Sustainable and Bio-Derived Injectable-Grade Excipients Market Forecast with Size and Share Analysis 2026 to 2035
What is Sustainable and Bio-Derived Injectable-Grade Excipients Market Size?
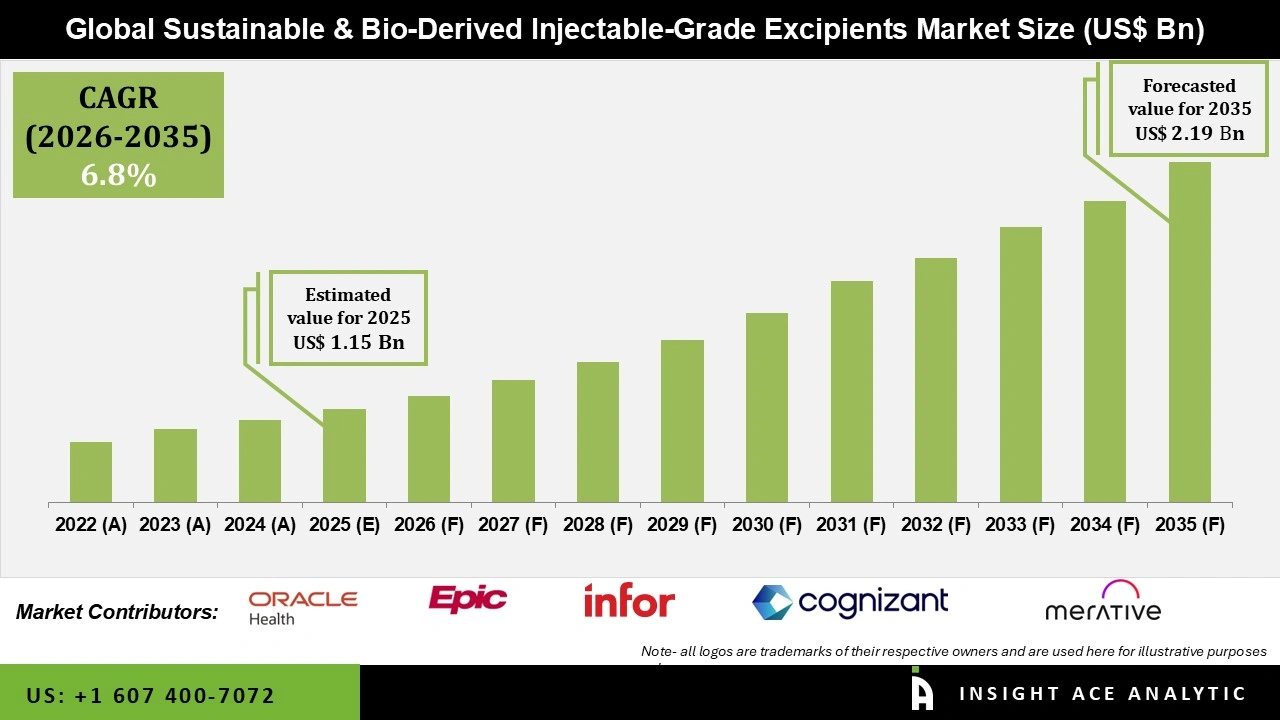
Global Sustainable and Bio-Derived Injectable-Grade Excipients Market Size is valued at USD 1.15 Bn in 2025 and is predicted to reach USD 2.19 Bn by the year 2035 at a 6.8% CAGR during the forecast period for 2026 to 2035.
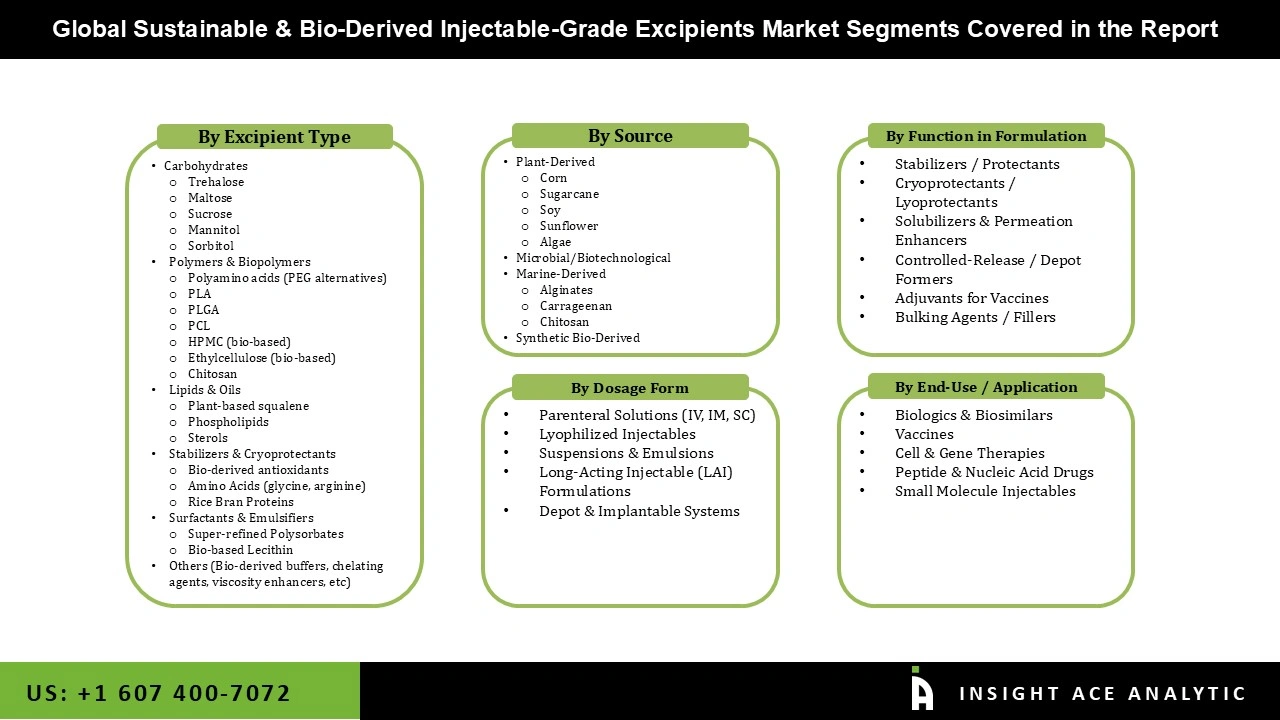
Sustainable and Bio-Derived Injectable-Grade Excipients Market, Share & Trends Analysis Report, By Excipient Type (Carbohydrates, Polymers & Biopolymers, Lipids & Oils, Stabilizers & Cryoprotectants, Surfactants & Emulsifiers, Others), By Source (Plant-Derived, Microbial/Biotechnological, Marine-Derived, Synthetic Bio-Derived),By Function in Formulation (Stabilizers / Protectants, Cryoprotectants / Lyoprotectants, Solubilizers & Permeation Enhancers, Controlled-Release / Depot Formers, Adjuvants for Vaccines, Bulking Agents / Fillers), By Dosage Form, By End-Use, By Region, and Segment Forecasts, 2026 to 2035
Sustainable and Bio-Derived Injectable-Grade Excipients Market Key Takeaways:
|
Sustainable, bio-derived injectable excipients are inert pharmaceutical ingredients that enhance drug stability, solubility, and bioavailability while reducing environmental impact. Derived from renewable sources such as plant-based sucrose, starch, and cellulose, these excipients are manufactured through eco-friendly processes that eliminate harmful organic solvents, reduce waste generation, and lower carbon emissions supporting global sustainability objectives. This approach contrasts sharply with traditional petroleum-based or synthetic alternatives. A notable example is a sucrose-derived excipient offering high hydrophobicity and biocompatibility, enabling improved solubility, prolonged drug release, depot formation, and abuse-deterrent functionality in injectable formulations.
The pharmaceutical industry is increasingly adopting sustainable excipients. In 2023, Croda International formed strategic partnerships to source key vaccine adjuvants sustainably rather than developing new synthetic alternatives. Croda partnered with Amyris to supply biotechnology-derived squalene and with Botanical Solutions Inc. (BSI) to provide pharmaceutical-grade QS-21, both essential for vaccine formulations. Beyond vaccines, biodegradable biopolymers including polylactic acid (PLA), polyglycolic acid (PGA), and poly(lactic-co-glycolic acid) (PLGA) enable depot formulations for controlled, sustained drug release, benefiting long-acting therapies for conditions like psychiatric disorders, hormonal imbalances, and chronic pain. Additionally, biopolymers such as hyaluronic acid and chitosan facilitate targeted delivery by encapsulating active ingredients in nanoparticles or micelles, improving bioavailability while reducing side effects in injectable biologics including monoclonal antibodies and gene therapies.
Competitive Landscape
Which are the Leading Players in Sustainable and Bio-Derived Injectable-Grade Excipients Market?
- Nagase Viita
- Eastman Chemical Company
- Roquette Frères
- Croda International / Croda Pharma
- Evonik Industries
- Merck KGaA
- BASF SE
- ADM (Archer Daniels Midland)
- Pfanstiehl
- Ashland
- Clariant
- Actylis Lab Solutions
- CD Bioparticles
- Other Prominent Players
Market Dynamics
Driver
Regulatory Support and Sustainability Mandates
Increasingly stringent environmental regulations and sustainability mandates from regulatory bodies worldwide are driving adoption of bio-derived injectable excipients. Government initiatives and pharmaceutical industry commitments to reduce carbon footprints and minimize environmental impact incentivize manufacturers to transition from petroleum-based to renewable, plant-derived alternatives. Regulatory frameworks now favor eco-friendly manufacturing processes that eliminate harmful solvents and reduce waste generation. Additionally, sustainability-focused labeling requirements and corporate social responsibility expectations encourage pharmaceutical companies to source sustainable excipients, enhancing brand reputation and market competitiveness. These regulatory pressures, combined with consumer demand for environmentally responsible products, create strong market incentives for bio-derived excipient adoption, accelerating market expansion and positioning sustainable excipients as essential components of modern pharmaceutical manufacturing.
Restrain/Challenge
Higher Production Costs and Supply Chain Limitations
Bio-derived injectable excipients face significant cost disadvantages compared to traditional petroleum-based alternatives, constraining market growth. Sustainable sourcing, eco-friendly manufacturing processes, and stringent quality control requirements increase production expenses, resulting in premium pricing that deters adoption among cost-sensitive manufacturers. Additionally, limited supply chain infrastructure for renewable materials creates sourcing inconsistencies and availability challenges, particularly for specialized bio-based ingredients like plant-derived sucrose and botanical extracts. Scale-up complexities and lower manufacturing volumes compared to conventional excipients further elevate per-unit costs. Small and mid-sized pharmaceutical companies may lack resources to absorb these higher expenses, limiting market penetration. Until production technologies mature and supply chains expand, cost barriers will remain a significant restraint on broader bio-derived excipient adoption.
The Carbohydrates Segment is Expected to Have the Highest Growth Rate During the Forecast Period
The carbohydrates segment is positioned for the highest growth rate due to its versatility and established safety profile in pharmaceutical applications. Bio-derived carbohydrates such as sucrose, starch, and cellulose serve multiple functions enhancing drug solubility, stability, and bioavailability while maintaining biocompatibility. Their plant-based origin aligns with global sustainability mandates, making them increasingly attractive to manufacturers committed to reducing environmental impact. Carbohydrates offer cost-effectiveness compared to synthetic alternatives, improving market accessibility for diverse pharmaceutical companies. Additionally, regulatory agencies have extensive experience approving carbohydrate-based excipients, streamlining development timelines and reducing commercialization barriers. Growing demand for long-acting injectable therapies and sustained-release formulations further drives carbohydrate adoption. The combination of regulatory familiarity, sustainability alignment, versatile functionality, and relative affordability positions carbohydrates as the fastest-growing excipient category.
The Plant-Derived Segment Dominates the Market
Plant-derived excipients serve multiple critical functions in injectable pharmaceutical formulations, including stabilization, cryoprotection, solubility enhancement, and depot formation. Sucrose, for example, is a key stabilizer in lipid nanoparticle-based mRNA vaccines such as Comirnaty and Spikevax, while cellulose derivatives enable sustained-release delivery in injectable therapies. The widespread availability and established production methods for plant-based materials support cost-effective, large-scale manufacturing, making them economically attractive particularly for generic and biosimilar developers operating under tight margin constraints.
Global regulatory and governmental support for pharmaceutical sustainability further accelerates plant-derived excipient adoption. Regulatory bodies worldwide actively encourage environmentally responsible manufacturing practices and favor plant-based ingredients over synthetic or animal-derived alternatives due to their lower environmental footprint and reduced carbon emissions. Beyond regulatory incentives, plant-derived excipients align with circular economy principles by utilizing agricultural byproducts and minimizing waste throughout the production process. This dual advantage—combining economic efficiency with environmental responsibility—positions plant-derived excipients as the preferred choice for modern pharmaceutical development and manufacturing.
Why North America Led the Sustainable & Bio-Derived Injectable-Grade Excipients?
North America dominates the sustainable and bio-derived injectable excipients market due to strong regulatory support and advanced manufacturing infrastructure. The FDA and Health Canada actively promote sustainable pharmaceutical practices, encouraging manufacturers to adopt eco-friendly excipients and establishing clear approval pathways. Significant investments in biotechnology and pharmaceutical innovation, coupled with robust venture capital funding, accelerate development of bio-derived alternatives.
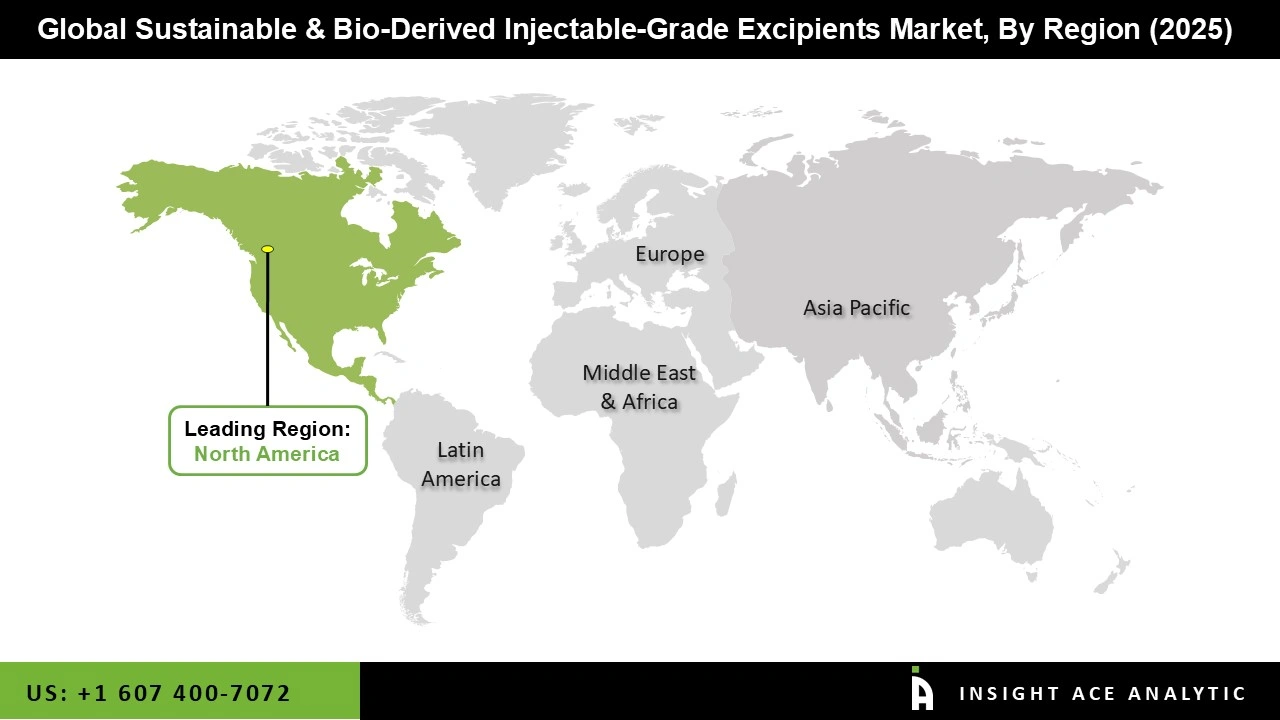
North American companies benefit from established agricultural supply chains providing reliable access to plant-derived raw materials like sucrose and cellulose at competitive costs. Additionally, major pharmaceutical manufacturers and CDMOs in the region prioritize sustainability commitments, driving demand for plant-based excipients. Strong intellectual property protection, skilled workforce, and proximity to key pharmaceutical markets further strengthen North America's competitive position as the global leader in sustainable injectable excipient innovation and production.
Recent Developments:
• In Nov 2024, Nagase Viita Co., Ltd., a member of the NAGASE Group, intends to introduce high-purity, low-endotoxin SUCROSE SG by June 2025 as part of its expansion of the SOLBIOTETM line of saccharide-based pharmaceutical excipients. In addition to TREHALOSE SG and MALTOSE PH, which are trademark products of pharmaceutical components, SOLBIOTETM's portfolio now includes SUCROSE SG, providing a wide spectrum of stability-related options for biopharmaceutical development.
• In Oct 2023, Clariant, has announced new additions to its range of high-performing pharmaceutical ingredient solutions. In order to position itself as a one-stop shop for industry solutions, Clariant will introduce three new VitiPure® excipients at CPHI Barcelona. These will enable a wide range of Active Pharmaceutical Ingredient (API) formulations and administration routes, including sensitive ones like mRNA vaccines and biologic medications.
Sustainable and Bio-Derived Injectable-Grade Excipients Market Report Scope :
| Report Attribute | Specifications |
| Market Size Value In 2025 | USD 1.15 Bn |
| Revenue Forecast In 2035 | USD 2.19 Bn |
| Growth Rate CAGR | CAGR of 6.8 % from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Mn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | By Excipient Type, By Source, By Function in Formulation, By Dosage Form, By End-Use Application |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; South East Asia |
| Competitive Landscape | Nagase Viita, Eastman Chemical Company, Roquette Frères, Croda International / Croda Pharma, Evonik Industries, Merck KGaA, BASF SE, ADM (Archer Daniels Midland), Pfanstiehl, Ashland, Clariant, Actylis Lab Solutions, CD Bioparticles |
| Customization Scope | Free customization report with the procurement of the report and modifications to the regional and segment scope. Particular Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of the Sustainable & Bio-Derived Injectable-Grade Excipients Market :
Sustainable & Bio-Derived Injectable-Grade Excipients Market, By Excipient Type-
- Carbohydrates
- Trehalose
- Maltose
- Sucrose
- Mannitol
- Sorbitol
- Polymers & Biopolymers
- Polyamino acids (PEG alternatives)
- PLA
- PLGA
- PCL
- HPMC (bio-based)
- Ethylcellulose (bio-based)
- Chitosan
- Lipids & Oils
- Plant-based squalene
- Phospholipids
- Sterols
- Stabilizers & Cryoprotectants
- Bio-derived antioxidants
- Amino Acids (glycine, arginine)
- Rice Bran Proteins
- Surfactants & Emulsifiers
- Super-refined Polysorbates
- Bio-based Lecithin
- Others (Bio-derived buffers, chelating agents, viscosity enhancers, etc)
Sustainable & Bio-Derived Injectable-Grade Excipients Market, By Source-
- Plant-Derived
- Corn
- Sugarcane
- Soy
- Sunflower
- Algae
- Microbial/Biotechnological
- Marine-Derived
- Alginates
- Carrageenan
- Chitosan
- Synthetic Bio-Derived
Sustainable & Bio-Derived Injectable-Grade Excipients Market, By Function in Formulation-
- Stabilizers / Protectants
- Cryoprotectants / Lyoprotectants
- Solubilizers & Permeation Enhancers
- Controlled-Release / Depot Formers
- Adjuvants for Vaccines
- Bulking Agents / Fillers
Sustainable & Bio-Derived Injectable-Grade Excipients Market, By Dosage Form-
- Parenteral Solutions (IV, IM, SC)
- Lyophilized Injectables
- Suspensions & Emulsions
- Long-Acting Injectable (LAI) Formulations
- Depot & Implantable Systems
Sustainable & Bio-Derived Injectable-Grade Excipients Market, By End-Use Application-
- Biologics & Biosimilars
- Vaccines
- Cell & Gene Therapies
- Peptide & Nucleic Acid Drugs
- Small Molecule Injectables
Sustainable & Bio-Derived Injectable-Grade Excipients Market, By Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Sustainable and Bio-Derived Injectable-Grade Excipients Market Size is valued at USD 1.15 Bn in 2025 and is predicted to reach USD 2.19 Bn by the year 2035
Sustainable and Bio-Derived Injectable-Grade Excipients Market is expected to grow at a 6.8% CAGR during the forecast period for 2026 to 2035
Nagase Viita, Eastman Chemical Company, Roquette Frères, Croda International / Croda Pharma, Evonik Industries, Merck KGaA, BASF SE, ADM (Archer Daniels Midland), Pfanstiehl, Ashland, Clariant, Actylis Lab Solutions, CD Bioparticles and Others.
Sustainable and Bio-Derived Injectable-Grade Excipients Market is segmented into Excipient Type, Source, Function in Formulation, Dosage Form, End-Use Application and Other.
North America region is leading the Sustainable and Bio-Derived Injectable-Grade Excipients Market.