Biodegradable Devices for IBD Market Size, Share and Trends Analysis 2026 to 2035
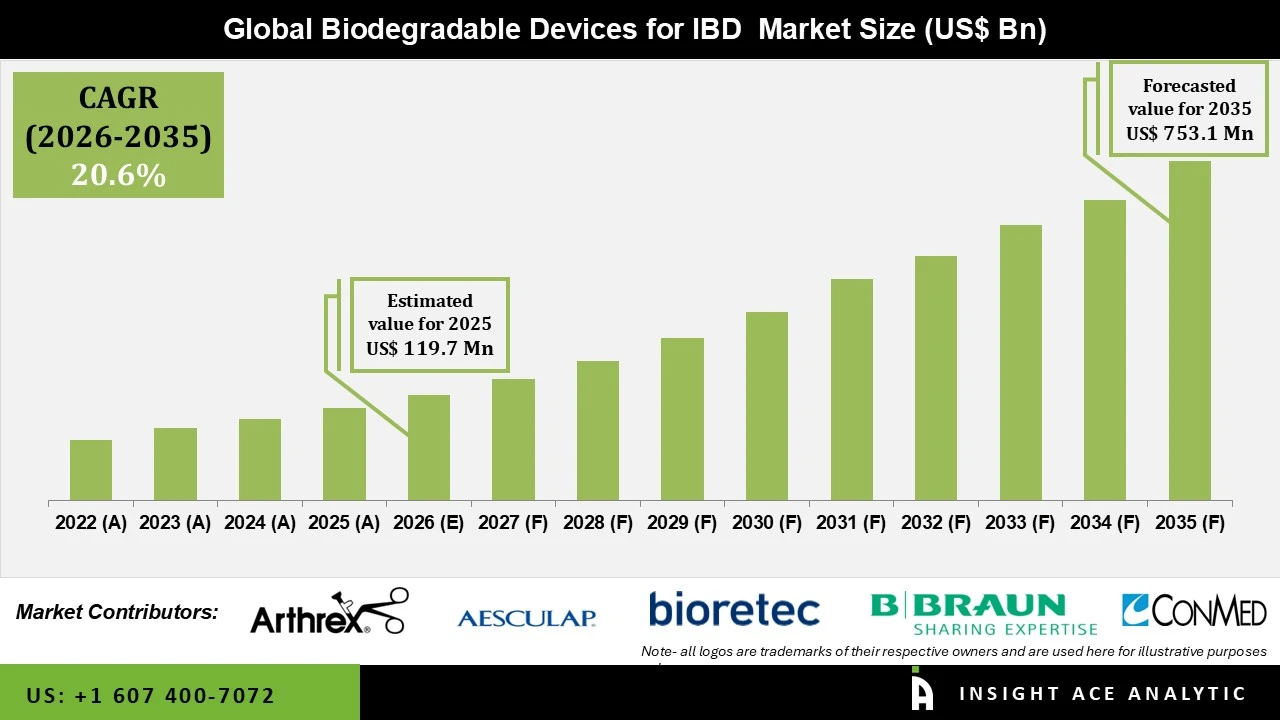
Biodegradable Devices for IBD Market Size is valued at USD 119.7 Mn in 2025 and is predicted to reach USD 753.1 Mn by the year 2034 at a 20.6% CAGR during the forecast period for 2026 to 2035.
Biodegradable Devices for IBD Market Size, Share & Trends Analysis Distribution by Disease Type (Crohn’s Disease and Ulcerative Colitis), By Product Type (Tissue Barriers, Wound Dressings, Drug Delivery Systems, Biodegradable Stents, Sutures / Staples, Implants & Patches, Anti-Adhesive, Scaffolds, Surgical Meshes, and Others), By Material Type (Polylactic Acid (PLA), Polyhydroxyalkanoates (PHA), Polyglycolic Acid (PGA), Polycaprolactone (PCL), and Others), By Technology (3D Printing, Biohybrid / Robotics, Nanotechnology, Capsule Endoscopy for IBD Diagnosis and Therapy), By Application (Inflammatory Bowel Disease (IBD) Treatment & Management, Drug Delivery for IBD, and Others), By End-user (Hospitals & Clinics, Ambulatory Surgical Centers, Research and Academic Institutes), and Segment Forecasts, 2026 to 2034
Inflammatory bowel disease (IBD) biodegradable medical implants or therapeutic delivery systems are designed in such a way as to safely biodegrade in the body after fulfilling their intended use, thereby making it unnecessary to undergo surgery for removal. Biodegradable polymers or bioresorbable materials are often utilized for making such devices, which then disintegrate into non-toxic components. These devices find their applications in the management of IBD and tissue healing scaffolding for various conditions such as Crohn’s disease and ulcerative colitis, stenting, and drug delivery systems. The rising number of cases related to inflammatory bowel diseases such as Crohn’s and ulcerative colitis, as well as the rapidly increasing need for modern and convenient treatment options, is responsible for the growth of the global biodegradable devices for IBD market.
The use of biodegradable devices in the treatment of IBD has been advanced even further by the increasing need for targeted approaches to the administration of medications and minimally invasive therapies, which are driving the market growth. Furthermore, the market for biodegradable devices in IBD has experienced steady growth in recent years because of advancements in drug delivery and biodegradable material development. Moreover, rising investments in gastrointestinal research and advancements in biodegradable polymers make it possible to create devices that can deliver anti-inflammatory drugs directly to the affected areas of the intestine.
In addition, the increasing global incidence of chronic gastrointestinal disorders is further driving the biodegradable devices for IBD market growth. On the other hand, technological advancements remain one of the major factors contributing to the development of biodegradable devices for IBD. The performance, safety, and degradation properties of biodegradable devices for IBD have been improved due to advances in areas such as tissue engineering and nanotechnology. Moreover, their clinical applications have been enhanced due to their combined use with intelligent drug delivery systems, which respond to specific physiological conditions present in the gastrointestinal tract.
Competitive Landscape
Which are the Leading Players in the Biodegradable Devices for IBD Market?
- Arthrex
- Braun
- Conmed
- Exactech
- Aesculap Implant Systems
- Bioretec
- Integra Life Sciences
- Osteomed
- Stryker
- Medtronic
- Smith and Nephew
- NuVasive
- Orthofix Medical
- Wright Medical
Market Dynamics
Driver
Rapid Advancements in Biomaterials
The global biodegradable devices for IBD market is expected to grow rapidly due to advancements in biomaterials. The discovery of biodegradable materials has created new opportunities for Inflammatory bowel disease (IBD) medical implants, and these are essential to the creation of medical devices. These biodegradable biomaterials are designed to break down organically over time in the body without any negative side effects.
The businesses are producing biodegradable implants with cutting-edge technologies. Additionally, the companies are receiving authorization for treatments and implants that employ a range of biodegradable materials. Furthermore, advancements in polycaprolactone (PCL), polylactic acid (PLA), and polyglycolic acid (PGA) have improved implant efficacy and safety. Because of their superior biocompatibility, these materials are less likely to cause inflammation or rejection.
Restrain/Challenge
Uncertainties Surrounding Material Performance and Degradation Management
The biodegradable devices for the IBD market are significantly hampered by the uncertainties surrounding material performance and degradation management in the gastrointestinal tract. The disintegration rate and structural integrity of biodegradable devices can be greatly impacted by the highly diverse conditions found in the human gut, which include varying pH levels, digestive enzymes, moisture, and mechanical stress from peristalsis.
On the other hand, delayed or uneven degradation might result in partial drug release, obstruction, or local discomfort. If a device degrades too quickly, it may stop working before providing the desired therapeutic benefit. The market for biodegradable devices for IBD may not expand as much overall due to its intricacy, which may restrict widespread acceptance and slow regulatory clearances.
Crohn's Disease Segment is Expected to Drive the Biodegradable Devices for IBD Market
The Crohn's disease category held the largest share in the Biodegradable Devices for IBD market in 2025 because Crohn's disease is becoming more commonplace worldwide, and there is a growing demand for cutting-edge, patient-friendly treatment alternatives.
The need for biodegradable stents, localized medication delivery systems, and temporary implants that don't need to be surgically removed has increased because Crohn's disease frequently results in chronic inflammation, strictures, and fistulas that necessitate repeated operations. By delivering targeted therapy directly to inflamed intestinal tissues, these devices improve long-term disease management and lessen procedure-related problems.
Polylactic Acid (PLA) Segment is Growing at the Highest Rate in the Biodegradable Devices for IBD Market
In 2025, the Polylactic Acid (PLA) category dominated the Biodegradable Devices for IBD market because of its good biocompatibility, consistent biodegradation profile, and widespread use in medicinal applications. Since PLA-based materials safely break down into lactic acid, which the body naturally metabolizes, they are becoming more and more popular in the production of biodegradable drug delivery systems, temporary intestinal stents, and implantable scaffolds used in the treatment of IBD. Additionally, the use of PLA-based devices is being further accelerated by the increasing prevalence of IBD worldwide as well as the growing need for minimally invasive and patient-friendly therapeutic options.
Why North America Led the Biodegradable Devices for IBD Market?
The Biodegradable Devices for IBD market was dominated by the North America region in 2025 because of the region's improved health care infrastructure, high rates of use of cutting-edge medical items, and the established presence of entry-level businesses. The region has seen significant expenditures in research and development, the adoption of favourable legislative frameworks, and an increase in patient and medical community awareness of the benefits of biodegradable solutions.
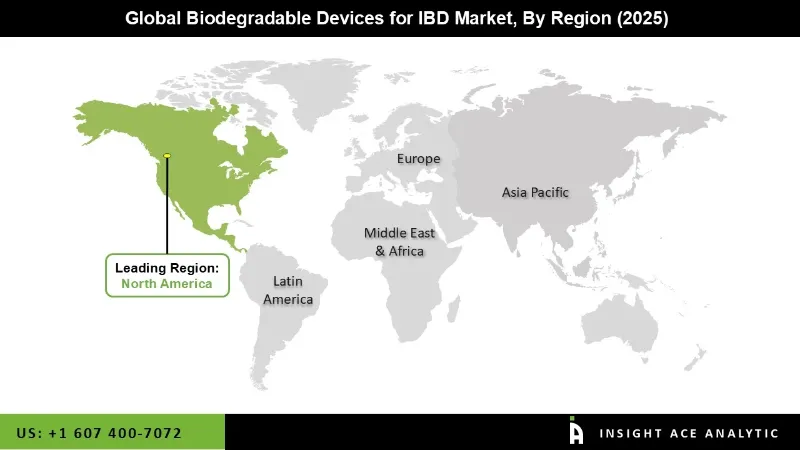
Furthermore, the biodegradable devices for the IBD market are growing because inflammatory bowel conditions like Crohn's and ulcerative colitis are more common in this area. Additionally, the presence of specialized manufacturing and strong collaboration between medical technology firms and research facilities contributes to the degree of product innovation, which is a compelling argument for North America's supremacy in the global market.
Biodegradable Devices for IBD Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 119.7 Mn |
| Revenue forecast in 2035 | USD 753.1 Mn |
| Growth Rate CAGR | CAGR of 20.6% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Disease Type, Product Type, Material Type, Technology, Application, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Arthrex, B. Braun, Conmed, Exactech, Aesculap Implant Systems, Bioretec, Integra Life Sciences, Osteomed, Stryker, Medtronic, Smith and Nephew, Nu Vasive, Orthofix Medical, and Wright Medical. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Biodegradable Devices for IBD Market by Disease Type-
- Crohn’s Disease
- Ulcerative Colitis

Biodegradable Devices for IBD Market by Product Type-
- Tissue Barriers
- Wound Dressings
- Drug Delivery Systems
- Biodegradable Stents
- Sutures / Staples
- Implants & Patches
- Anti-Adhesive
- Scaffolds
- Surgical Meshes
- Others
Biodegradable Devices for IBD Market by Material Type-
- Polylactic Acid (PLA)
- Polyhydroxyalkanoates (PHA)
- Polyglycolic Acid (PGA)
- Polycaprolactone (PCL)
- Others
Biodegradable Devices for IBD Market by Technology-
- 3D Printing
- Biohybrid / Robotics
- Nanotechnology
- Capsule Endoscopy for IBD Diagnosis and Therapy
Biodegradable Devices for IBD Market by Application-
- Inflammatory Bowel Disease (IBD) Treatment & Management
- Drug Delivery for IBD
- Others
Biodegradable Devices for IBD Market by End-user-
- Hospitals & Clinics
- Ambulatory Surgical Centers
- Research and Academic Institutes
By Region-
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Biodegradable Devices for IBD Market Size is valued at USD 119.7 Mn in 2025 and is predicted to reach USD 753.1 Mn by the year 2034.
Biodegradable Devices for IBD Market is expected to grow at a 20.6% CAGR during the forecast period for 2026 to 2035.
Arthrex, B. Braun, Conmed, Exactech, Aesculap Implant Systems, Bioretec, Integra Life Sciences, Osteomed, Stryker, Medtronic, Smith and Nephew, Nu Vasive, Orthofix Medical, and Wright Medical.
Disease Type, Product Type, Material Type, Technology, Application, End-user, and By Region are the key segments of the Biodegradable Devices for IBD Market.
North America region is leading the Biodegradable Devices for IBD Market.