Advanced Drug Delivery CDMO Market Size, Trend, Revenue Report 2026 to 2035
What is Advanced Drug Delivery CDMO Market Size?
Global Advanced Drug Delivery CDMO Market Size is predicted to show a 5.00% CAGR during the forecast period for 2026 to 2035.
Advanced Drug Delivery CDMO Market Size, Share & Trends Analysis Report By Service Type (Formulation Development, Process Development and Scale-Up, Analytical and Testing Services, Manufacturing Services, Packaging and, Device Development, Regulatory Support), By Technology (Targeted Drug Delivery Systems, Controlled/Sustained-Release Technologies, Injectable Delivery Platforms, Pulmonary & Nasal Delivery Systems, Transdermal & Microneedle Delivery, Mucosal Delivery Methods, Implantable Delivery Devices, 3D-Printed Drug Delivery), By Dosage Form, By Application, By End-User By Region, And By Segment Forecasts, 2026 to 2035.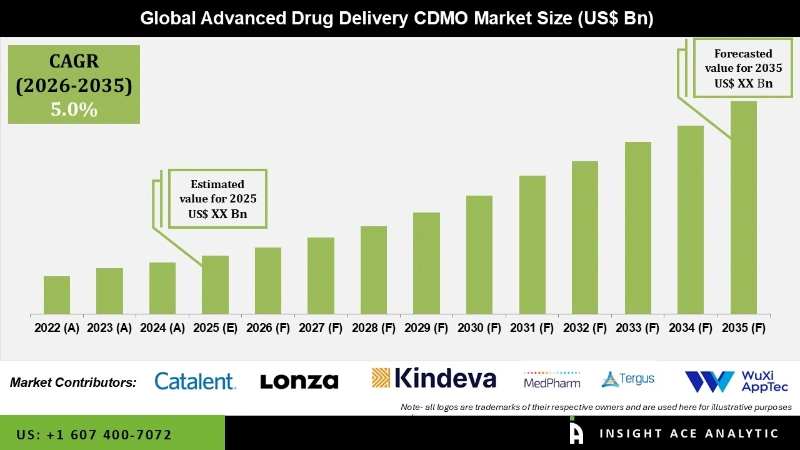
Advanced Drug Delivery CDMO Market Key Takeaways:
|
The pharmaceutical sector greatly benefits from a Contract Development and Manufacturing Organisation (CDMO) specialising in cutting-edge drug delivery technologies. These CDMOs offer services for creating, formulating, and developing drug delivery systems beyond conventional oral tablets or capsules. Modern medication delivery systems have arisen as a quick and precise way to get the drug to the right place. These systems have several advantages over conventional systems, including the focused and regulated delivery of novel molecules to target areas, consistent drug absorption, reduced frequency of dosing, decreased production of hazardous metabolites, and lower variability in systemic drug concentrations.
The need for advanced drug delivery CDMOs has increased as a result of improved drug performance and superior results brought about by advanced drug delivery systems. However, the Advanced Drug Delivery Contract Development and Manufacturing Organisation (CDMO) market was significantly impacted by the COVID-19 pandemic, which had a wide-ranging effect on the sector. The pandemic affected the availability of raw materials and parts needed for medicine delivery systems by upsetting global supply chains. For several CDMOs, this resulted in manufacturing difficulties and delivery delays.
Competitive Landscape
Some Major Key Players in the Advanced Drug Delivery CDMO Market are:
- Catalent, Inc.
- Lonza Group AG
- Kindeva Drug Delivery
- MedPharm (merged with Tergus Pharma)
- WuXi AppTec
- Hovione
- Piramal Pharma Solutions
- CordenPharma International
- Divi’s Laboratories
- Lifecore Biomedical
- Recipharm AB
- Syngene International
- Thermo Fisher Scientific (Patheon)
- Aenova Group
- Cellipoint Bioservices
- Evonik Health Care
- SHL Medical
- Lubrizol Life Science
- Acino International AG
- Pharmathen
- Phillips-Medisize (A Molex Company)
- Others
Market Segmentation:
The advanced drug delivery CDMO market is segmented on the basis of services which is segmented as service type, technology, dosage form, application and end-user. Based on the service type, the market segmented into formulation development, process development and scale-up, analytical and testing services, manufacturing services, packaging and, device development, regulatory support. Based on the technology, the market segmented into targeted drug delivery systems, controlled/sustained-release technologies, injectable delivery platforms, pulmonary & nasal delivery systems, transdermal & microneedle delivery, mucosal delivery methods, implantable delivery devices, 3d-printed drug delivery. Based on the dosage form, the market segmented into injectables, oral solids, topical & transdermal, implantables & ocular. Based on the application, the market segmented into oncology, infectious diseases, neurological disorders, cardiovascular diseases, other. Based on the end-user, the market segmented into clinical, commercial.
The Segment, Analytical and Testing Services, Accounted As A Major Contributor In The Advanced Drug Delivery CDMO Market.
The analytical and testing services category is expected to hold a major share in the global Advanced Drug Delivery CDMO Market in 2025. Advanced drug delivery technologies such as liposomes, nanoparticles, implants, and long-acting injectables require sophisticated testing to ensure their performance, safety, and stability. These systems often exhibit complex release profiles, encapsulation efficiencies, and bioavailability parameters that must be validated using high-precision analytical methods. Analytical services are essential at every stage of the drug development lifecycle, from preformulation and clinical trials to regulatory submission and post-marketing surveillance. As a result, the Analytical and Testing Services segment holds the largest share in the advanced drug delivery CDMO market, as it plays a critical role in ensuring the quality, safety, regulatory compliance, and functional performance of increasingly complex drug delivery systems.
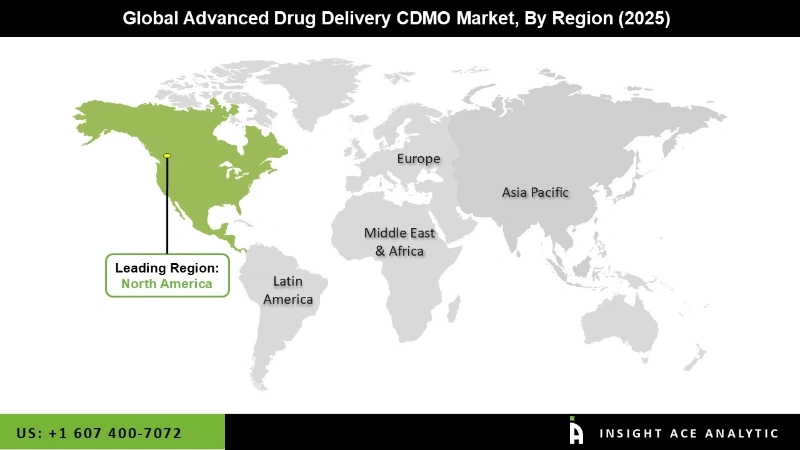
In The Region, The North American Advanced Drug Delivery CDMO Market Holds A Significant Revenue Share.
The North America Advanced Drug Delivery CDMO Market is expected to record the maximum market share in terms of revenue in the near future. The region's long-standing CDMO presence distinguishes the market in North America. The region is heavily invested in researching and developing biological medications, gene treatments, and other specialized pharmaceuticals. These complex compounds frequently necessitate specialized drug delivery devices, which drives demand for CDMO services. Along with this element, expanding partnerships between pharmaceutical firms and CDMOs providing a wide range of services are to blame for the region's leading market share globally. Due to the region's highly qualified workforce and low manufacturing and R&D costs, the Asia Pacific market is estimated to grow significantly over the course of the forecast period.
Recent Developments:
- In July 2023, SHL Medical said it had bought LCA Automation AG, a Swiss company that makes innovative automation solutions. The purchase was SHL Medical's answer to the growing need for drug delivery options in the market. It also helped its manufacturing operations around the world, especially the new factory that will open in Zug, Switzerland.
- In March 2023, Evonik has solidified its prominent position in drug delivery for RNA treatments by initiating the construction process for a new pharmaceutical lipids plant in the United States. The newly established facility was anticipated to ensure the industry's uninterrupted availability of essential excipients required for mRNA vaccines and other nucleic acid therapeutics. The commencement of plant operations was planned for the year 2025.
Advanced Drug Delivery CDMO Market Report Scope :
| Report Attribute | Specifications |
| Growth Rate CAGR | CAGR of 5.00% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Mn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | By Service Type, Technology, Dosage Form, Application, End-User |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; South East Asia |
| Competitive Landscape | Catalent, Inc., Lonza Group AG, Kindeva Drug Delivery, MedPharm (merged with Tergus Pharma), WuXi AppTec, Hovione, Piramal Pharma Solutions, CordenPharma International, Divi’s Laboratories, Lifecore Biomedical, Recipharm AB, Syngene International, Thermo Fisher Scientific (Patheon), Aenova Group, Cellipoint Bioservices, Evonik Health Care, SHL Medical, Lubrizol Life Science, Acino International AG, Pharmathen, Phillips-Medisize (A Molex Company), Others |
| Customization Scope | Free customization report with the procurement of the report and modifications to the regional and segment scope. Particular Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of Advanced Drug Delivery CDMO Market -
Advanced Drug Delivery CDMO Market, By Service Type
- Formulation Development
- Process Development and Scale-Up
- Analytical and Testing Services
- Manufacturing Services
- Packaging and Device Development
- Regulatory Support
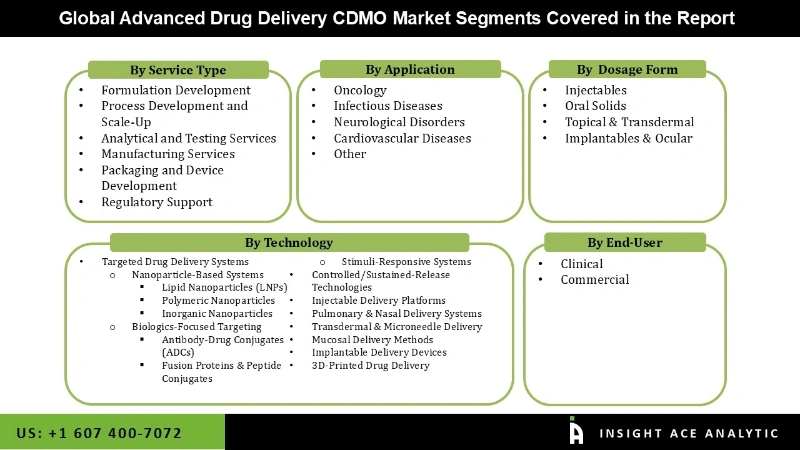
Advanced Drug Delivery CDMO Market, By Technology
- Targeted Drug Delivery Systems
- Nanoparticle-Based Systems
- Lipid Nanoparticles (LNPs)
- Polymeric Nanoparticles
- Inorganic Nanoparticles
- Biologics-Focused Targeting
- Antibody-Drug Conjugates (ADCs)
- Fusion Proteins & Peptide Conjugates
- Stimuli-Responsive Systems
- Nanoparticle-Based Systems
- Controlled/Sustained-Release Technologies
- Injectable Delivery Platforms
- Pulmonary & Nasal Delivery Systems
- Transdermal & Microneedle Delivery
- Mucosal Delivery Methods
- Implantable Delivery Devices
- 3D-Printed Drug Delivery
Advanced Drug Delivery CDMO Market, By Dosage Form
- Injectables
- Oral Solids
- Topical & Transdermal
- Implantables & Ocular
Advanced Drug Delivery CDMO Market, By Application
- Oncology
- Infectious Diseases
- Neurological Disorders
- Cardiovascular Diseases
- Other
Advanced Drug Delivery CDMO Market, By End-User
- Clinical
- Commercial
Advanced Drug Delivery CDMO Market, By Region-
North America-
- The US
- Canada
- Mexico
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Advanced Drug Delivery CDMO Market Size is predicted to show a 5.00% CAGR during the forecast period for 2026-2035.
Evonik Health Care, SHL Medical, Lubrizol Life Science, Acino International AG, Pharmathen, Phillips-Medisize (A Molex Company), and others.
Advanced Drug Delivery CDMO Market is segmented on the basis of services which is segmented as Analytical Services, Assembly, Labelling and Packaging,
North American region is leading the Advanced Drug Delivery CDMO Market.