Aorto iliac Occlusive Disease Treatment Market Report with Forecast 2026 to 2035
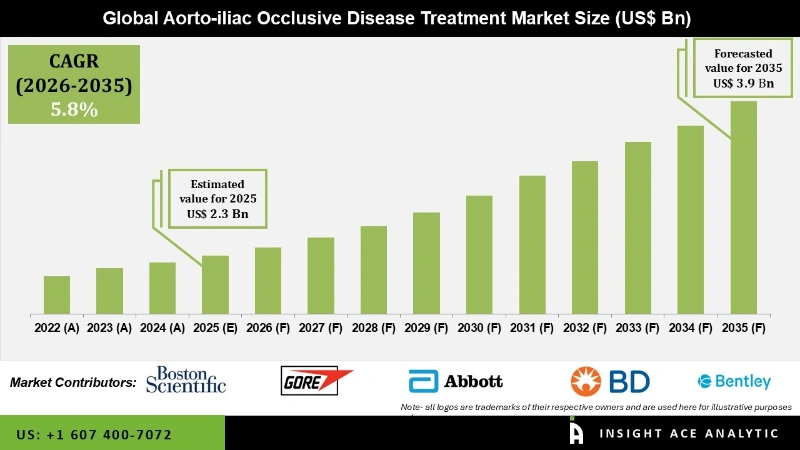
Aorto iliac Occlusive Disease Treatment Market Size is valued at USD 2.3 Bn in 2025 and is predicted to reach USD 3.9 Bn by the year 2035 at a 5.8% CAGR during the forecast period for 2026 to 2035.
Aorto-iliac Occlusive Disease Treatment Market Size, Share & Trends Analysis Distribution by Device Type (Endovascular Devices (Atherectomy Systems, Balloon Angioplasty Devices, Stents, and Surgical Devices), By Procedure (Endovascular Procedures, Hybrid Procedures, and Open Surgical Procedures), By End-user (Hospitals, Outpatient Facilities, and Others), and Segment Forecasts, 2026 to 2035
The goals of treating aorto-iliac occlusive disease (AIOD) are to prevent limb-threatening consequences, relieve symptoms like claudication and ischemic discomfort, and restore appropriate blood flow to the lower extremities. To address risk factors like hypertension, diabetes, and hyperlipidemia, management usually starts with conservative measures like quitting smoking, participating in organized exercise programs, and using antiplatelet and statin medications to slow the advancement of the diabetes condition.
The revascularization is necessary in patients with severe to moderate symptoms or critical limb ischemia. The minimally invasive nature of endovascular therapies, their quicker recovery times, and their positive results in appropriate lesions make them popular. One of the main drivers propelling the aorto-iliac occlusive disease treatment market's growth is the rising incidence of peripheral artery disease (PAD).
The aorto-iliac occlusive disease treatment market is expanding steadily as a result of the aging population and the rising prevalence of lifestyle-related risk factors such as obesity, diabetes, smoking, and hypertension. One of the main causes of lower-limb ischemia is aorto-iliac occlusive disease, and since this condition is diagnosed more frequently, there is a growing need for efficient treatment.
The acceptance of medicinal therapy, endovascular therapies, and surgical revascularization techniques has accelerated due to early detection made possible by improvements in diagnostic imaging and increased awareness among patients and doctors. Additionally, advancements in vascular care accessibility and healthcare infrastructure are fostering the aorto-iliac occlusive disease treatment market growth in both developed and developing nations.
The aorto-iliac occlusive disease treatment market is being greatly driven by technological developments in minimally invasive endovascular therapies, such as angioplasty balloons, bare-metal and drug-eluting stents, and coated stent grafts.
Compared to open surgery, these treatments are becoming more and more popular since they need fewer hospital stays, have fewer procedural risks, and require less recovery time. The use of innovative treatment modalities is also being encouraged by rising healthcare costs and advantageous payment systems in a number of nations. However, the high cost of cutting-edge endovascular devices, the scarcity of qualified vascular experts, and the dangers associated with procedures in patients with serious comorbidities are limiting the aorto-iliac occlusive disease treatment market's expansion.
Competitive Landscape
Which are the Leading Players in Aorto-iliac Occlusive Disease Treatment Market?
• Medtronic
• Cook
• BD
• Bentley InnoMed
• Boston Scientific Corporation
• W. L. Gore & Associates, Inc.
• Terumo Corporation
• Scitech Medical
• Abbott
• iVascular
• Getinge AB
• Biotronik SE & Co KG
Market Dynamics
Driver
Growing Prevalence of Peripheral Artery Disease (PAD)
Peripheral artery disease (PAD), which frequently affects the aorto-iliac arteries, is becoming more common as more people suffer from lifestyle-related illnesses like diabetes, high blood pressure, and obesity. The American Heart Association estimates that peripheral artery disease affects 200 million people globally. Over time, these medical conditions damage blood vessels, increasing a person's risk of significant artery blockages. Because of this, more people are being diagnosed with aorto-iliac occlusive disease, which is increasing the need for efficient treatment alternatives. Improvements in PAD awareness and early detection have resulted in prompt medical therapies. Healthcare providers and manufacturers of medical devices are being pushed by this trend to increase the number of treatment options available for these illnesses.
Restrain/Challenge
High Cost of Advanced Endovascular Devices and Intricate Surgical Procedures
The high cost of sophisticated endovascular devices and intricate surgical procedures is one of the main barriers to the aorto-iliac occlusive disease treatment market. Costly equipment, specialized operating rooms, and highly qualified vascular surgeons or interventional radiologists are needed for operations like hybrid revascularization, angioplasty with drug-eluting or coated stents, and stent graft systems. These expenses greatly raise the total cost of treatment, which limits access to sophisticated therapies, especially in low- and middle-income nations. The hospitals are further deterred from implementing modern technologies by the restricted or uneven payment for specific endovascular procedures in some areas.
Endovascular Procedures Segment is Expected to Drive the Aorto-iliac Occlusive Disease Treatment Market
The endovascular procedures segment held the largest share in the aorto-iliac occlusive disease treatment market in 2025 because of its increasing recognition as the primary therapy for aorto-iliac occlusive disease. These techniques' less intrusive nature enables quicker return to normal activities, shorter hospital stays, and less post-operative problems. Because angioplasty and stenting are repeatable, affordable, and have a positive risk-benefit profile—especially in patients with comorbidities—doctors like them. Additionally, increasing access to cath laboratories in metropolitan hospitals and extensive training in endovascular procedures have increased procedure volumes. Their quick incorporation into standard practice has also been facilitated by ongoing clinical trials and advantageous reimbursement regulations.
Hospitals Segment is Growing at the Highest Rate in the Aorto-iliac Occlusive Disease Treatment Market
In 2025, the hospitals segment dominated the aorto-iliac occlusive disease treatment market. To manage complex aorto-iliac occlusive disease, hospitals are progressively adding sophisticated and minimally invasive treatments to their treatment portfolios. For instance, NYU Langone Health has adopted minimally invasive techniques, including balloon angioplasty with stenting and atherectomy. Because these procedures are carried out under local anesthesia, patients can frequently return home the same day, which shortens hospital stays and speeds up recovery. Additionally, complex aorto-iliac reconstructions are supported by the availability of hybrid operating rooms, vascular intensive care units, and round-the-clock specialized care.
Why North America Led the Aorto-iliac Occlusive Disease Treatment Market?
The aorto-iliac occlusive disease treatment market was dominated by North America region in 2024 because of early diagnosis methods, robust reimbursement mechanisms, and the extensive use of modern endovascular procedures. Patients now have easier access to efficient therapy because of a strong healthcare system and a large number of vascular specialists. Furthermore, early management and improved results have resulted from greater screening for high-risk patients and growing awareness of peripheral artery disease.
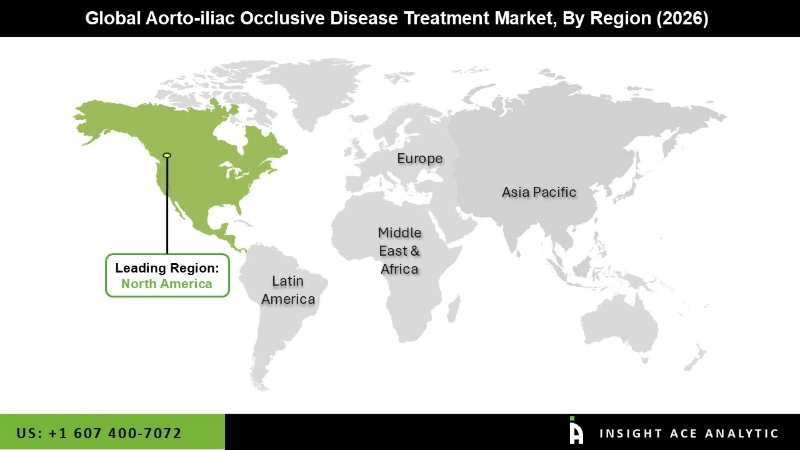
Additionally, a high rate of new medical device adoption and continuous clinical research have contributed to consistent growth. Faster introduction of novel solutions is ensured by ongoing investments in vascular research and development, as well as robust regulatory support for device approvals. Leadership in this area has also been bolstered by favorable insurance policies and the increasing need for outpatient vascular care.
Key Development
March 2025: Balloon angioplasty, particularly in the femoro-popliteal and infra-popliteal segments, is still a key treatment for lower extremity PAD, according to the Washington Healthcare Authority. The study highlighted how it can be used to improve blood flow and preserve limbs, indicating that drug-coated balloon technologies are becoming more widely used.
January 2024: The NMPA approved Zylox-Tonbridge Medical Technology Co., Ltd.'s ZYLOX Peripheral Venous Stent System for use in treating pelvic and lower limb venous diseases brought on by iliac vein compression.
Aorto-iliac Occlusive Disease Treatment Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 2.3 Bn |
| Revenue forecast in 2035 | USD 3.9 Bn |
| Growth Rate CAGR | CAGR of 5.8% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Device Type, Procedure, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Medtronic, Cook, BD, Bentley InnoMed, Boston Scientific Corporation, W. L. Gore & Associates, Inc., Terumo Corporation, Scitech Medical, Abbott, iVascular, Getinge AB, and Biotronik SE & Co KG. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Aorto-iliac Occlusive Disease Treatment Market by Device Type
- Endovascular Devices
-
- Atherectomy Systems
- Balloon Angioplasty Devices
- Stents
-
-
- Self-Expanding Stents
- Balloon-Expandable Stents
- Bifurcated Aortic Stent Grafts
- Covered Stent Grafts
-
- Surgical Devices
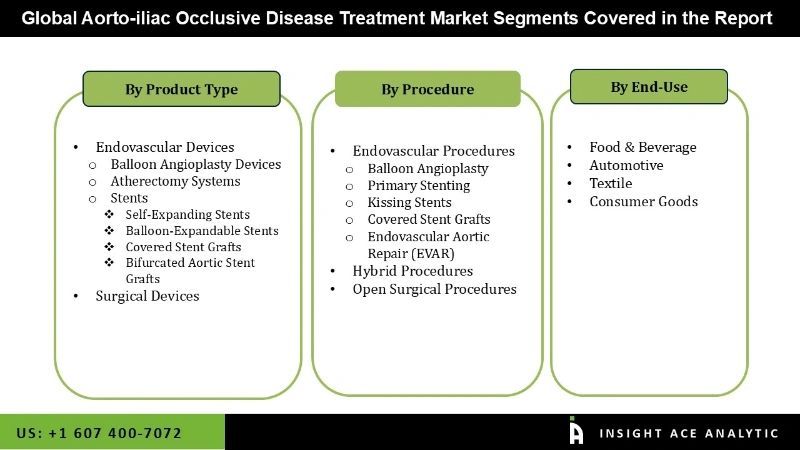
Aorto-iliac Occlusive Disease Treatment Market by Procedure
- Endovascular Procedures
-
- Primary Stenting
- Balloon Angioplasty
- Covered Stent Grafts
- Kissing Stents
- Endovascular Aortic Repair (EVAR)
-
- Hybrid Procedures
- Open Surgical Procedures
Aorto-iliac Occlusive Disease Treatment Market by End-user
- Hospitals
- Outpatient Facilities
- Others
Aorto-iliac Occlusive Disease Treatment Market-By Region
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Aorto iliac Occlusive Disease Treatment Market Size is valued at USD 2.3 Bn in 2025 and is predicted to reach USD 3.9 Bn by the year 2035
Aorto iliac Occlusive Disease Treatment Market is likely to grow at a 5.8% CAGR during the forecast period for 2026 to 2035.
Medtronic, Cook, BD, Bentley InnoMed, Boston Scientific Corporation, W. L. Gore & Associates, Inc., Terumo Corporation, Scitech Medical, Abbott, iVascular, Getinge AB, and Biotronik SE & Co KG.
Device Type, Procedure, End-user, and By Region are the key segments of the Aorto iliac Occlusive Disease Treatment Market.
North America region is leading the Aorto iliac Occlusive Disease Treatment Market.