In-Vivo CAR-T Viral and Non-Viral Vector Manufacturing Market Size, Share Detailed Report 2026 to 2035
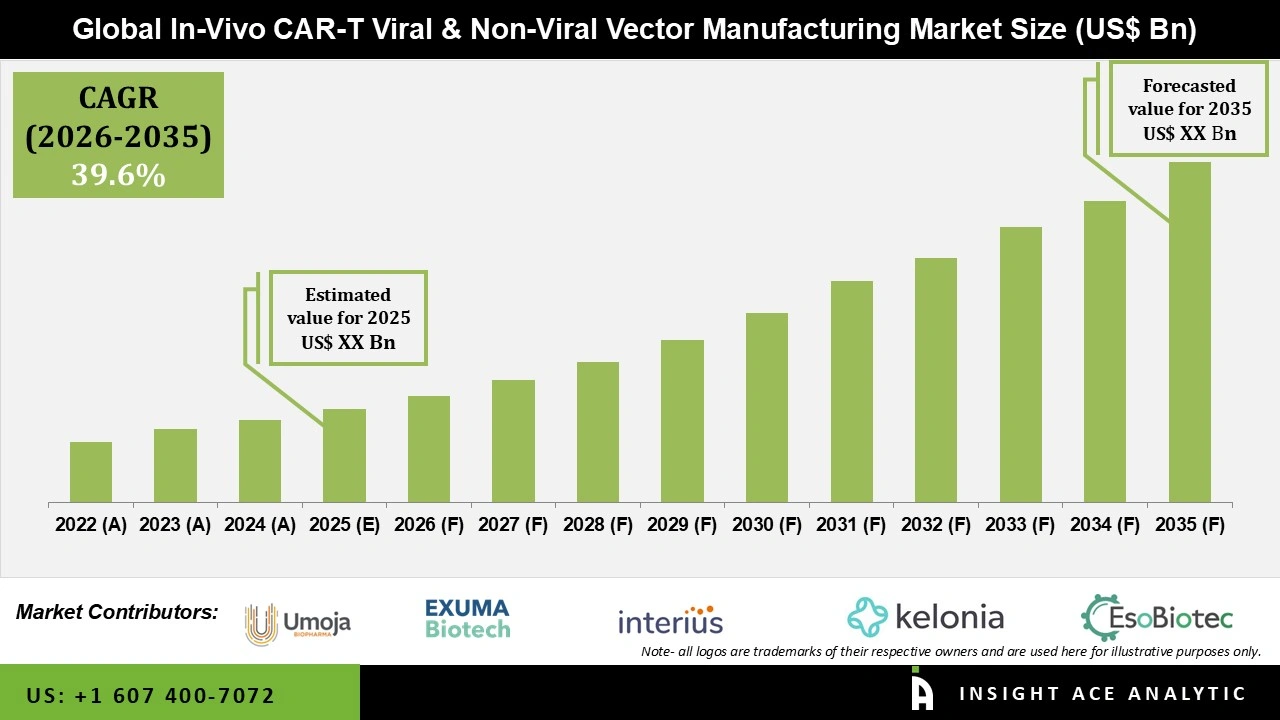
Global In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market Size is predicted to grow at a 39.6% CAGR during the forecast period for 2026 to 2035.
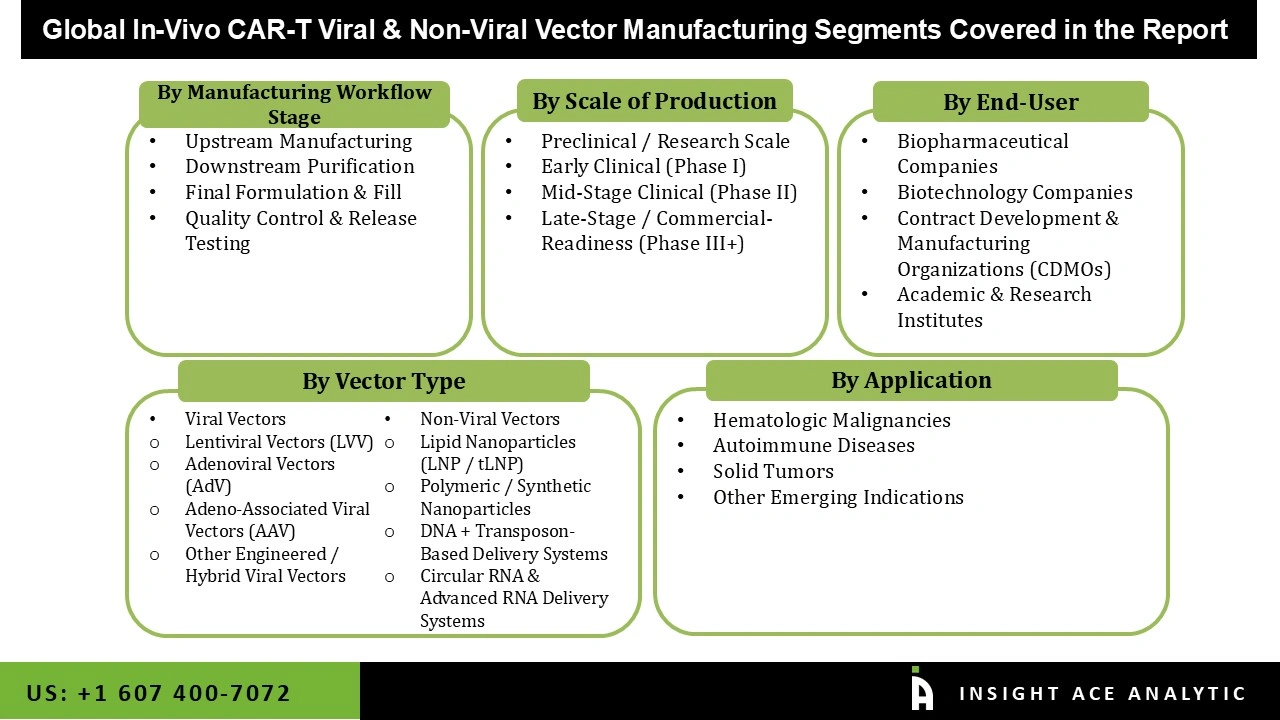
In-Vivo CAR-T Viral and Non-Viral Vector Manufacturing Market Size, Share & Trends Analysis Distribution by Vector Type (Viral Vectors (Lentiviral Vectors (LVV), Adenoviral Vectors (AdV), Adeno-Associated Viral Vectors (AAV), Other Engineered / Hybrid Viral Vectors), Non-Viral Vectors (Lipid Nanoparticles (LNP / tLNP), Polymeric / Synthetic Nanoparticles, DNA + Transposon-Based Delivery Systems, Circular RNA & Advanced RNA Delivery Systems)) By Manufacturing Workflow Stage (Upstream Manufacturing, Downstream Purification, Final Formulation & Fill, Quality Control & Release Testing), By Scale of Production (Preclinical / Research Scale, Early Clinical (Phase I), Mid-Stage Clinical (Phase II), Late-Stage / Commercial-Readiness (Phase III+)) By End-User, By Region and Segment Forecasts, 2026 to 2036
The In-Vivo CAR-T viral & non-viral vector manufacturing market refers to the production and distribution of viral and non-viral vectors, which are employed in the delivery of CAR-T cell therapies directly into the body (in-vivo). In contrast to conventional ex-vivo CAR-T cell therapies, which require the engineering of immune cells outside the body, in-vivo approaches employ vectors to genetically engineer T cells in the body, making manufacturing easier and possibly more accessible. The market is divided into two primary vector categories: viral vectors, including lentiviruses and adeno-associated viruses (AAVs), which are preferred for their high transduction efficiency and stable expression, and non-viral vectors, including lipid nanoparticles (LNPs), synthetic polymers, and transposases, which are preferred for their safety, scalability, and reduced immunogenicity.
The vectors generated are used in cancer immunotherapy, treatment of autoimmune diseases, management of infectious diseases, and gene editing and regenerative medicine as carriers for the CRISPR/Cas system. Market expansion is fuelled by the growing need for scalable CAR-T cell therapies and by improvements in vector engineering that enhance delivery efficiency and minimise off-target toxicity. Nevertheless, the complexity and cost of manufacturing, as well as safety and regulatory issues such as insertional mutagenesis and immunogenicity, remain major limitations to widespread use.
Competitive Landscape
Which are the Leading Players in the In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market?
Viral Vector Manufacturers / Developers
- Umoja Biopharma
- EXUMA Biotech
- Interius BioTherapeutics
- Kelonia Therapeutics
- EsoBiotec
- Vyriad
- Vector BioMed
- Vector BioPharma
Non-Viral Vector Manufacturers / Developers
- Capstan Therapeutics
- Ensoma
- Myeloid Therapeutics
- NanoCell Therapeutics
- Orbital Therapeutics
- GenEdit
- Verve Therapeutics
- Beam Therapeutics
- Tessera Therapeutics
CDMOs / Contract Vector Manufacturing Providers
- Lonza
- Thermo Fisher Scientific
- Catalent
- Oxford BioMedica
- AGC Biologics
- FUJIFILM Diosynth Biotechnologies
- Samsung Biologics
- Cobra Biologics
Market Dynamics
Driver
Rising Demand for Scalable In Vivo CAR T Therapies
The market is primarily driven by the growing demand for scalable and accessible CAR T therapies. In vivo CAR T approaches simplify treatment by delivering CAR constructs directly into patients’ T cells, reducing the complexity, cost, and time associated with traditional ex vivo methods. Advances in vector engineering and the rising prevalence of cancer and chronic diseases further support market growth.
Restrain/Challenge
Manufacturing Complexity and Regulatory Hurdles
The primary challenge facing the In‑Vivo CAR‑T viral & non‑viral vector manufacturing market is the complexity and high cost of producing high-quality vectors at scale. The production of viral vectors such as lentiviruses and AAVs is a complex process that requires sophisticated bioprocessing infrastructure and expertise. Even non-viral vectors, which are safer and more easily producible, need sophisticated formulation and delivery systems for effective in-vivo targeting.
Viral Vector Segment is Expected to Drive the In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market
The viral vector segment is expected to drive the In‑Vivo CAR‑T viral & non‑viral vector manufacturing market due to its proven efficiency, clinical adoption, and ability to deliver durable therapeutic outcomes. Lentiviruses and AAVs have the ability to transduce T cells from patients efficiently in vivo, ensuring stable CAR expression and potent anti-tumor immune responses. Their success in both hematologic malignancies and solid tumors has made them the vectors of choice in clinical trials and commercial CAR-T therapies. Moreover, continuous advancements in vector design, scalability, and safety are improving their reliability and ease of use. Although non-viral vectors are being explored as a safer alternative, the viral vectors are likely to remain the major revenue-generating component, leading to the growth of the market because of their extensive clinical use and continued development of viral vector manufacturing infrastructure.
CBD (Cannabidiol) Segment is Growing at the Highest Rate in the In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market
The upstream manufacturing market is expanding at the fastest rate in the in-vivo CAR-T vector market. This phase, which involves cell culture, vector generation, and gene transfer, is a crucial step in scaling up the production of vectors to cater to the increasing demand for therapies. Improved bioreactors, cell lines, and production methods are making upstream manufacturing the driving force behind the market.
Why North America Led the In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market?
North America currently leads the in vivo CAR-T viral and non-viral vector manufacturing market due to the presence of a strong biopharma industry, well-developed infrastructure, and a rich R&D environment. The region is home to many CAR-T therapy developers, biotech firms, contract development organizations, and also has the maximum number of clinical trials being conducted.
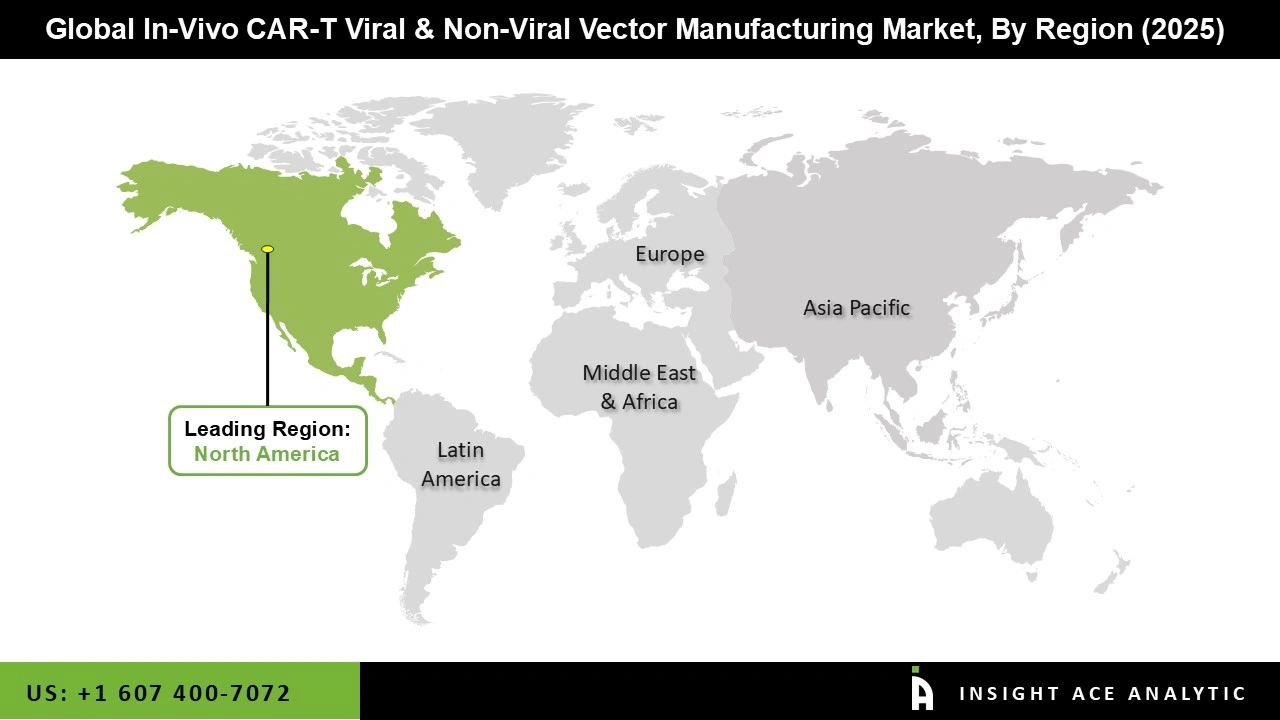
Favorable government initiatives and investments have further fueled the development and commercialization of CAR-T therapies, making North America the leading contributor to the market.
Key Development
August 2025 : ViroCell Biologics announced a manufacturing collaboration with AvenCell Therapeutics and successfully delivered a novel retroviral vector. The vector was used to produce AVC-203, an investigational CD19/CD20 dual-targeted CAR T therapy for B cell malignancies and autoimmune diseases.
In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market Report Scope:
| Report Attribute | Specifications |
| Growth Rate CAGR | CAGR of 39.6% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Vector Type, Manufacturing Workflow Stage, Scale of Production, End-User, Application and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Umoja Biopharma, EXUMA Biotech, Interius BioTherapeutics, Kelonia Therapeutics, EsoBiotec, Vyriad, Vector BioMed, Vector BioPharma, Capstan Therapeutics, Ensoma, Myeloid Therapeutics, NanoCell Therapeutics, Orbital Therapeutics, GenEdit, Verve Therapeutics, Beam Therapeutics, Tessera Therapeutics, Lonza, Thermo Fisher Scientific, Catalent, Oxford BioMedica, AGC Biologics, FUJIFILM Diosynth Biotechnologies, Samsung Biologics, Cobra Biologics |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market :
In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market by, Vector Type -
- Viral Vectors
- Lentiviral Vectors (LVV)
- Adenoviral Vectors (AdV)
- Adeno-Associated Viral Vectors (AAV)
- Other Engineered / Hybrid Viral Vectors
- Non-Viral Vectors
- Lipid Nanoparticles (LNP / tLNP)
- Polymeric / Synthetic Nanoparticles
- DNA + Transposon-Based Delivery Systems
- Circular RNA & Advanced RNA Delivery Systems
In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market, by Manufacturing Workflow Stage -
- Upstream Manufacturing
- Downstream Purification
- Final Formulation & Fill
- Quality Control & Release Testing
In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market, by Scale of Production -
- Preclinical / Research Scale
- Early Clinical (Phase I)
- Mid-Stage Clinical (Phase II)
- Late-Stage / Commercial-Readiness (Phase III+)
In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market, by End-User -
- Biopharmaceutical Companies
- Biotechnology Companies
- Contract Development & Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market, by Application -
- Hematologic Malignancies
- Autoimmune Diseases
- Solid Tumors
- Other Emerging Indications
In-Vivo CAR-T Viral & Non-Viral Vector Manufacturing Market, By Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
In-Vivo CAR-T Viral and Non-Viral Vector Manufacturing Market is expected to grow at a 39.6% CAGR during the forecast period for 2026 to 2035
Umoja Biopharma, EXUMA Biotech, Interius BioTherapeutics, Kelonia Therapeutics, EsoBiotec, Vyriad, Vector BioMed, Vector BioPharma, Capstan Therapeutics, Ensoma, Myeloid Therapeutics, NanoCell Therapeutics, Orbital Therapeutics, GenEdit, Verve Therapeutics, Beam Therapeutics, Tessera Therapeutics, Lonza, Thermo Fisher Scientific, Catalent, Oxford BioMedica, AGC Biologics, FUJIFILM Diosynth Biotechnologies, Samsung Biologics, Cobra Biologics and Others,
In-Vivo CAR-T Viral and Non-Viral Vector Manufacturing Market is segmented in Vector Type (Viral Vectors (Lentiviral Vectors (LVV), Adenoviral Vectors (AdV), Adeno-Associated Viral Vectors (AAV), Other Engineered / Hybrid Viral Vectors), Non-Viral Vectors (Lipid Nanoparticles (LNP / tLNP), Polymeric / Synthetic Nanoparticles, DNA + Transposon-Based Delivery Systems, Circular RNA & Advanced RNA Delivery Systems)) By Manufacturing Workflow Stage (Upstream Manufacturing, Downstream Purification, Final Formulation & Fill, Quality Control & Release Testing), By Scale of Production (Preclinical / Research Scale, Early Clinical (Phase I), Mid-Stage Clinical (Phase II), Late-Stage / Commercial-Readiness (Phase III+)) By End-User and Other.
North America region is leading the In-Vivo CAR-T Viral and Non-Viral Vector Manufacturing Market.