Bioregenerative Aesthetic Injectable Market Size, Scope, Forecast Report 2026 to 2035
What is Bioregenerative Aesthetic Injectable Market Size?
Global Bioregenerative Aesthetic Injectable Market Size is valued at USD 502.48 Mn in 2025 and is predicted to reach USD 1,503.99 Mn by the year 2035 at a 11.9% CAGR during the forecast period for 2026 to 2035.
Bioregenerative Aesthetic Injectable Market Size, Share & Trends Analysis Distribution by Product Type (PN (PDRN and PN Injectables), Exosome-based Injectables, Peptide-based Injectables, Biostimulatory Injectables (CaHA, PLLA), PRP Injectables, and Others), Application (Skin Hydration & Moisturization (For Face), Acne Scar (For Face), Elasticity & Firmness (For Face), Fine Lines (For Face), Under eye-areas (For Face), Wrinkle Correction (For Face), and Body Application (Arms & Thighs, Knees, Décolletage, Scalp & Hair Rejuvenation, Intimate Areas)), End-user (MedSpas, Hospitals, Aesthetic Surgery Centers, Dermatology Centers, and Others), and Segment Forecasts, 2026 to 2035.
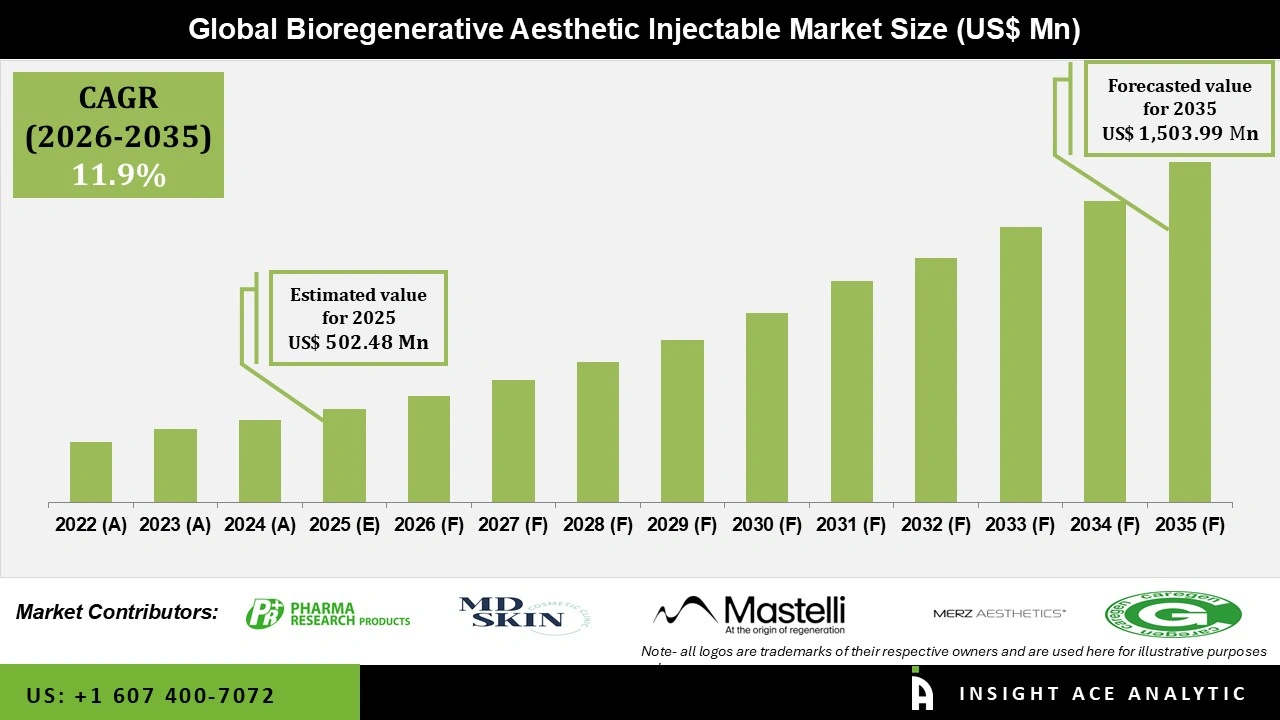
Bioregenerative Aesthetic Injectable Market Key Takeaways:
|
Bioregenerative aesthetic injectables are a class of cosmetic injectable therapies intended to improve skin quality over time by stimulating the body's natural regenerative mechanisms in addition to offering instant aesthetic benefit. These injectables function by stimulating collagen synthesis, tissue remodeling, and cellular regeneration within the skin, in contrast to conventional dermal fillers that just add volume. They usually include bioactive materials that promote fibroblast activity and extracellular matrix reconstruction, such as poly-L-lactic acid (PLLA), calcium hydroxylapatite (CaHA), polynucleotides, or components produced from platelets. The growing customer demand for minimally invasive, natural-looking treatments that support long-term skin health is driving the bioregenerative aesthetic injectable market's expansion.
The bioregenerative aesthetic injectable market is expanding due in large part to the growing desire for minimally invasive cosmetic operations. The use of regenerative injectable solutions has increased as a result of consumers' growing desire for cosmetic procedures that produce outcomes that look natural with little recovery time. Bioregenerative injectables are very appealing in contemporary aesthetic medicine since they promote tissue regeneration and collagen formation, in contrast to traditional fillers that merely temporarily increase volume. Additionally, a wider range of consumers are choosing face rejuvenation operations due to increased awareness of anti-aging treatments, social media impact, and beauty standards. The advanced injectable therapies are becoming more widely available in dermatology clinics and medical spas, which is further driving market growth.
Furthermore, the bioregenerative aesthetic injectable market is anticipated to be shaped by ongoing developments in biotechnology and aesthetic dermatology. Manufacturers are concentrating on creating cutting-edge formulas that boost collagen stimulation, enhance skin quality, and produce results that stay longer. New prospects for market expansion are being created by the growing incorporation of regenerative medicine concepts into cosmetic procedures, such as the utilization of polynucleotides, platelet-derived components, and bio-stimulatory materials. Additionally, access to these treatments is being improved globally by the growth of aesthetic clinics and the increasing number of qualified specialists. The bioregenerative aesthetic injectable market is expanding as a result of regulatory approvals for novel injectable medications and the growing demand for preventive anti-aging treatments.
Competitive Landscape
Which are the Leading Players in Bioregenerative Aesthetic Injectable Market?
- PharmaResearch Products Co., Ltd.
- MD Skin Solutions
- Caregen Co. Ltd
- BR Pharma
- Fillmed
- Croma Pharma
- PROMOITALIA
- Galderma
- Fidia
- IBSA
- Bio-Formula
- Allergan (AbbVie)
- VAIM
- Mastelli s.r.l.
- Merz Aesthetics
Market Dynamics
Driver
Growing Use of Minimally Invasive Procedures
The growing use of minimally invasive procedures such as injectables for facial and body rejuvenation is a notable and rapidly growing trend in the worldwide bioregenerative aesthetic injectable market. Due to consumer preferences for quick, efficient, and low-downtime procedures, the demand for botulinum toxin injections, dermal fillers, and microneedling treatments has increased in both developed and emerging countries. Additionally, in order to improve therapeutic results, combination therapies—such as laser resurfacing combined with platelet-rich plasma (PRP) or hyaluronic acid-based treatments—are also becoming more widely used. In order to provide individualized aesthetic solutions and encourage adoption, medical experts are increasingly utilizing technological developments in imaging, diagnostic tools, and patient-specific treatment planning. The bioregenerative aesthetic injectable market's growth trajectory is being shaped by the trend toward natural-looking, tailored aesthetic results as well as rising patient knowledge of cutting-edge dermatological treatments.
Restrain/Challenge
High Cost of Modern Aesthetic Injectable Treatments
One major obstacle to the bioregenerative aesthetic injectable market penetration is the comparatively high cost of modern aesthetic dermatological treatments, especially for middle-class or price-sensitive populations. Certain populations may not be able to afford procedures such as PRP injectables, exosome-based injectables, and peptide-based injectables. Strict regulatory frameworks and compliance criteria for the approval of biologics, injectable medicines, and dermatological devices can lengthen the time it takes for innovations to reach the market and delay the introduction of new goods. Additionally, prospective patients may be reluctant to employ newer or combination medicines due to worries about procedural safety, possible side effects, and treatment success variability. The provision of high-quality care may be limited in some areas due to a lack of qualified dermatologists and cosmetic professionals, which could hinder the bioregenerative aesthetic injectable market expansion.
Biostimulatory Injectables Segment is Expected to Drive the Bioregenerative Aesthetic Injectable Market
The biostimulatory injectables category held the largest share in the Bioregenerative Aesthetic Injectable market in 2025 because it can stimulate the body's natural production of collagen, resulting in long-lasting, natural-looking cosmetic benefits. The biostimulatory injectables like poly-L-lactic acid and calcium hydroxylapatite encourage progressive tissue regeneration and structural development, in contrast to conventional fillers that offer instant volume. Additionally, its adoption has been fueled by patients' growing demand for minimally invasive procedures with long-lasting results, less frequent maintenance, and better skin quality. The segment's dominant market position is further supported by growing clinical indications, growing utilization in combination medicines, and strong physician confidence.
Skin Hydration & Moisturization (For Face) Segment is Growing at the Highest Rate in the Bioregenerative Aesthetic Injectable Market
In 2025, the skin hydration & moisturization (for face) category dominated the Bioregenerative Aesthetic Injectable market. This dominance is mostly due to consumers' increasing focus on long-term dermal health, skin quality, and radiance rather than structural modification. Strong acceptance in urban areas has been facilitated by growing demand for minimally invasive procedures that improve skin texture, moisture, and elasticity. Additionally, both younger consumers looking for preventive treatment and older patients focusing on early indications of aging are drawn to products that emphasize deep skin hydration and collagen activation. Furthermore, practitioner endorsements and growing clinic networks support segment leadership throughout the region.
Why North America Led the Bioregenerative Aesthetic Injectable Market?
The Bioregenerative Aesthetic Injectable market was dominated by North America region in 2025 due to the high cost of healthcare, the prevalence of top dermatology clinics, and the rising popularity of customized cosmetic procedures. Technological developments, growing acceptance of injectables, and innovations from both well-known medical device manufacturers and startups concentrating on minimally invasive cosmetic solutions all contributed to the bioregenerative aesthetic injectable market's significant growth in aesthetic dermatology procedures.
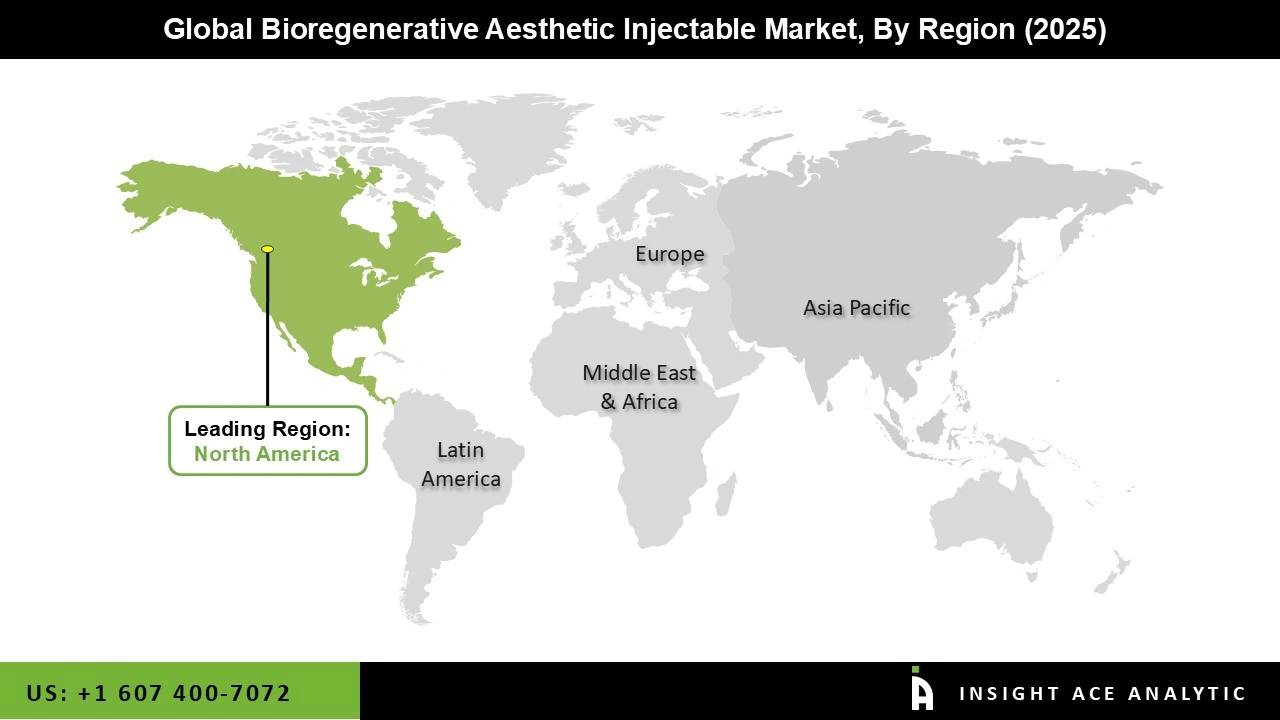
Additionally, consumers in the area are looking more and more for combination treatments, non-invasive procedures, and innovative skin therapies to get results that seem natural. Furthermore, a strong healthcare system, broad insurance coverage for certain operations, and a skilled network of dermatologists and aesthetic practitioners all contribute to the region's acceptance.
Key Development
November 2025: To meet the growing demand for regenerative aesthetics, Germany's Merz Aesthetics introduced two flagship injectables in China: Xeomin and Radiesse. In light of stricter regulations and rising consumer interest in biostimulators, Merz's long-term China strategy is strengthened by this action.
October 2025: In order to increase its presence in Canada, Croma-Pharma GmbH and Renellence formed a strategic distribution alliance. While both businesses work together on physician education, training, and long-term expansion in the Canadian aesthetics industry, Renellence will distribute Croma's MDR-approved Saypha hyaluronic acid fillers.
Bioregenerative Aesthetic Injectable Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 502.48 Mn |
| Revenue forecast in 2035 | USD 1,503.99 Mn |
| Growth Rate CAGR | CAGR of 11.9% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Mn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026 to 2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Application, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | PharmaResearch Products Co., Ltd., MD Skin Solutions, Caregen Co. Ltd, BR Pharma, Fillmed, Croma Pharma, PROMOITALIA, Galderma, Fidia, IBSA, Bio-Formula, Allergan (AbbVie), VAIM, Mastelli s.r.l., and Merz Aesthetics |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentations of Bioregenerative Aesthetic Injectable Market :
Bioregenerative Aesthetic Injectable Market, by Product Type-
- PN
- PDRN
- PN Injectables
- Exosome-based Injectables
- Peptide-based Injectables
- Biostimulatory Injectables
- CaHA
- PLLA
- PRP Injectables
- Others
Bioregenerative Aesthetic Injectable Market, by Application-
- Skin Hydration & Moisturization (For Face)
- Acne Scar (For Face)
- Elasticity & Firmness (For Face)
- Fine Lines (For Face)
- Under eye-areas (For Face)
- Wrinkle Correction (For Face)
- Body Application
- Arms & Thighs
- Knees
- Décolletage
- Scalp & Hair Rejuvenation
- Intimate Areas
Bioregenerative Aesthetic Injectable Market, by End-user-
- MedSpas
- Hospitals
- Aesthetic Surgery Centers
- Dermatology Centers
- Others
Bioregenerative Aesthetic Injectable Market, by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Bioregenerative Aesthetic Injectable Market Size is valued at USD 502.48 Mn in 2025 and is predicted to reach USD 1,503.99 Mn by the year 2035
Bioregenerative Aesthetic Injectable Market is predicted to grow at an 11.9% CAGR during the forecast period for 2026 to 2035.
PharmaResearch Products Co., Ltd., MD Skin Solutions, Caregen Co. Ltd, BR Pharma, Fillmed, Croma Pharma, PROMOITALIA, Galderma, Fidia, IBSA, Bio-Formula, Allergan (AbbVie), VAIM, Mastelli s.r.l., Merz Aesthetics and Others
Bioregenerative Aesthetic Injectable Market is segmented into Product Type, Application, End-user and other.
North America region is leading the Bioregenerative Aesthetic Injectable Market.
