GLP-1 Autoinjector and Pen Injector Market Size, Revenue, Forecast Report 2026 to 2035
What is GLP-1 Autoinjector and Pen Injector Market?
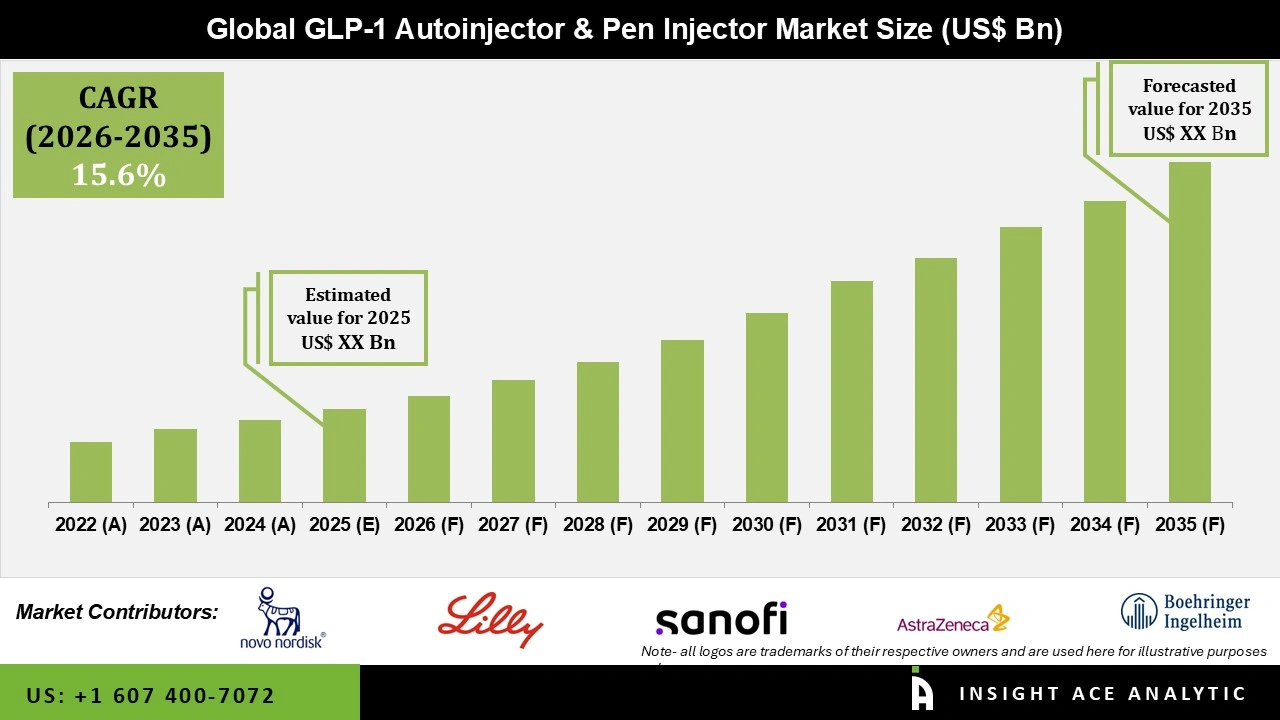
Global GLP-1 Autoinjector and Pen Injector Market is estimated to witness a rapid growth with at a 15.6% CAGR during the forecast period for 2026 to 2035.
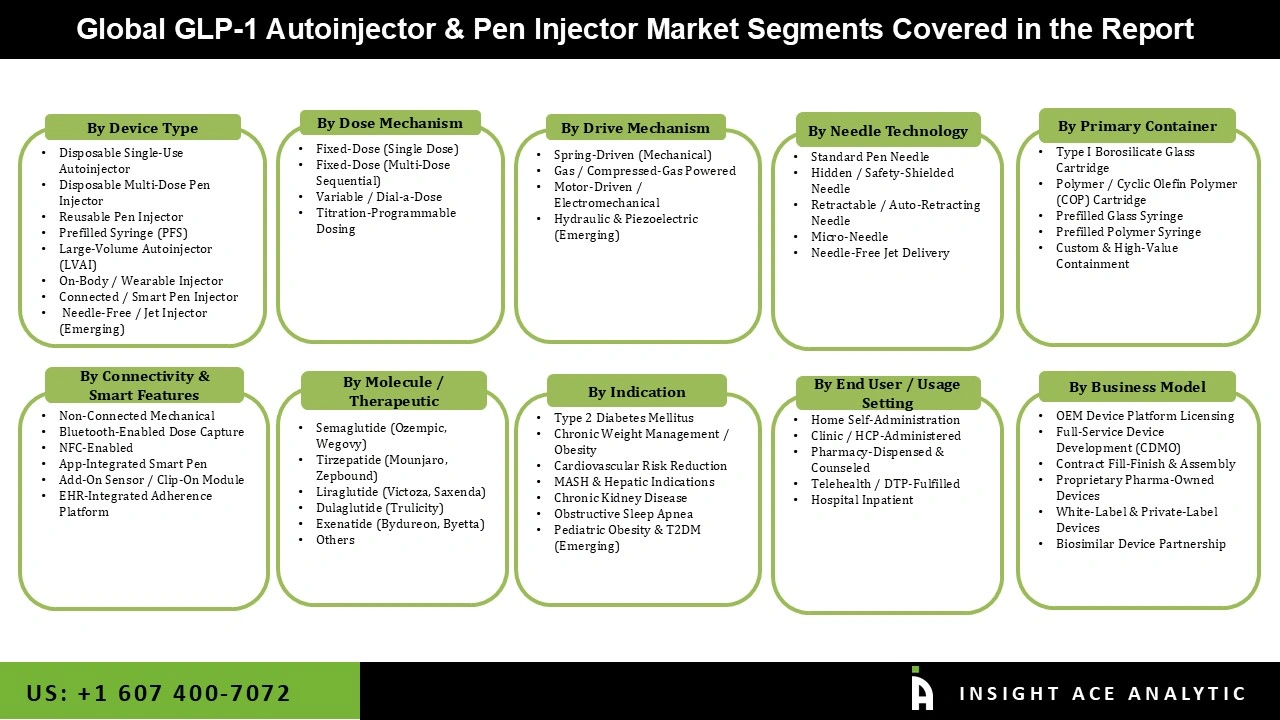
GLP-1 Autoinjector and Pen Injector Market Size, Share and Trends Analysis Distribution by Device Type (Disposable Single-Use Autoinjector, Disposable Multi-Dose Pen Injector, Reusable Pen Injector, Prefilled Syringe (PFS), Large-Volume Autoinjector (LVAI), On-Body / Wearable Injector, Connected / Smart Pen Injector, Needle-Free / Jet Injector (Emerging)), By Dose Mechanism (Fixed-Dose (Single Dose), Fixed-Dose (Multi-Dose Sequential), Variable / Dial-a-Dose, Titration-Programmable Dosing), By Drive Mechanism (Spring-Driven (Mechanical), Gas / Compressed-Gas Powered, Motor-Driven / Electromechanical, Hydraulic and Piezoelectric (Emerging)) By Needle Technology (Standard Pen Needle, Hidden / Safety-Shielded Needle, Retractable / Auto-Retracting Needle, Micro-Needle, Needle-Free Jet Delivery), By Primary Container(Type I Borosilicate Glass Cartridge, Polymer / Cyclic Olefin Polymer (COP) Cartridge, Prefilled Glass Syringe, Prefilled Polymer Syringe, Custom and High-Value Containment), Segment Forecasts, 2026 to 2035.
The market for GLP-1 autoinjectors and pen injectors has grown immensely in the wake of the rising number of patients with metabolic syndromes like Type 2 Diabetes and Obesity. GLP-1 (Glucagon-like peptide-1) agonist receptor therapies are popular incretin treatments, which work to control glucose in the bloodstream, appetite, and weight management. These incretin drugs have been administered with the help of advanced delivery systems such as autoinjectors and pen injectors to increase the effectiveness and ease of application for patients. The GLP-1 autoinjector/pen injector is a pre-filled, handheld medical device that is used for administering drugs via subcutaneous injection; whereas pen injectors usually have multi-dose capacity, and autoinjectors can be one-time use only.
The main usage of such instruments is associated with the management of diabetes through improvement of glycemic control through secretion of insulin and lowering of glucagon. Moreover, their applications in treating obesity have increased substantially due to rising demand for effective treatment of the disease. Besides, there is a trend in exploring GLP-1 treatments via such instruments as a means for reducing risk of cardiovascular problems. The development of new techniques by pharmaceutical firms such as Novo Nordisk, Eli Lilly and Company, and Sanofi is contributing to the growth of this industry through adoption of self-treatment technologies.
Competitive Landscape
Which are the Leading Players in the GLP-1 Autoinjector and Pen Injector Market?
- Novo Nordisk A/S
- Eli Lilly and Company
- Sanofi S.A.
- AstraZeneca plc
- Boehringer Ingelheim
- Amgen Inc.
- Pfizer Inc.
- Ypsomed AG
- SHL Medical
- Owen Mumford
- Stevanato Group
- Phillips-Medisize
- Nemera
- AptarGroup
- Gerresheimer AG
- Haselmeier
- Credence MedSystems
- Enable Injections
- Congruence Medical Solutions
- Weibel CDS
- Elcam Medical
- Duoject Medical Systems
- Oval Medical Technologies
- SCHOTT Pharma
- Nipro Corporation
- Corning Incorporated
- West Pharmaceutical Services
- Becton, Dickinson and Company
- AGC Inc.
- Catalent, Inc.
- Vetter Pharma
- PCI Pharma Services
- Grand River Aseptic Manufacturing
- Pfizer CentreOne
- Thermo Fisher Scientific
- Recipharm AB
- Delpharm
- Boehringer Ingelheim BioXcellence
- Simtra BioPharma Solutions
- Biocorp
- Medtronic
- Embecta Corp.
- Insulet Corporation
- Terumo Corporation
- Shanghai Haoyue Medical
- Truking Technology
- Shandong Weigao Group
- Dali Medical Devices
- Berry Star Medical
- Portal Instruments
- PharmaJet
- Sorrel Medical
- Subcuject
Market Dynamics
Driver
Rising Prevalence of Metabolic Disorders
The main factor fueling the growth of the GLP-1 Autoinjectors and Pen Injectors market is the rise in the prevalence of Type 2 Diabetes and Obesity around the world. Both of these disorders need continuous medical intervention, and GLP-1 agonists are gaining prominence as an ideal therapy owing to their ability to provide both diabetes and obesity control at once. With the rise in the number of patients being affected by these disorders worldwide, there will be a corresponding rise in demand for more patient-friendly drug delivery devices like autoinjectors and pen injectors.
Restrain/Challenge
High Cost of GLP-1 Therapies and Delivery Devices
The key constraint on the growth of the GLP-1 auto-injector and pen injector market is the expensive nature of the GLP-1 receptor agonist medication and its delivery mechanism. These drugs, which are typically prescribed for treating Type 2 Diabetes and Obesity, have a higher value than conventional medications. Besides the cost of the drug, the production of auto-injectors and pen injectors entails sophisticated engineering and technology. The resulting costs of treatment may be a barrier to accessing healthcare products, especially in cost-conscious or emerging economies.
Disposable Multi-Dose Pen Injector Segment is Expected to Drive the GLP-1 Autoinjector and Pen Injector Market
The disposable multidose pen injector category forms an important component of the GLP-1 autoinjector and pen injector market due to their extensive usage and patient preference for convenience and home treatment. These pen injectors are pre-filled with several doses that can be easily controlled by the user with the help of dials. These products are very effective in treating chronic diseases like Type 2 diabetes and obesity. The advantage of using these pen injectors for more than one injection and not requiring replacement makes them more economical and easy to use. Besides, easy-to-train users, enhanced dosing precision, and greater physician acceptance have led to their successful penetration in the market.
CBD (Cannabidiol) Segment is Growing at the Highest Rate in the GLP-1 Autoinjector and Pen Injector Market
The variable (dial-a-dose) dosing mechanism is the fastest-growing segment in the GLP-1 autoinjector and pen injector market due to its flexibility and strong alignment with clinical treatment needs, as GLP-1 therapies often require gradual dose escalation to optimize efficacy and minimize side effects; these devices enable easy dose adjustment, allowing personalized treatment for conditions such as Type 2 Diabetes and Obesity, while offering intuitive functionality, improved dosing accuracy, and higher patient acceptance compared to fixed-dose systems, thereby driving their rapid adoption and growth.
Why North America Led the GLP-1 Autoinjector and Pen Injector Market?
North America leads the GLP-1 autoinjector and pen injector market primarily due to the high prevalence of Type 2 Diabetes and Obesity, which has significantly increased the demand for GLP-1 therapies and their delivery devices. The region benefits from advanced healthcare infrastructure, high healthcare spending, and strong access to innovative treatments, enabling early adoption of autoinjectors and pen injectors.
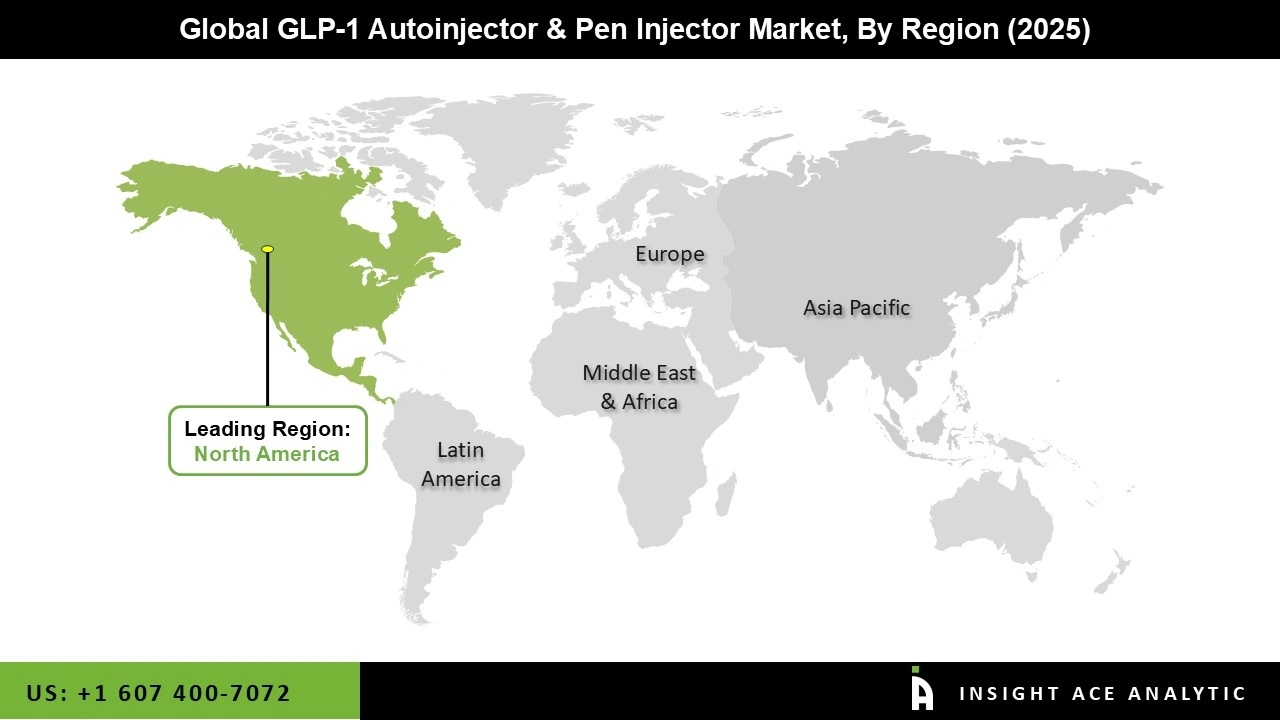
Additionally, favorable reimbursement policies, strong presence of leading pharmaceutical companies, and high patient awareness regarding self-administration devices further support market growth. The widespread shift toward home-based care and the rapid uptake of newer GLP-1 drugs, particularly in the United States, continue to reinforce North America’s dominant position in the market.
GLP-1 Autoinjector and Pen Injector Market Report Scope:
| Report Attribute | Specifications |
| Growth Rate CAGR | CAGR of 15.6% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Device Type, Dose Mechanism, Drive Mechanism, Needle Technology, Primary Container, Connectivity and Smart Features, Molecule / Therapeutic, Indication, End User / Usage Setting, Business Model and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East and Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Novo Nordisk A/S, Eli Lilly and Company, Sanofi S.A., AstraZeneca plc, Boehringer Ingelheim, Amgen Inc., Pfizer Inc., Device Platform Developers and Design Houses, Ypsomed AG (UnoPen, YpsoMate, ServoPen, SmartPilot), SHL Medical, Owen Mumford (Aidaptus, Autoject) |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentations of GLP-1 Autoinjector and Pen Injector Market:
GLP-1 Autoinjector and Pen Injector Market, by Device Type-
- Disposable Single-Use Autoinjector
- Disposable Multi-Dose Pen Injector
- Reusable Pen Injector
- Prefilled Syringe (PFS)
- Large-Volume Autoinjector (LVAI)
- On-Body / Wearable Injector
- Connected / Smart Pen Injector
- Needle-Free / Jet Injector (Emerging)
GLP-1 Autoinjector and Pen Injector Market, by Dose Mechanism -
- Fixed-Dose (Single Dose)
- Fixed-Dose (Multi-Dose Sequential)
- Variable / Dial-a-Dose
- Titration-Programmable Dosing
GLP-1 Autoinjector and Pen Injector Market, by Drive Mechanism -
- Spring-Driven (Mechanical)
- Gas / Compressed-Gas Powered
- Motor-Driven / Electromechanical
- Hydraulic and Piezoelectric (Emerging)
GLP-1 Autoinjector and Pen Injector Market, by Needle Technology-
- Standard Pen Needle
- Hidden / Safety-Shielded Needle
- Retractable / Auto-Retracting Needle
- Micro-Needle
- Needle-Free Jet Delivery
GLP-1 Autoinjector and Pen Injector Market, by Primary Container-
- Type I Borosilicate Glass Cartridge
- Polymer / Cyclic Olefin Polymer (COP) Cartridge
- Prefilled Glass Syringe
- Prefilled Polymer Syringe
- Custom and High-Value Containment
GLP-1 Autoinjector and Pen Injector Market, by Connectivity and Smart Features-
- Non-Connected Mechanical
- Bluetooth-Enabled Dose Capture
- NFC-Enabled
- App-Integrated Smart Pen
- Add-On Sensor / Clip-On Module
- EHR-Integrated Adherence Platform
GLP-1 Autoinjector and Pen Injector Market, by Molecule / Therapeutic-
- Semaglutide (Ozempic, Wegovy)
- Tirzepatide (Mounjaro, Zepbound)
- Liraglutide (Victoza, Saxenda)
- Dulaglutide (Trulicity)
- Exenatide (Bydureon, Byetta)
- Orforglipron (Emerging Oral — Pen Not Applicable)
- CagriSema (Semaglutide + Cagrilintide)
- Retatrutide (Triple Agonist)
- MariTide (Monthly GIPR / GLP-1)
- Survodutide (GLP-1 / Glucagon Dual)
- Biosimilar and Generic GLP-1 Molecules
GLP-1 Autoinjector and Pen Injector Market, by Indication-
- Type 2 Diabetes Mellitus
- Chronic Weight Management / Obesity
- Cardiovascular Risk Reduction
- MASH and Hepatic Indications
- Chronic Kidney Disease
- Obstructive Sleep Apnea
- Pediatric Obesity and T2DM (Emerging)
GLP-1 Autoinjector and Pen Injector Market, by End User / Usage Setting-
- Home Self-Administration
- Clinic / HCP-Administered
- Pharmacy-Dispensed and Counseled
- Telehealth / DTP-Fulfilled
- Hospital Inpatient
GLP-1 Autoinjector and Pen Injector Market, by Business Model-
- OEM Device Platform Licensing
- Full-Service Device Development (CDMO)
- Contract Fill-Finish and Assembly
- Proprietary Pharma-Owned Devices
- White-Label and Private-Label Devices
- Biosimilar Device Partnership
GLP-1 Autoinjector and Pen Injector Market, by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East and Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
GLP-1 Autoinjector and Pen Injector Market is estimated to witness a rapid growth with at a 15.6% CAGR during the forecast period for 2026 to 2035.
Novo Nordisk A/S, Eli Lilly and Company, Sanofi S.A., AstraZeneca plc, Boehringer Ingelheim, Amgen Inc., Pfizer Inc., Device Platform Developers and Design Houses, Ypsomed AG (UnoPen, YpsoMate, ServoPen, SmartPilot), SHL Medical, Owen Mumford (Aidaptus, Autoject) and Others.
GLP-1 Autoinjector and Pen Injector Market is segmented into Device Type, Dose Mechanism, Drive Mechanism, Needle Technology, Primary Container, Connectivity and Smart Features, Molecule / Therapeutic, Indication, End User / Usage Setting, Business Model and Other.
North America region is leading the GLP-1 Autoinjector and Pen Injector Market.