Nose-to-Brain Drug Delivery Devices Market Size and Scope 2026 to 2035
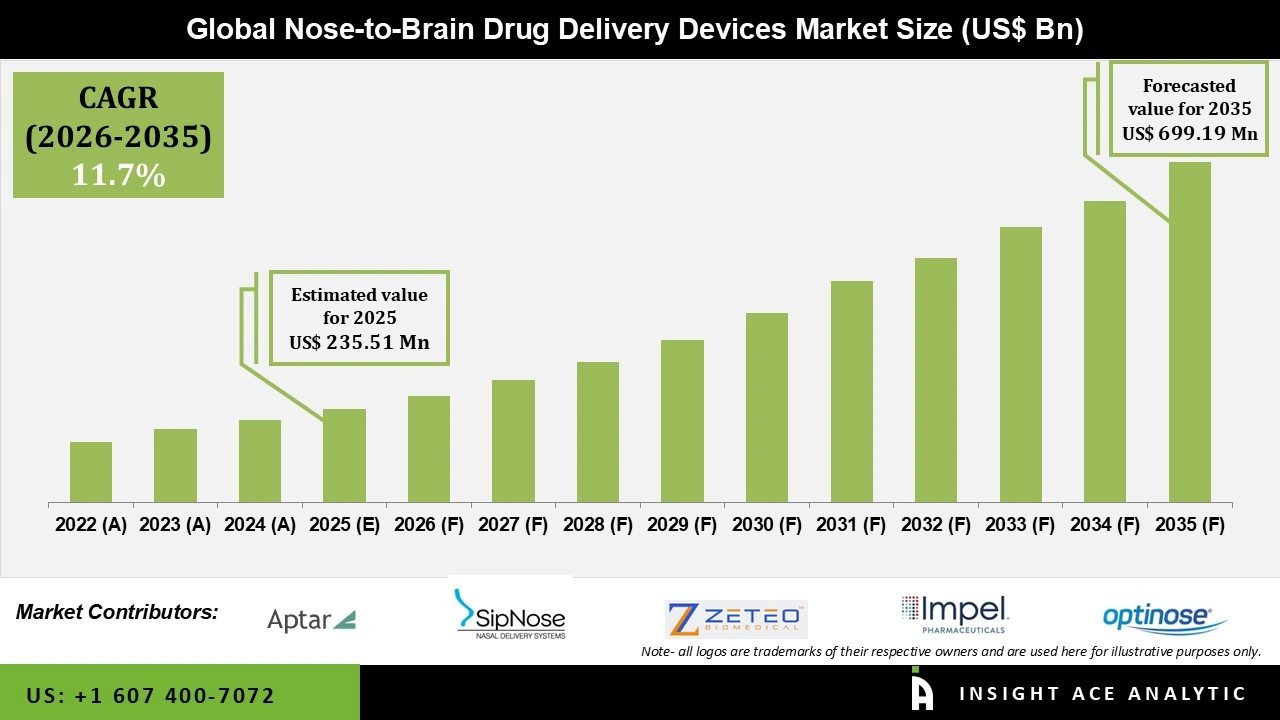
Global Nose-to-Brain Drug Delivery Devices Market Size is valued at USD 235.51 Mn in 2025 and is predicted to reach USD 699.19 Mn by the year 2035 at a 11.7% CAGR during the forecast period for 2026 to 2035.
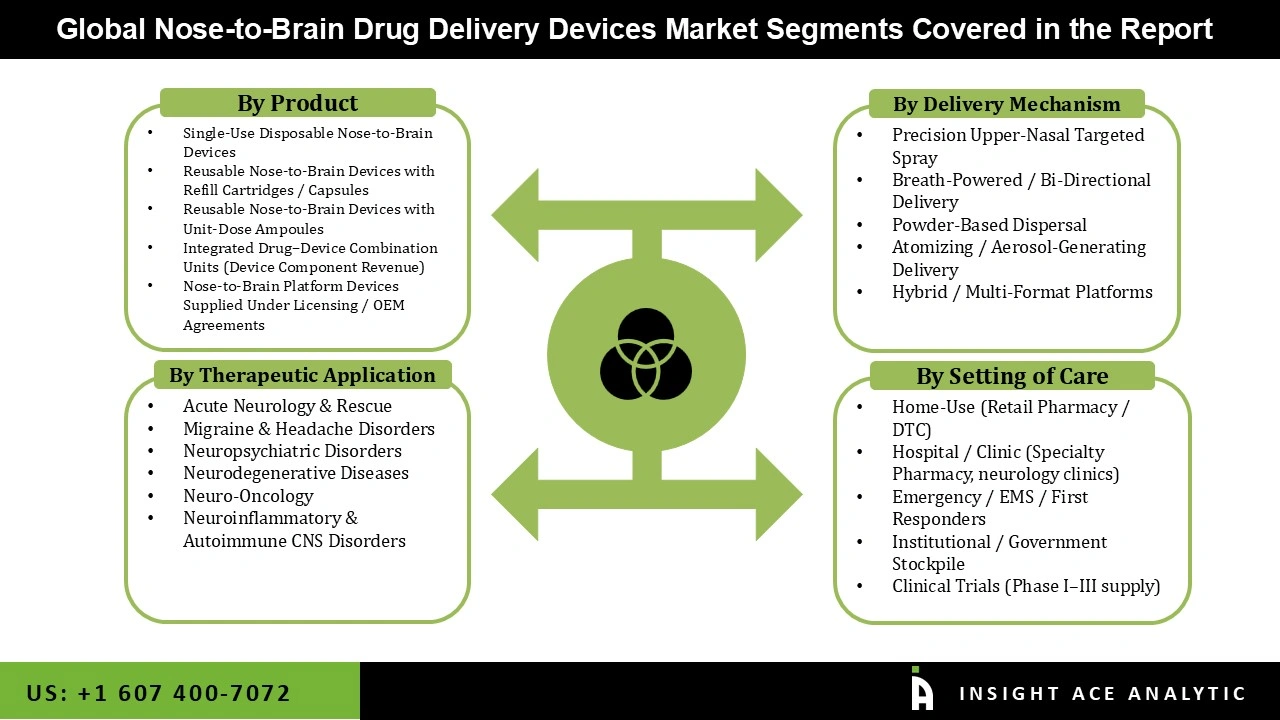
Nose-to-Brain Drug Delivery Devices Market Size, Share & Trends Analysis Distribution by Product (Single-Use Disposable Nose-to-Brain Devices, Reusable Nose-to-Brain Devices with Refill Cartridges / Capsules, Reusable Nose-to-Brain Devices with Unit-Dose Ampoules, Integrated Drug–Device Combination Units (Device Component Revenue, Nose-to-Brain Platform Devices Supplied Under Licensing / OEM Agreements), By Delivery Mechanism (Precision Upper-Nasal Targeted Spray, Breath-Powered / Bi-Directional Delivery, Powder-Based Dispersal, Atomizing / Aerosol-Generating Delivery, Hybrid / Multi-Format Platforms), By Therapeutic Application, By Setting of Care, By Region and Segment Forecasts, 2026 to 2035
Nose-to-brain delivery is a non-invasive intranasal drug administration approach in which therapeutics are transported directly from the nasal cavity to the brain via the olfactory and trigeminal nerve pathways, effectively bypassing the blood–brain barrier (BBB). By targeting the olfactory epithelial region of the nasal passage, this route enables rapid and direct drug transport to the central nervous system (CNS), offering significantly faster onset of action compared with conventional oral or injectable therapies. Nose-to-brain drug delivery devices include advanced technologies such as unidose nasal sprays, powder-based delivery systems, catheter-based atomizers, and precision metered-dose devices. These systems are engineered to tightly control droplet or particle size, spray pattern, deposition angle, and delivery velocity, thereby optimizing transport to central brain pathways.These devices are increasingly being explored for the treatment of a wide range of neurological and cerebral disorders, including Parkinson’s disease, Alzheimer’s disease, epilepsy and seizure disorders, stroke, brain tumors, and Huntington’s disease.
In addition, they are used in acute and emergency therapeutic applications such as rapid migraine relief and other conditions requiring immediate systemic action where fast drug absorption is critical. The technology is also gaining momentum for the delivery of biologics, oligopeptides, and nucleic-acid–based therapeutics, particularly for molecules that cannot be effectively administered through traditional oral or injectable routes without complex delivery enhancements.
The nose-to-brain drug delivery devices market for CNS therapeutics is being driven primarily by the significant unmet medical need in neurological disorders, where many drugs cannot be effectively administered through conventional routes due to the restrictive nature of the blood–brain barrier (BBB). Market growth has been further accelerated by the increasing preference for minimally invasive, patient-friendly, and needle-free drug delivery technologies. The rapid onset of action and improved bioavailability achievable through intranasal administration are particularly advantageous for acute and emergency CNS applications. In parallel, expanding research and development efforts focused on intranasal formulations for biologics, peptides, and advanced CNS drugs are strengthening long-term market potential. However, market expansion is constrained by several technical and physiological challenges. These include limited permeability caused by nasal mucosal barriers, rapid mucociliary clearance particularly for large molecules formulation stability issues, device design complexity, and difficulties in achieving consistent and accurate drug deposition within the olfactory region.
Competitive Landscape
Which are the Leading Players in the Nose-to-Brain Drug Delivery Devices Market?
- Aptar Pharma
- SipNose
- Zeteo Biomedical
- Impel Pharmaceuticals
- Optinose
- Kurve Therapeutics
- Nemera
- Kindeva Drug Delivery
- Hovione
- Upperton Pharma Solutions
- Shin Nippon Biomedical Laboratories
- Neurelis
- ARS Pharmaceuticals
- Belhaven Biopharma
- Tiziana Life Sciences
- Others
Market Dynamics
Driver
Unmet Need for Effective Central Nervous System Drug Delivery Due to Blood–Brain Barrier Constraints
The primary factor driving the nose-to-brain drug delivery devices market is the growing need for effective management of central nervous system disorders, primarily due to the limitations imposed by the blood-brain barrier. Traditional oral and parenteral administration routes are often inadequate for achieving therapeutic drug concentrations in the brain. In contrast, nose-to-brain delivery enables direct and rapid transport of drugs to the central nervous system, thereby enhancing efficacy and reducing onset time. Consequently, the rising prevalence of neurological and psychiatric disorders, along with increased investment in central nervous system drug development, has significantly contributed to the demand for nose-to-brain drug delivery devices.
Restrain/Challenge
Physiological Limitations of the Nasal Cavity and Rapid Mucociliary Clearance
The primary challenge in the nose to brain drug delivery devices market has been the biological constraints of the nasal cavity, particularly its limited drug-carrying capacity and mucociliary clearance rate. These aspects limit the amount of drugs as well as their nature capable of reaching the brain and thus pose a challenge in targeting the CNS efficiently.
Single-use Disposable Devices Segment is Expected to Drive the Nose-to-Brain Drug Delivery Devices Market
The nose-to-brain delivery devices market is currently dominated by single-use disposable devices, accounting for the largest revenue share. Their dominance is driven by strong adoption in approved and late-stage CNS therapies, particularly for acute and emergency indications such as migraine, seizures, and overdose reversal. These devices offer advantages including ease of use, rapid drug administration, accurate dosing, lower contamination risk, and regulatory preference. Additionally, their lower manufacturing complexity, cost efficiency, and compatibility with large-scale fill-finish operations support widespread commercialization. While reusable and platform-based systems are emerging, disposable devices remain the primary choice for current nose-to-brain drug delivery applications.
Precision Upper-Nasal Targeted Spray Segment is Growing at the Highest Rate in the Nose-to-Brain Drug Delivery Devices Market
The nose-to-brain delivery devices market is currently led by precision upper-nasal targeted spray systems, as they represent the most commercially established delivery mechanism. Their dominance is supported by broad integration into marketed intranasal CNS therapies, manufacturing scalability, and compatibility with existing pharmaceutical fill-finish infrastructure. These systems offer predictable performance and standardized dosing, making them the preferred option for commercial deployment. These devices enable controlled droplet size, spray pattern, and delivery velocity, supporting consistent brain targeting and rapid onset of action.
Why North America Led the Nose-to-Brain Drug Delivery Devices Market?
North America has been the largest and leading region in the global nose-to-brain (intranasal) drug delivery devices market, as highlighted across multiple industry analyses of the broader intranasal delivery segment. With a prominent market share, North America held a leading position in global revenues. This was due to the region's already-established state-of-the-art healthcare infrastructure, supported by advanced clinical and diagnostic capabilities on par with those of developed markets, thereby enabling early adoption of novel intranasal and nose-to-brain delivery technologies.
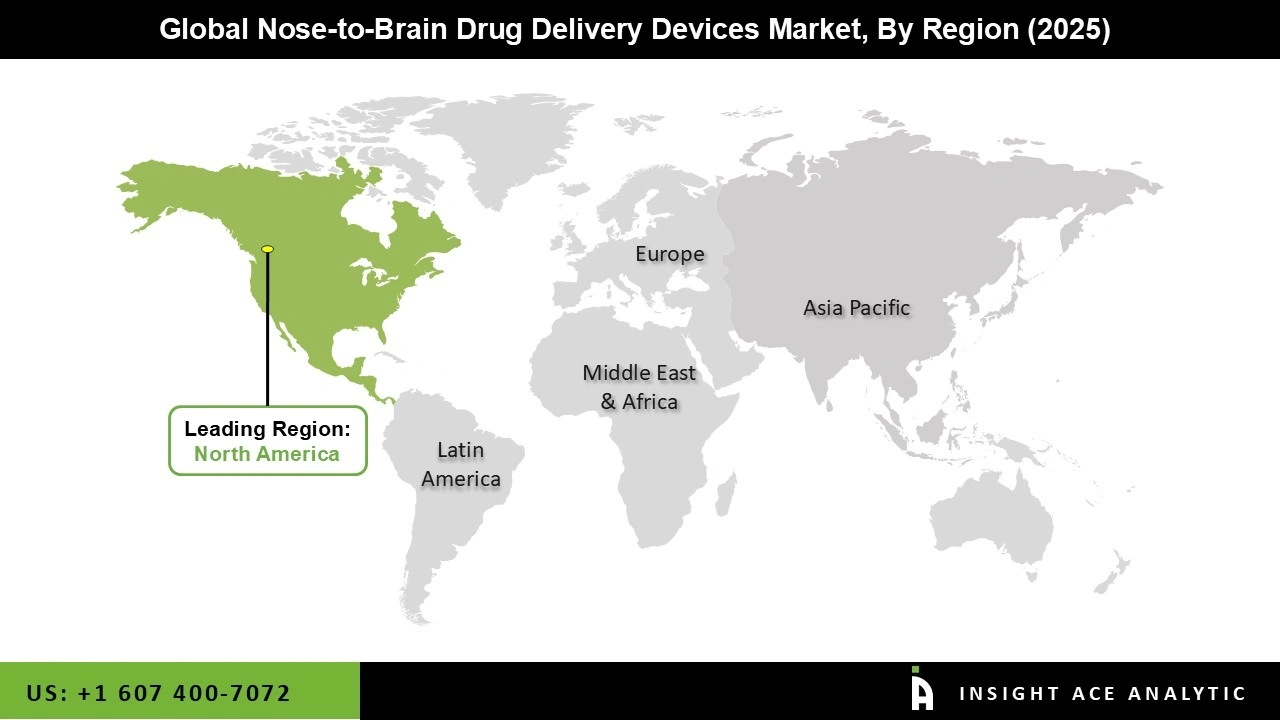
In the meanwhile, strong investments in R&D by big pharmaceuticals and medical device companies in the United States and Canada fast-tracked the development of sophisticated formulations and precision delivery devices for CNS and other complex therapies. Such leadership was further aided by a strong regulatory environment, particularly in the United States, which facilitated faster approvals and the commercialization of novel intranasal drug–device combinations to strengthen North America's dominant market position.
Key Development
- October 2024: Aptar Pharma acquired all device technology assets from SipNose Nasal Delivery Systems, a company specializing in intranasal platforms for local, systemic, and CNS therapies. SipNose had offered devices designed to target specific areas of the nasal cavity, enabling enhanced systemic, local, or direct-to-brain drug delivery.
Nose-to-Brain Drug Delivery Devices Market Report Scope :
| Report Attribute | Specifications |
| Market size value in 2025 | USD 235.51 Mn |
| Revenue forecast in 2035 | USD 699.19 Mn |
| Growth Rate CAGR | CAGR of 11.7% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2024 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | By Product, Delivery Mechanism, Therapeutic Application, Setting of Care and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Aptar Pharma, SipNose, Zeteo Biomedical, Impel Pharmaceuticals, Optinose, Kurve Therapeutics, Nemera, Kindeva Drug Delivery, Hovione, Upperton Pharma Solutions, Shin Nippon Biomedical Laboratories, Neurelis, ARS Pharmaceuticals, Belhaven Biopharma, Tiziana Life Sciences |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentations of Nose-to-Brain Drug Delivery Devices Market:
Nose-to-Brain Drug Delivery Devices Market by Product -
- Single-Use Disposable Nose-to-Brain Devices
- Reusable Nose-to-Brain Devices with Refill Cartridges / Capsules
- Reusable Nose-to-Brain Devices with Unit-Dose Ampoules
- Integrated Drug–Device Combination Units (Device Component Revenue)
- Nose-to-Brain Platform Devices Supplied Under Licensing / OEM Agreements
Nose-to-Brain Drug Delivery Devices Market by Delivery Mechanism -
- Precision Upper-Nasal Targeted Spray
- Breath-Powered / Bi-Directional Delivery
- Powder-Based Dispersal
- Atomizing / Aerosol-Generating Delivery
- Hybrid / Multi-Format Platforms
Nose-to-Brain Drug Delivery Devices Market by Therapeutic Application -
- Acute Neurology & Rescue
- Migraine & Headache Disorders
- Neuropsychiatric Disorders
- Neurodegenerative Diseases
- Neuro-Oncology
- Neuroinflammatory & Autoimmune CNS Disorders
Nose-to-Brain Drug Delivery Devices Market by Setting of Care -
- Home-Use (Retail Pharmacy / DTC)
- Hospital / Clinic (Specialty Pharmacy, neurology clinics)
- Emergency / EMS / First Responders
- Institutional / Government Stockpile
- Clinical Trials (Phase I–III supply)
Nose-to-Brain Drug Delivery Devices Market by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Nose-to-Brain Drug Delivery Devices Market Size is valued at USD 235.51 Mn in 2025 and is predicted to reach USD 699.19 Mn by the year 2035
Nose-to-Brain Drug Delivery DevicesMarket is expected to grow at a 11.7% CAGR during the forecast period for 2026 to 2035
Aptar Pharma, SipNose, Zeteo Biomedical, Impel Pharmaceuticals, Optinose, Kurve Therapeutics, Nemera, Kindeva Drug Delivery, Hovione, Upperton Pharma Solutions, Shin Nippon Biomedical Laboratories, Neurelis, ARS Pharmaceuticals, Belhaven Biopharma, Tiziana Life Sciences and Others
Nose-to-Brain Drug Delivery Devices Market is Segmented in By Product (Single-Use Disposable Nose-to-Brain Devices, Reusable Nose-to-Brain Devices with Refill Cartridges / Capsules, Reusable Nose-to-Brain Devices with Unit-Dose Ampoules, Integrated Drug–Device Combination Units (Device Component Revenue, Nose-to-Brain Platform Devices Supplied Under Licensing / OEM Agreements), By Delivery Mechanism (Precision Upper-Nasal Targeted Spray, Breath-Powered / Bi-Directional Delivery, Powder-Based Dispersal, Atomizing / Aerosol-Generating Delivery, Hybrid / Multi-Format Platforms), Therapeutic Application, Setting of Care, Region and Other
North America region is leading the Nose-to-Brain Drug Delivery Devices Market.