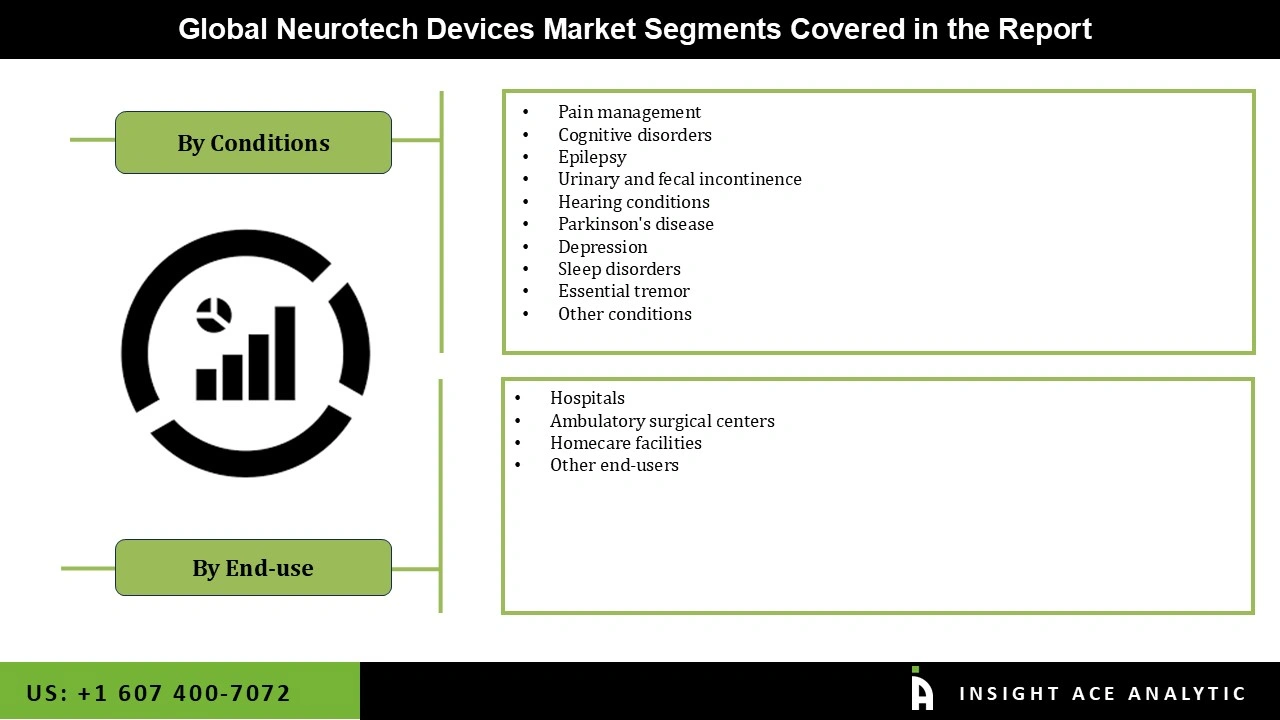
Neurotech Devices Market Size, Share & Trends Analysis Distribution by Condition (Pain Management, Epilepsy, Parkinson's Disease, Urinary and Fecal Incontinence, Cognitive Disorders, Depression, Sleep Disorders, Hearing Conditions, Essential Tremor, and Others), End-user (Hospitals, Homecare Facilities, Ambulatory Surgical Centers, and Others), by region and Segment Forecasts, 2026 to 2035.
Neurotech Devices Market Key Takeaways:
|
The medical and technological instruments intended to observe, evaluate, and communicate with the nervous system specifically, the brain and spinal cord are referred to as neurotechnology (Neurotech) devices. People with neurological disorders are diagnosed and treated with these gadgets. In both clinical and non-clinical contexts, they are also employed to improve cognitive and motor abilities. By assisting medical professionals and researchers in better understanding and treating disorders affecting the brain and nervous system, neurotech gadgets are having a significant impact on healthcare. The neurotech devices market is expected to expand significantly over the next ten years due to an increase in neurological illnesses and investments in brain research.
The need for cutting-edge diagnostic and therapeutic neurotech devices is being driven by the increasing incidence of neurological conditions such as epilepsy, Parkinson's disease, and Alzheimer's disease. About 6.9 million Americans 65 and older have Alzheimer's, with 73% of those affected being 75 years of age or older, according to Alzheimer's Disease Facts and Figures 2024. Additionally, the neurotech devices market is expanding due to the increased prevalence of depression, especially in the fields of brain monitoring and neurostimulation technology. The need for neurostimulation devices including Deep Brain Stimulation (DBS) implants, Vagus Nerve Stimulation (VNS), and Transcranial Magnetic Stimulation (TMS) is rising as more individuals experience treatment-resistant depression. Furthermore, the market for neurotech devices is mostly driven by government funding for research into neurological illnesses.
In addition, the neurotech devices market is significantly changing as a result of the convergence of growing demand for non-invasive therapies, medical electronics downsizing, and neuroscience breakthroughs. However, one of the biggest obstacles to their broad use worldwide is still the expensive cost of neurotech devices. The concerns about data security and privacy with brain-computer interface devices may also hinder the neurotech devices market expansion. On the other hand, the neurotech devices market sees fresh prospects from continued research and development to create innovative devices for conditions like Alzheimer's, Parkinson's, and epilepsy. The effectiveness of therapy will be improved by integrating gadgets with cloud computing and artificial intelligence. Neurostimulation therapy indications are growing as a result of partnerships between pharmaceutical and device manufacturers.
Competitive Landscape
Which are the Leading Players in Neurotech Devices Market?
- Medtronic plc
- Cochlear Limited
- Control Bionics, Inc
- Advanced Bionics AG (Sonova)
- Integra LifeSciences
- Boston Scientific Corporation
- Abbott Laboratories
- BrainCo, Inc
- LivaNova PLC
- NeuroSigma, Inc.
- NeuroVigil Inc.
- Nihon Kohden Corporation
- Blackrock Microsystems
- Natus Medical Incorporated
- Neuralink Corp
Market Dynamics
Driver
Rising Geriatric Population
The neurotech devices market is expanding due in large part to the aging of the world's population. According to a United Nations assessment, the number of individuals 65 and older is expected to nearly quadruple from 727 million in 2020 to over 1.5 billion by 2050 as life expectancies rise globally. In the upcoming decades, it is anticipated that the prevalence of neurological conditions such as Parkinson's disease and Alzheimer's disease would increase significantly as the baby boomer population continues to enter their senior years. The need for cutting-edge neurotech solutions that can enhance quality of life and enable independent aging is being fueled by the expanding geriatric population. Additionally, brain computer interfaces, which convert brain signals into movement orders for assistance daily living devices or prosthetic limbs, are among the technologies that are becoming more and more significant. In order to cure neurological movement disorders by deep brain stimulation, companies are also creating neural implants.
Restrain/Challenge
Growing Ethical Concerns
The development and uptake of neurotech devices are severely hampered by ethical issues, which may limit the market expansion. Patient autonomy and consent are fundamental ethical concerns. Patients must be fully informed and provide their agreement before using neurotech devices, particularly those that involve deep brain stimulation or brain implants. It is a difficult and continuous task to guarantee that patients completely understand the possible risks, advantages, and long-term effects of these procedures. This is made more difficult by the cognitive deficits that people with neurological diseases frequently exhibit, which cast doubt on their ability to give informed permission. Furthermore, there are ethical conundrums with data security and privacy as well. Sensitive brain data is frequently collected and sent by neurotech devices, which raises questions regarding how this data is shared, kept, and used. Strict precautions and ethical requirements are necessary to preserve patient privacy due to the possibility of neurological data being misused, either by unauthorized third parties or even inside the healthcare system.
Pain Management Segment is Expected to Drive the Neurotech Devices Market
The pain management category held the largest share in the Neurotech Devices market in 2025 principally caused by the increasing incidence of chronic neurological and musculoskeletal pain disorders worldwide, including complex regional pain syndrome, migraine, neuropathic pain, failed back surgery syndrome, and pain associated with spinal cord injuries. Neurostimulation-based treatments, such as spinal cord stimulators (SCS), peripheral nerve stimulation (PNS), deep brain stimulation (DBS), and transcutaneous electrical nerve stimulation (TENS) devices, have become much more popular as a result of growing awareness of the drawbacks and adverse effects of long-term opioid therapy. Additionally, technological innovations include wireless wearable neuromodulation devices, closed-loop stimulation systems, rechargeable implantable pulse generators, and implants compatible with MRIs are enhancing patient compliance and therapeutic results.
Hospitals Segment is Growing at the Highest Rate in the Neurotech Devices Market
In 2025, the hospitals category dominated the Neurotech Devices market. This expansion can be linked to increased public and private investment in healthcare settings to build reputable healthcare facilities. Neuromodulation, an invasive process that entails surgically implanting an electrode in the afflicted area of the brain, is the most prevalent use of adaptive neurotech devices in hospitals. Nonetheless, this market segment is anticipated to expand in the upcoming years due to these possible benefits. To execute a wide range of brain diagnostics and treatments, hospitals also have highly qualified and experienced staff, specialized equipment, and state-of-the-art neurotech devices. Furthermore, because of the continuous investments in medical technology and the rising incidence of neurological conditions that need for specialist care, hospitals play a crucial role in preserving a stable market share.
Why Asia Pacific Led the Neurotech Devices Market?
The Neurotech Devices market was dominated by Asia Pacific region in 2025 due to its dedication to neurotechnology R&D, understanding of neurological illnesses, and healthcare infrastructure. The BRAIN Initiative and other angel groups provide Asia Pacific with a great deal of financing and assistance. A number of agencies, including the NIH and DARPA, are still making significant investments in the development of brain-computer interfaces, neurostimulation devices, and neuroprosthetics with the goal of pushing the boundaries.
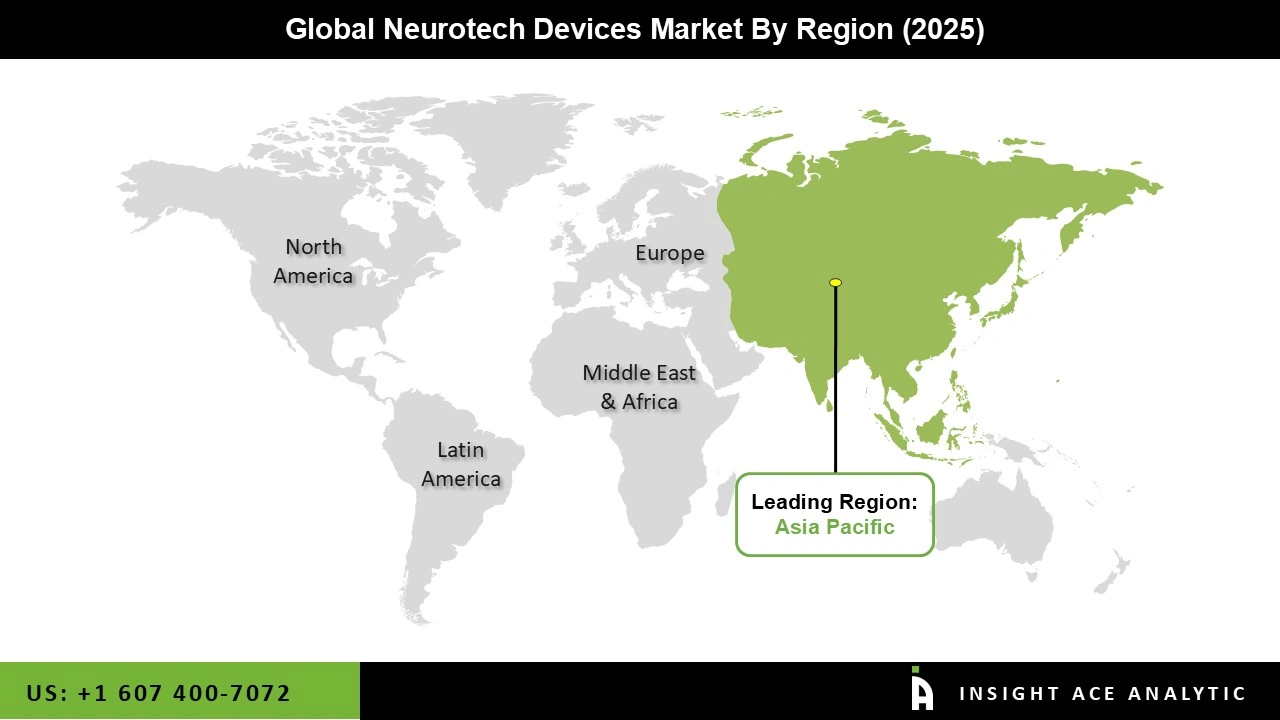
Additionally, the demand for health innovations will only increase due to advances in neurotechnology's revolutionary diagnostic and treatments, particularly given the increasing prevalence of chronic pain problems and diseases such as Parkinson's and Alzheimer's.
Neurotech Devices Market Report Scope :
| Report Attribute | Specifications |
| Market size value in 2025 | USD 19.18 Bn |
| Revenue forecast in 2035 | USD 66.81 Bn |
| Growth Rate CAGR | CAGR of 13.5% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Condition, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Medtronic plc, Cochlear Limited, Control Bionics, Inc, Advanced Bionics AG (Sonova), Integra LifeSciences, Boston Scientific Corporation, Abbott Laboratories, BrainCo, Inc, LivaNova PLC, NeuroSigma, Inc., NeuroVigil Inc., Nihon Kohden Corporation, Blackrock Microsystems, Natus Medical Incorporated, and Neuralink Corp. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation Neurotech Devices Market :
Neurotech Devices Market by Condition-
- Pain Management
- Epilepsy
- Parkinson's Disease
- Urinary and Fecal Incontinence
- Cognitive Disorders
- Depression
- Sleep Disorders
- Hearing Conditions
- Essential Tremor
- Others
Neurotech Devices Market by End-user-
- Hospitals
- Homecare Facilities
- Ambulatory Surgical Centers
- Others
Neurotech Devices Market, by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Neurotech Devices Market Size is valued at USD 19.18 Bn in 2025 and is predicted to reach USD 66.81 Bn by the year 2035
Neurotech Devices Market is expected to grow at a 13.5% CAGR during the forecast period for 2026 to 2035
Medtronic plc, Cochlear Limited, Control Bionics, Inc, Advanced Bionics AG (Sonova), Integra LifeSciences, Boston Scientific Corporation, Abbott Laboratories, BrainCo, Inc, LivaNova PLC, NeuroSigma, Inc., NeuroVigil Inc., Nihon Kohden Corporation, Blackrock Microsystems, Natus Medical Incorporated, Neuralink Corp. and Other.
Neurotech Devices Market is segmented into by Condition (Pain Management, Epilepsy, Parkinson\'s Disease, Urinary and Fecal Incontinence, Cognitive Disorders, Depression, Sleep Disorders, Hearing Conditions, Essential Tremor, and Others), End-user (Hospitals, Homecare Facilities, Ambulatory Surgical Centers, and Others) and Other.
Asia Pacific region is leading the Neurotech Devices Market.