Nerve Capping Device Market Size, Scope, Revenue Report 2026 to 2035
What is Nerve Capping Device Market Size?
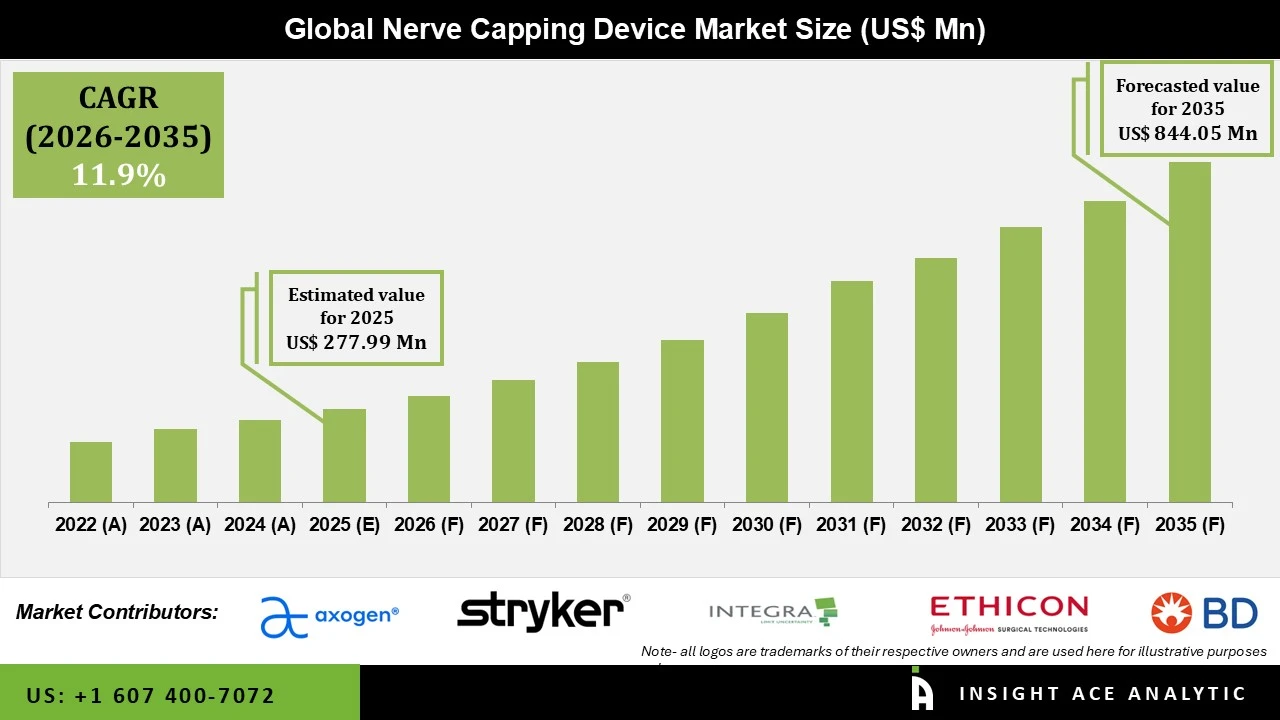
Nerve Capping Device Market Size is valued at USD 277.99 Mn in 2025 and is predicted to reach USD 844.05 Mn by the year 2035 at a 11.9% CAGR during the forecast period for 2026 to 2035.
Nerve Capping Device Market Size, Share & Trends Analysis Distribution by Product Type (Bioabsorbable Polymers (PLGA, PCL, PLA, Composites), Silicone/Non-absorbable, Biologic/Collagen-Based, and Others), Surgical Specialty (Vascular & Amputation Care, Orthopedics/Trauma, Plastic/Reconstructive Surgery, and Others), Device Architecture (Closed-End Cap, Sleeve Cap, Semi-Closed / Restricted Outlet, and Others), End-user (Hospitals, Specialty Clinics, Ambulatory Surgery Centers, and Others), and Segment Forecasts, 2026 to 2035
A nerve capping device is a specialist medical implant used, especially following surgical procedures like amputations, to control or prevent pain resulting from injured or severed peripheral nerves. Cutting a nerve can result in a painful growth called a neuroma, which can cause hypersensitivity or persistent discomfort. By enclosing and shielding the nerve's end, nerve capping devices serve as a physical barrier that lessens aberrant nerve regrowth and keeps surrounding tissues from irritating the nerve. A paradigm shift in pain management toward intraoperative prevention in peripheral nerve handling procedures, such as trauma surgeries, amputations, and difficult reconstructions, is driving the nerve capping device market's expansion.
The nerve capping device market is being driven by the rising incidence of peripheral nerve injuries and post-amputation problems. These devices assist in preventing painful neuroma growth and enhancing surgical results. Advanced nerve management technologies are also becoming more and more necessary due to the increasing number of trauma cases, diabetes-related amputations, and complicated surgical procedures. As healthcare systems prioritize enhancing patient quality of life and lowering chronic pain, the trend is anticipated to continue. Additionally, the safety and efficacy of nerve capping devices are being improved by developments in biomaterials, such as bioresorbable and biocompatible polymers. Furthermore, these advancements are increasing their use in pain management treatments and reconstructive surgery, especially in sophisticated healthcare environments.
In addition, continuing technical developments and rising R&D expenditures are projected to influence the nerve capping device market. The market is expanding due in part to favorable regulatory backing for novel medical implants and an increasing emphasis on minimally invasive surgical methods. Additionally, manufacturers are putting more of an emphasis on creating specialized tools for certain surgical requirements, which suggests a move toward precision-based and customized healthcare solutions. The adoption is also anticipated to increase as healthcare personnel become more knowledgeable about improved nerve restoration treatments. However, the nerve capping device market confronts certain obstacles, such as the high cost of treatments and devices, as well as low knowledge in developing nations.
Competitive Landscape
Which are the Leading Players in the Nerve Capping Device Market?
• Medtronic plc
• Smith+Nephew plc
• Axogen, Inc.
• BD (Becton, Dickinson and Company)
• Stryker Corporation
• Johnson & Johnson (Ethicon)
• DePuy Synthes (Johnson & Johnson)
• B. Braun Melsungen AG
• Coloplast A/S
• Integra LifeSciences Holdings Corporation
Market Dynamics
Driver
Shift to Pain Prevention During Surgery
The nerve capping device market is mostly driven by a major paradigm shift in post-surgical treatment, from reactive postoperative pain management to proactive intraoperative avoidance. In the past, peripheral nerve transection procedures such as amputations and intricate trauma surgeries mainly relied on systemic analgesics, which frequently offered insufficient relief from long-term ailments like neuroma discomfort. Furthermore, a growing body of clinical evidence indicates that treating the nerve stump during surgery can significantly lower the risk of long-term problems. Nerve capping devices are particularly made to prevent symptomatic neuromas, which are thought to occur in up to 25% of major limb amputees. Moreover, surgeons are being compelled to include these devices in their routine workflows, especially within enhanced recovery after surgery (ERAS) protocols, due to the desire for first-pass treatments that improve long-term patient outcomes and decrease the need for secondary interventions.
Restrain/Challenge
High Device and Procedure Costs Combined with Reimbursement Obstacles
The nerve capping device market is constrained by high device and procedure costs. More significantly, the funding for these specialty devices is sometimes complicated and uneven among various payers and healthcare systems. The hospitals and ambulatory surgery centers are worried about financial remuneration because unambiguous payment codes specifically for nerve capping treatments are still being developed in several locations. This may cause procurement hesitancy, particularly in cost-conscious settings where conventional, less expensive nerve stump treatment techniques may still be preferred despite perhaps worse long-term results. Additionally, the value proposition for budget holders is complicated by the dearth of solid, extensive health economic studies proving the long-term cost-effectiveness of these implants in lowering chronic pain treatment and revision surgeries.
Bioabsorbable Polymers Segment is Expected to Drive the Nerve Capping Device Market
The bioabsorbable polymers category held the largest share in the Nerve Capping Device market in 2025. These polymers are favored because of their special ability to dissolve safely inside the body, which removes the need for removal surgery following nerve capping procedures. Because of their biocompatibility and degradation characteristics, bioabsorbable polymers are very useful in nerve regeneration and the prevention of amputation neuromas. Surgeons looking for long-term, patient-friendly solutions are increasingly choosing bioabsorbable polymers as the need for minimally invasive and efficient surgical solutions grows. Additionally, it is anticipated that the continued development of more sophisticated bioabsorbable polymers with enhanced mechanical qualities and quicker rates of degradation would spur additional growth in the nerve capping device market.
Closed-End Cap Segment is Growing at the Highest Rate in the Nerve Capping Device Market
In 2025, the closed-end cap category dominated the Nerve Capping Device market because it works well to isolate damaged nerve terminals and stop the development of neuromas. These devices minimize aberrant nerve regrowth and lessen discomfort following surgery because of their sealed structure, which completely encloses the nerve stump. They are highly favored in peripheral nerve surgeries and amputation treatments because they can offer a regulated microenvironment for nerve repair. Furthermore, as closed-end caps gradually deteriorate without needing to be removed, developments in bioresorbable materials are improving their safety and long-term results. The need for closed-end cap solutions is also being accelerated by the expanding use of minimally invasive surgical methods and the increased focus on enhancing patient recovery outcomes.
Why North America Led the Nerve Capping Device Market?
The Nerve Capping Device market was dominated by North America region in 2025 motivated by the high frequency of peripheral nerve injuries, rising rates of amputations, and a solid healthcare system. The growing prevalence of long-term illnesses such as diabetes and vascular disorders, which frequently result in limb loss, is driving up demand for sophisticated pain control techniques, such as nerve capping devices. Further promoting market growth are the existence of top producers of medical devices and ongoing biomaterials technology developments.
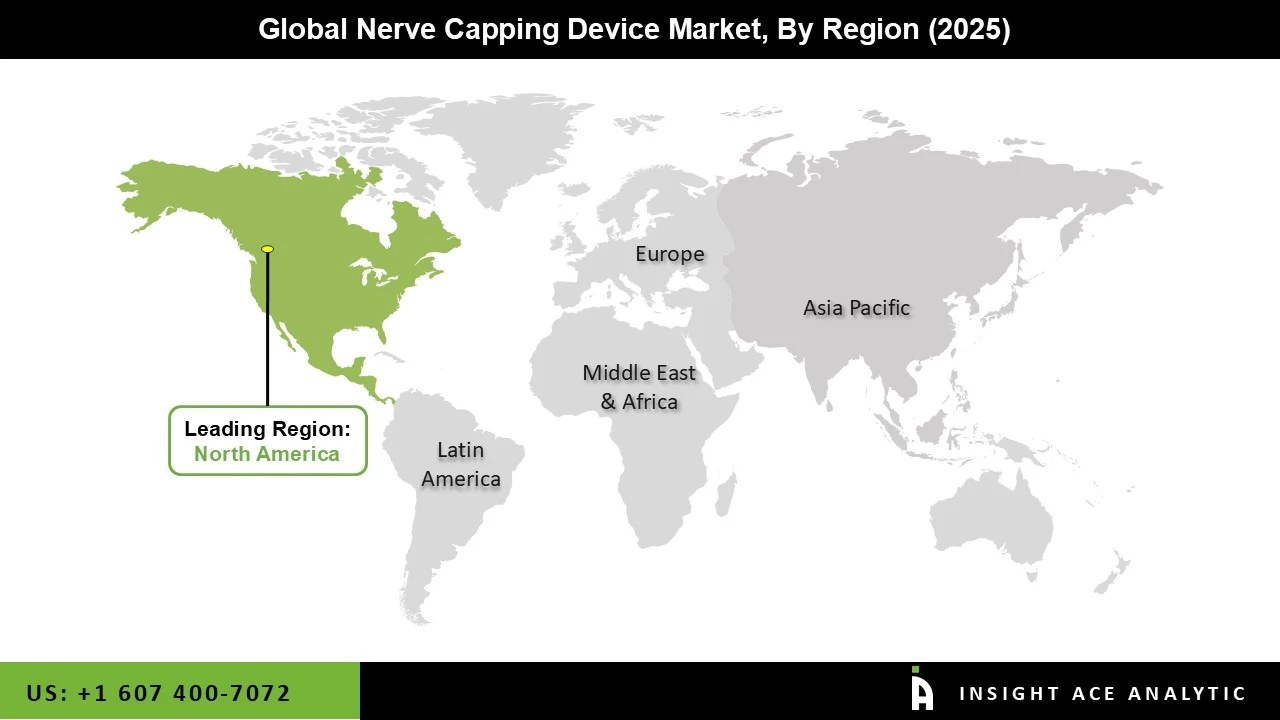
Additionally, the area benefits from high healthcare spending and early adoption of cutting-edge surgical techniques, which increases the uptake of these gadgets. Moreover, the increased popularity is also a result of healthcare providers becoming more knowledgeable about post-surgical pain management and neuroma avoidance.
Nerve Capping Device Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 277.99 Mn |
| Revenue forecast in 2035 | USD 844.05 Mn |
| Growth Rate CAGR | CAGR of 11.9% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Mn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Surgical Specialty, Device Architecture, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Medtronic plc, Smith+Nephew plc, Axogen, Inc., BD (Becton, Dickinson and Company), Stryker Corporation, Johnson & Johnson (Ethicon), DePuy Synthes (Johnson & Johnson), B. Braun Melsungen AG, Coloplast A/S, and Integra LifeSciences Holdings Corporation |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Nerve Capping Device Market by Product Type-
• Bioabsorbable Polymers
o PLGA
o PCL
o PLA
o Composites
• Silicone/Non-absorbable
• Biologic/Collagen-Based
• Others
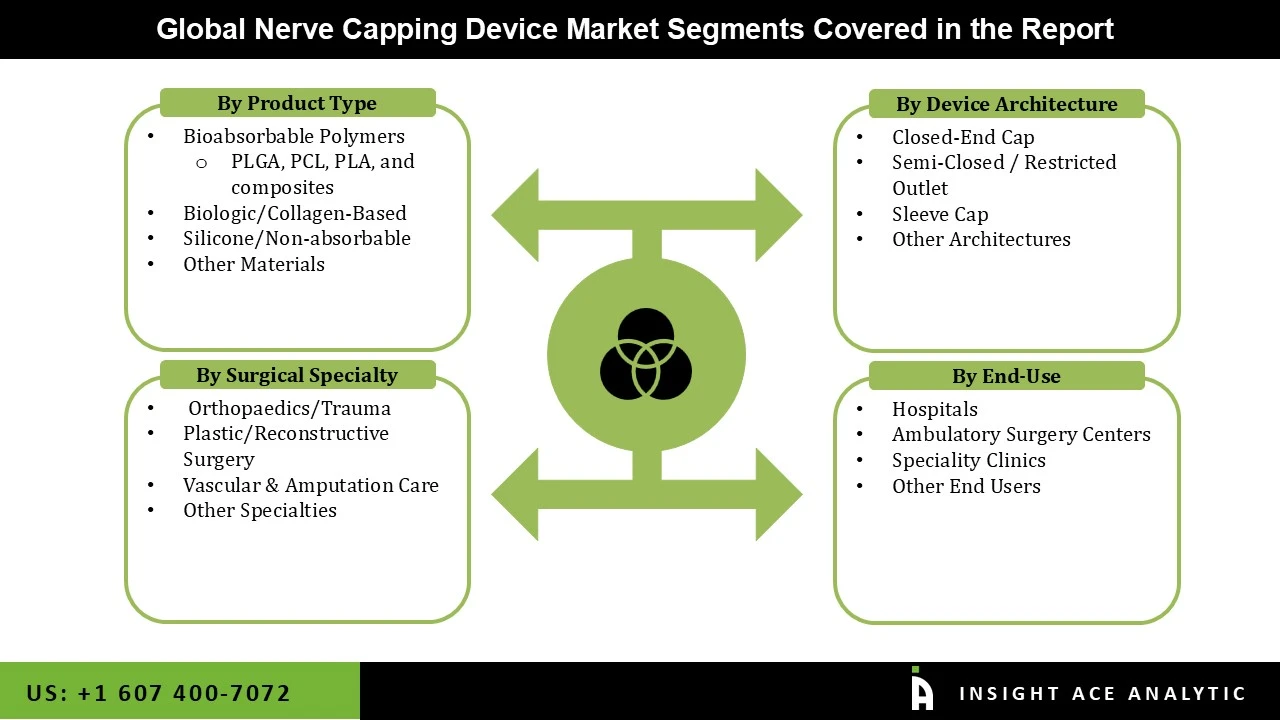
Nerve Capping Device Market by Surgical Specialty-
• Vascular & Amputation Care
• Orthopedics/Trauma
• Plastic/Reconstructive Surgery
• Others
Nerve Capping Device Market by Device Architecture-
• Closed-End Cap
• Sleeve Cap
• Semi-Closed / Restricted Outlet
• Others
Nerve Capping Device Market by End-user-
• Hospitals
• Specialty Clinics
• Ambulatory Surgery Centers
• Others
By Region-
North America-
• The US
• Canada
Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Nerve Capping Device Market Size is valued at USD 277.99 Mn in 2025 and is predicted to reach USD 844.05 Mn by the year 2035
Nerve Capping Device Market Size is predicted to grow at a 11.9% CAGR during the forecast period for 2026 to 2035.
Medtronic plc, Smith+Nephew plc, Axogen, Inc., BD (Becton, Dickinson and Company), Stryker Corporation, Johnson & Johnson (Ethicon), DePuy Synthes (Johnson & Johnson), B. Braun Melsungen AG, Coloplast A/S, and Integra LifeSciences Holdings Corporation
Nerve Capping Device Market is segmented into Product Type, Surgical Specialty, Device Architecture, End-user, and By Region
North America region is leading the Nerve Capping Device Market.