CAUTI Prevention Urology Products Market Size, Revenue, Forecast Report 2026 to 2035
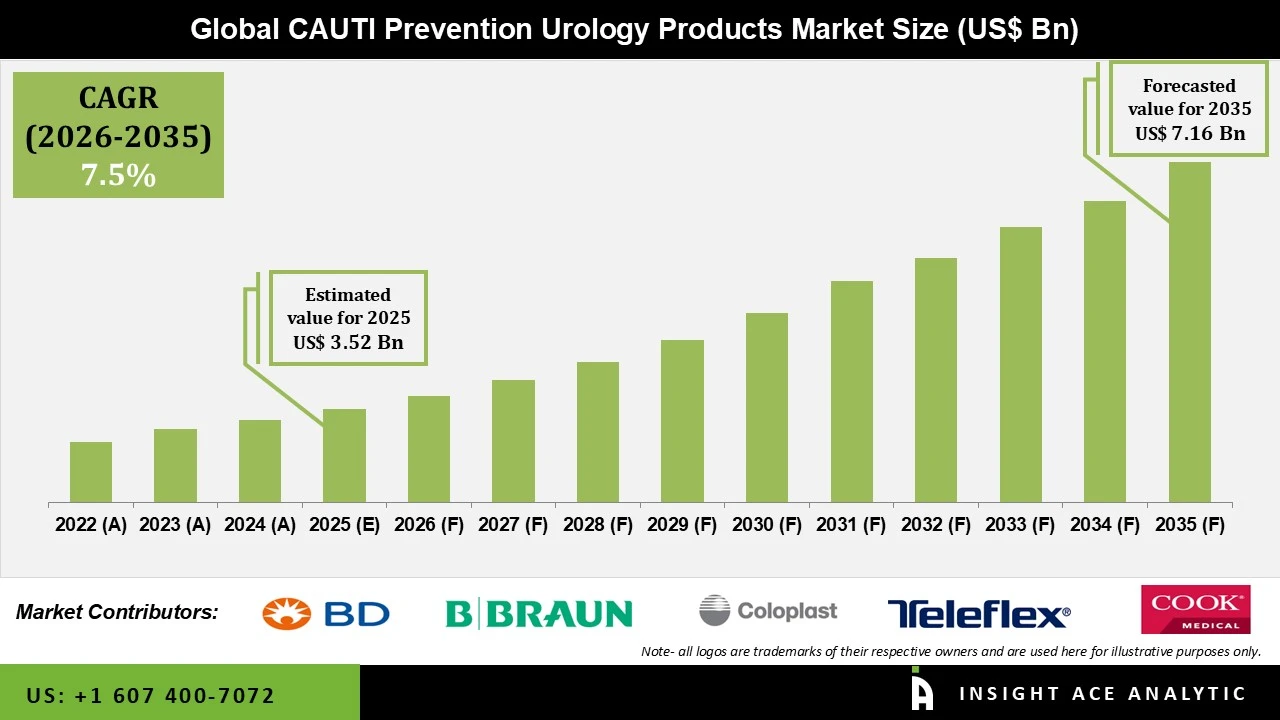
CAUTI Prevention Urology Products Market Size is valued at USD 3.52 Bn in 2025 and is predicted to reach USD 7.16 Bn by the year 2035 at a 7.5% CAGR during the forecast period for 2026 to 2035.
CAUTI Prevention Urology Products Market Size, Share & Trends Analysis Distribution by Product Type (Preventive Drainage Devices (Primary Prevention), External (Non-Invasive) Collection Systems [Intermittent (Single-Use) Catheters [Compact/Travel Intermittent, Antibacterial/Antimicrobial-Coated], Indwelling (Foley) Catheters [Silver-Alloy Coated Foley, Drug-Eluting Catheters, Silicone-Only Foley]), Procedural Support & "Insertion Bundles" (Aseptic Prevention) (Pre-Packaged Aseptic Trays, Sterile Lubricant & Cleansing Solutions, Bladder Scanners (Portable Ultrasound)), and Sterile Circuit & Maintenance Systems (Mechanical Prevention) (Closed Drainage Bags [Urine Meters, Anti-Reflux Systems], Needleless Sampling Ports, Catheter Stabilization/Securement Devices)), Application (Neurogenic Bladder & Spinal Cord Injury (SCI), Urinary Incontinence Management, Benign Prostatic Hyperplasia (BPH) and Obstructive Urology, Acute Surgical/Perioperative Care), End-user (Hospitals, Home Care Setting, Long Term Care Facilities, Ambulatory Surgical Centers (ASCs), and Others), and Segment Forecasts, 2026 to 2035
CAUTI prevention urology products are medical devices and technologies created especially to lower the risk of CAUTIs (Catheter-Associated Urinary Tract Infections) in patients who need to have a urinary catheter. Antimicrobial or silver-coated urinary catheters, antimicrobial drainage bags, catheter insertion kits, closed urine drainage systems, and antiseptic cleaning solutions are some of these products. They function by reducing the amount of bacterial colonization, preserving a sterile closed drainage system, and avoiding needless catheter movement that could contaminate the urinary tract. The growing awareness of hospital-acquired infections, an increase in hospital admissions, and an increase in the use of indwelling catheters are the factors driving the CAUTI prevention urology products market growth.
The CAUTI prevention urology products market is expanding due in large part to the rising incidence of catheter-associated urinary tract infections (CAUTIs), as medical institutions concentrate on enhancing patient safety and infection control. The demand for efficient preventative measures such as antimicrobial catheters, closed drainage systems, and catheter securement devices has increased due to the extensive use of urinary catheters in hospitals, especially among critically ill and post-surgical patients. Furthermore, the CAUTI prevention urology products market is also being influenced by the aging population and the increasing prevalence of chronic illnesses that necessitate long-term catheterization. In order to lower infection risks and enhance therapeutic results, healthcare providers are also placing a strong emphasis on the use of cutting-edge technologies, such as silver-alloy and antimicrobial-coated catheters.
Additionally, it is anticipated that the implementation of stringent healthcare laws and infection prevention guidelines, along with growing awareness of hospital-acquired infections, will facilitate the CAUTI prevention urology products market expansion. Improved catheter management methods and preventive products are being promoted by governments and healthcare organizations in an effort to lower treatment costs and hospital stays related to infections. In addition to continuing research and development efforts, technological developments in urology devices are also influencing the industry by bringing in more effective and patient-friendly solutions. The use of CAUTI prevention urology products is anticipated to increase consistently in the upcoming years as healthcare infrastructure continues to develop, especially in emerging economies.
Competitive Landscape
Which are the Leading Players in CAUTI Prevention Urology Products Market?
• BD
• Medtronic
• Coloplast
• Teleflex Incorporated
• B. Braun SE
• Cook Medical
• Hollister Incorporated
• Cardinal Health
• Merit Medical
• Medline Industries, LP.
• Poiesis Medical LLC
• ConvaTec Group PLC
• Boston Scientific Corporation
• Others
Market Dynamics
Driver
Rising Healthcare Costs and Focus on Patient Safety
In order to effectively manage the massive clinical and financial burden of catheter-associated urinary tract infections (CAUTIs), healthcare systems in both established and emerging nations are seeing an increase in infection prevention spending. CAUTIs are one of the most prevalent types of hospital-acquired infections (HAIs) that can be prevented with appropriate preventive measures. They frequently result in longer hospital stays, higher antibiotic use, higher rates of patient readmission, and serious complications like sepsis or bloodstream infections, all of which collectively result in a significant decrease in treatment costs. As a result, hospitals and healthcare institutions are shifting their spending from treatment to prevention. It is the primary cause of the increase in a variety of sophisticated CAUTI prevention urology products, including hydrophilic and antimicrobial coated catheters, closed urinary drainage systems, securement devices, and catheter care kits. Therefore, rising healthcare costs and the focus on patient safety continue to be important drivers of the CAUTI prevention urology products market.
Restrain/Challenge
Growing Inconsistency Reduces the Perception of Product Value
Despite the availability of clinically recommended CAUTI preventive measures, there is a great deal of diversity in how these protocols are implemented at healthcare facilities due to variations in institutional policy, staff training levels, and resource availability. Compliance inspections are typically relatively sporadic, and many hospitals lack established protocols for catheter insertion, maintenance, and removal. Therefore, when these items are not integrated into a well-organized, protocol-driven system, the effectiveness of CAUTI prevention urology products such as antimicrobial catheters, closed drainage systems, and catheter care kits is impaired. Particularly in smaller institutions and less creative settings, this kind of inconsistency reduces the perception of product value, slows the process of mass adoption, and restricts the CAUTI prevention urology products market growth.
Preventive Drainage Devices (Primary Prevention) Segment is Expected to Drive the CAUTI Prevention Urology Products Market
The preventive drainage devices (primary prevention) category held the largest share in the CAUTI prevention urology products market in 2025 because preventing infections during the early stages of catheter use is becoming more and more important. To prevent bacterial invasion and lower the incidence of catheter-associated UTIs, healthcare providers are implementing sophisticated drainage systems, such as closed urine drainage systems, anti-reflux valves, and catheter securement devices. In order to reduce contamination and promote improved infection control procedures in hospitals and long-term care facilities, these devices are made to maintain a sterile passage between the catheter and the collection bag. Additionally, the adoption of preventive drainage devices as a first line of defense against CAUTIs is being encouraged by the increased emphasis on hospital infection prevention procedures and patient safety programs.
Urinary Incontinence Management Segment is Growing at the Highest Rate in the CAUTI Prevention Urology Products Market
In 2025, the urinary incontinence management category dominated the CAUTI prevention urology products market because urinary problems are very common in older persons. According to clinical data, millions of adults worldwide suffer from urine incontinence, especially those who are elderly or have neurological conditions. Additionally, in order to maintain hygiene and avoid difficulties, medical professionals rely on specific catheterization and drainage treatments. As the world's population ages and the prevalence of chronic urological disorders rises, segment growth is expected to accelerate. Furthermore, increased urological management device utilization is supported by better diagnostic awareness and prompt treatment. As patients want better comfort and infection control measures in both clinical and home care settings, demand is probably going to increase.
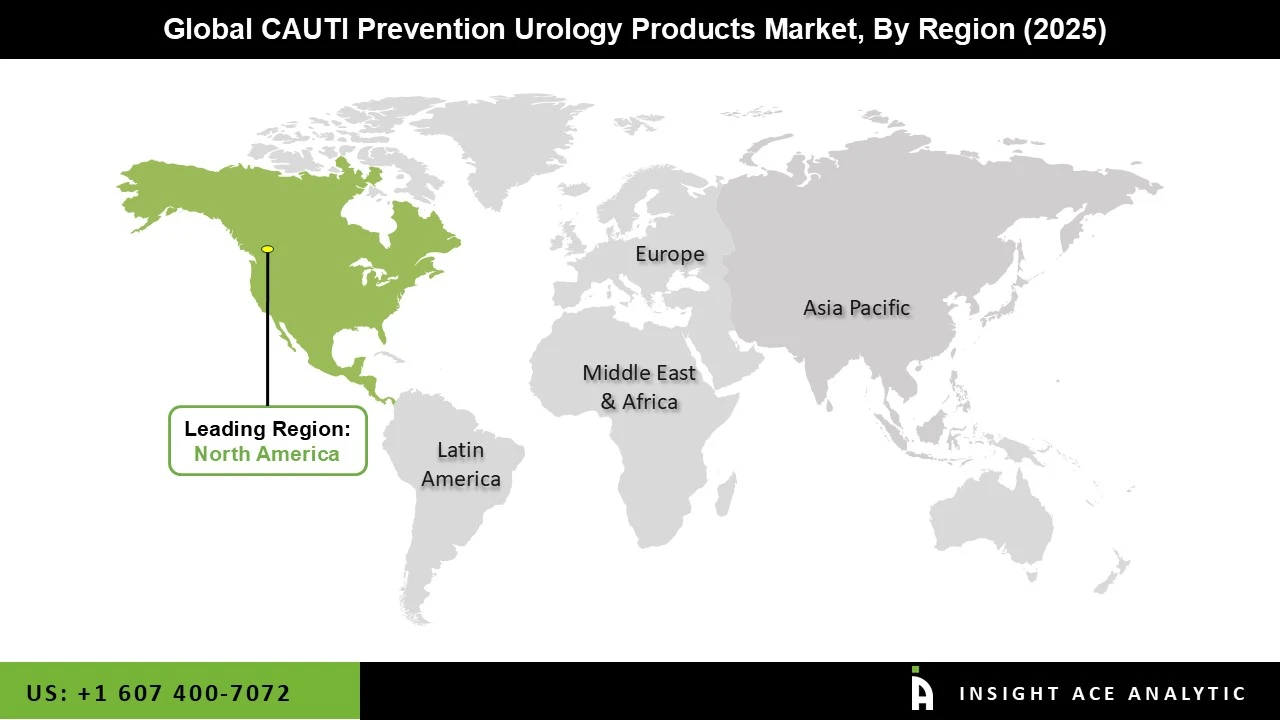
Why North America Led the CAUTI Prevention Urology Products Market?
The CAUTI prevention urology products market was dominated by North America region in 2025 as hospitals enhanced surveillance procedures throughout inpatient care units and stepped up infection control initiatives. According to the Centers for Disease Control and Prevention, over 75% of hospital UTIs are related to the use of urinary catheters, making catheter-associated UTIs one of the most prevalent illnesses linked to healthcare in the United States. The market trends are being shaped by the growing use of sterile drainage systems, antimicrobial catheters, and novel mechanical preventive devices. Additionally, the region's growing senior population and rising need for home healthcare solutions are driving the CAUTI prevention urology products market expansion. Moreover, the need for preventive devices intended to lessen infection consequences was bolstered by an increase in catheter usage due to an increase in surgical operations and intensive care hospitalizations.
Key Development
November 2025: The PureWickTM Portable Collection System, the first battery-operated personal urine management system for wheelchair users and those with limited mobility, was introduced by BD (Becton, Dickinson and Company). BD claims that users will be mobile at home and on the go, and they will be able to control urine incontinence. It is estimated that 25 million Americans suffer from urinary incontinence, which is linked to decreased self-esteem and social engagement. Its Portable Collection System is designed to provide users with more autonomy. This is the newest product in the growing PureWickTM Urology portfolio.
CAUTI Prevention Urology Products Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 3.52 Bn |
| Revenue forecast in 2035 | USD 7.16 Bn |
| Growth Rate CAGR | CAGR of 7.5% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Application, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | BD, Medtronic, Coloplast, Teleflex Incorporated, B. Braun SE, Cook Medical, Hollister Incorporated, Cardinal Health, Merit Medical, Medline Industries, LP., Poiesis Medical LLC, ConvaTec Group PLC, and Boston Scientific Corporation |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
CAUTI Prevention Urology Products Market- by Product Type
- Preventive Drainage Devices (Primary Prevention)
-
- External (Non-Invasive) Collection Systems
- Intermittent (Single-Use) Catheters
-
-
- Compact/Travel Intermittent
- Antibacterial/Antimicrobial-Coated
-
-
- Indwelling (Foley) Catheters
-
-
- Silver-Alloy Coated Foley
- Drug-Eluting Catheters
- Silicone-Only Foley
-
- Procedural Support & "Insertion Bundles" (Aseptic Prevention)
-
- Pre-Packaged Aseptic Trays
- Sterile Lubricant & Cleansing Solutions
- Bladder Scanners (Portable Ultrasound)
- Sterile Circuit & Maintenance Systems (Mechanical Prevention)
-
- Closed Drainage Bags
-
-
- Urine Meters
- Anti-Reflux Systems
-
-
- Needleless Sampling Ports
- Catheter Stabilization/Securement Devices
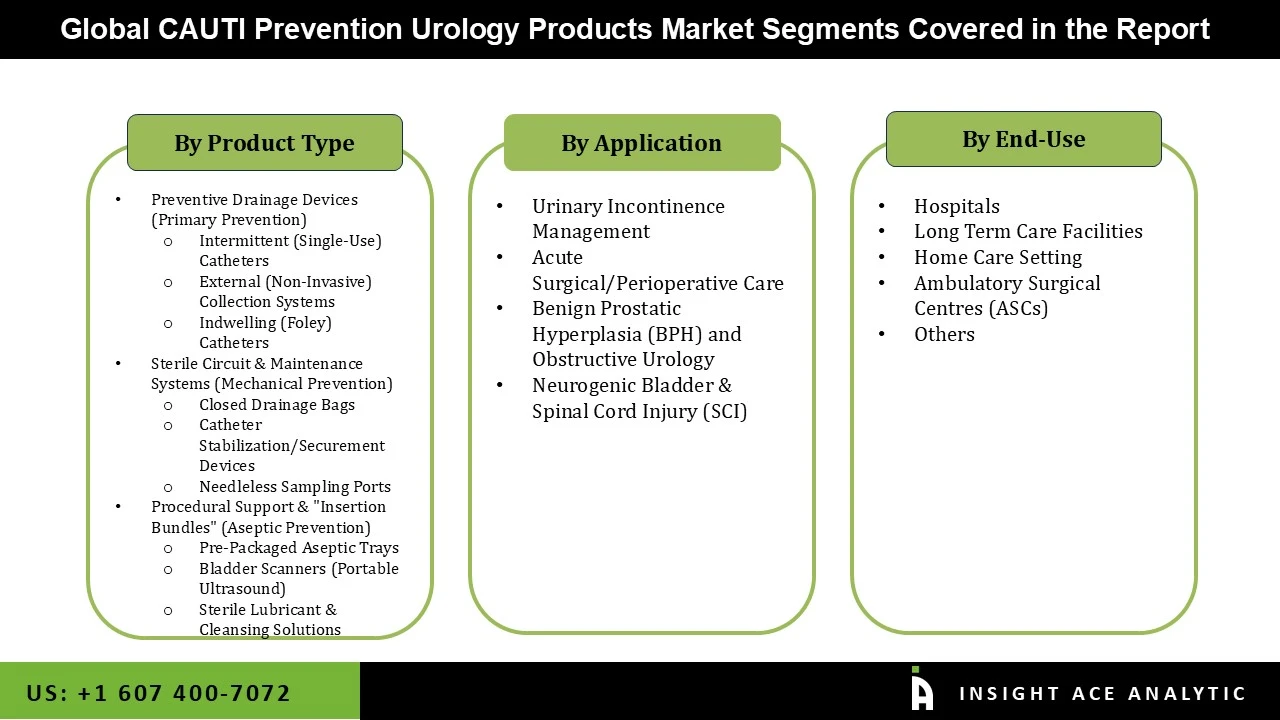
CAUTI Prevention Urology Products Market- by Application
• Neurogenic Bladder & Spinal Cord Injury (SCI)
• Urinary Incontinence Management
• Benign Prostatic Hyperplasia (BPH) and Obstructive Urology
• Acute Surgical/Perioperative Care
CAUTI Prevention Urology Products Market- by End-user
• Hospitals
• Home Care Setting
• Long Term Care Facilities
• Ambulatory Surgical Centers (ASCs)
• Others
CAUTI Prevention Urology Products Market-By Region
North America-
• The US
• Canada
Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
CAUTI Prevention Urology Products Market Size is valued at USD 3.52 Bn in 2025 and is predicted to reach USD 7.16 Bn by the year 2035
CAUTI Prevention Urology Products Market is predicted to grow at a 7.5% CAGR during the forecast period for 2026 to 2035.
BD, Medtronic, Coloplast, Teleflex Incorporated, B. Braun SE, Cook Medical, Hollister Incorporated, Cardinal Health, Merit Medical, Medline Industries, LP., Poiesis Medical LLC, ConvaTec Group PLC, and Boston Scientific Corporation
CAUTI Prevention Urology Products Market is segmented into Product Type, Application, End-user, and By Region
North America region is leading the CAUTI Prevention Urology Products Market.