Lipid based Pharma Excipients Market Size, Share, Trend, Revenue Report 2026 to 2035
What is Lipid based Pharma Excipients Market Size?
Lipid based Pharma Excipients Market Size is valued at USD 1.03 Bn in 2025 and is predicted to reach USD 1.96 Bn by the year 2035 at a 7.0% CAGR during the forecast period for 2026 to 2035.
Lipid based Pharma Excipients Market Size, Share & Trends Analysis Distribution by Product Type (Fats, Oils, Phospholipids, Waxes, and Others), Formulation (Injectables, Enteral, and Topical), Functionality (Solubilizers & Wetting Agents, Penetration Enhancers, & Solvents, Structuring Agents & Lubricants, Emollients, Emulsifiers & Surfactants, and Others), End-user (Pharma & Biotech Companies, Contract Research Organizations (CROs) & Contract Development and Manufacturing Organizations (CDMOs), Contract Manufacturing Organizations (CMOs), and Academic & Research Institutes), and Segment Forecasts, 2026 to 2035
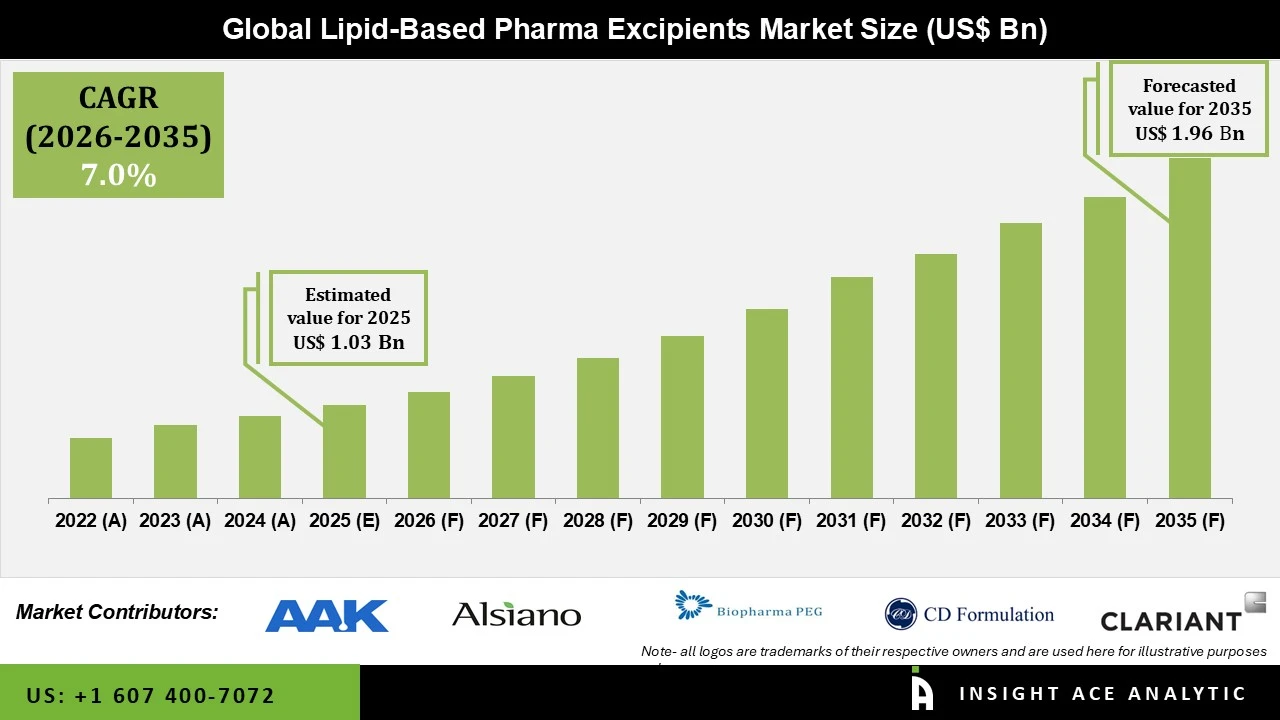
Lipid based Pharma Excipients Market Key Takeaways:
|
Specialized lipid materials called lipid-based pharma excipients are used in drug formulation to increase the solubility, stability, and bioavailability of active pharmaceutical ingredients (APIs). These excipients, which are crucial for creating oral, topical, and parenteral drug delivery systems, include oils, fats, phospholipids, surfactants, and other lipid derivatives. To increase therapeutic efficacy and patient adherence, they are frequently utilized in solid lipid nanoparticles, liposomes, self-emulsifying drug delivery systems (SEDDS), and other cutting-edge drug delivery technologies. Strict regulatory requirements are met in the production of lipid-based pharma excipients, guaranteeing their quality, safety, and uniformity in pharmaceutical applications. The lipid-based pharma excipients market is growing because pharmaceutical companies are dealing with more and more poorly water-soluble active pharmaceutical ingredients (APIs), which call for sophisticated lipid-based excipients and carriers to increase solubility, bioavailability, and targeted administration.
The lipid-based pharma excipients market is expanding significantly as a result of the growing need for improved medication administration, bioavailability, and solubility. A variety of drug delivery systems, including liposomes, lipid nanoparticles, and self-emulsifying drug delivery systems (SEDDS), depend heavily on lipid-based excipients, such as phospholipids, fatty acids, triglycerides, and sterols. Furthermore, the lipid-based pharma excipients market grew steadily due to improvements in drug delivery methods and the growing use of lipid-based formulations in topical, injectable, and enteral medicinal products. Additionally, the lipid-based pharma excipients market is controlled by large companies that prioritize alliances, innovation, and growth tactics. To improve their medication delivery capabilities, corporations are increasingly purchasing smaller businesses with experience in lipid nanoparticle technology.
In addition, the patent expirations for major pharmaceutical medications have also contributed to the growing need for lipid-based pharma excipients by raising competition and lowering drug prices. This has further increased the need for cost-effective excipients. The developments in formulation technology and artificial intelligence (AI) have boosted the lipid-based pharma excipients market growth. Lipid-based formulations can be developed more quickly by incorporating AI-driven modeling, which will improve predictability and shorten time-to-market. Furthermore, the potential for successful lipid-based formulations has been further enhanced by the development of new excipients and formulation methods. Moreover, because of the possibility of interactions between lipid-based pharma excipients and the active ingredient, biological environments, and container closure mechanisms, regulatory authorities have modified rules in response to new knowledge. These factors are anticipated to boost the lipid-based pharma excipients market expansion over the forecast period.
Competitive Landscape
Which are the Leading Players in Lipid-based Pharma Excipients Market?
• ABF Ingredients - ABITEC
• Gattefosé
• AAK
• CREMER OLEO
• IOI Oleo GMbH
• Merck KGaA
• BASF
• Croda International
• Alsiano
• Evonik
• Kao Corporation
• Biopharma PEG Scientific
• CD Formulation
• CordenPharma
• Lipoid
• NOF CORPORATION
• CLARIANT
• Colorcon
Market Dynamics
Driver
Growing Adoption of Gene Therapy
The lipid-based pharma excipients market is anticipated to develop in the future due to the increase in gene therapy. The advances in delivery technologies have made it possible to introduce genetic material into cells in a safe, effective, and focused manner, which has led to an increase in gene therapy. Lipid-based pharma excipients improve gene therapy by facilitating effective delivery systems, guaranteeing genetic material integrity, and facilitating targeted transport. By safeguarding nucleic acids and promoting cellular uptake, they enhance therapeutic results and increase the efficacy of contemporary treatments. For instance, the Food and Drug Administration (FDA) approved six gene therapy products in 2023, up from five in 2022, according to the American Society of Gene & Cell Therapy (ASGCT), a non-profit medical and scientific organization based in the United States. Consequently, the lipid-based pharma excipients market is expanding due to the surge in gene therapy.
Restrain/Challenge
Increasing Stability Issues and Formulation Complexity
The complexity of formulation creation and stability control is one of the main obstacles in the lipid-based pharmaceutical excipients market. To guarantee appropriate drug solubility and reliable performance, lipid-based systems require careful selection of lipid types, surfactants, and cosolvents. These compositions' stability and shelf life may be impacted by environmental elements like temperature, oxidation, and moisture. Additionally, the large-scale manufacturing also poses technological difficulties in maintaining uniformity, which raises development time and production costs. In order to comply with regulations, pharmaceutical businesses must also carry out comprehensive compatibility and stability tests. These technical issues might hinder the development of new products and restrict the use of lipid-based excipients, especially for businesses with little manufacturing or formulation experience.
Oils Segment is Expected to Drive the Lipid-based Pharma Excipients Market
The oils category held the largest share in the Lipid-based Pharma Excipients market in 2025 driven by growing demand in the industrial, personal care, and pharmaceutical industries. The acceptance of nutraceuticals, improvements in lipid-based medication delivery, and a move toward sustainable components are the main causes of this growth. Refined vegetable oils, or PURECO oils from ABITEC, are essential for melting and lubricating a variety of products, including fiberglass and pharmaceuticals. Additionally, vegetable oils, medium-chain triglycerides, and long-chain triglycerides are frequently utilized in lipid-based drug delivery systems, such as soft gelatin capsules and self-emulsifying drug delivery systems. These oils aid in the dissolution of lipophilic active medicinal substances and promote better gastrointestinal absorption. Furthermore, the oil-based excipients category is being driven by the growing number of medication candidates in the pharmaceutical pipeline that have limited water solubility.
Pharma & Biotech Companies Segment is Growing at the Highest Rate in the Lipid-based Pharma Excipients Market
In 2025, the pharma & biotech companies category dominated the Lipid-based Pharma Excipients market. Lipid excipients have gained popularity in pharmaceutical manufacturing and are now mostly utilized to improve the solubility of active compounds. Lipophilic active substances that would otherwise be poorly water-soluble can have their bioavailability increased by using lipid-based formulations. For oral formulations in pharmaceutical manufacturing, these elements are crucial. The greatest demand for lipid-based excipients is driven by pharmaceutical and biotech businesses because of their expanding production capacity, growing competition for market share, desire to provide the best goods, and ability to maintain the highest volume and revenue in the sector. Additionally, the lipid-based pharma excipients market is growing due in large part to investments in research and development, especially in the pharmaceutical end-use sector. As the industry continues to change, this trend is anticipated to continue.
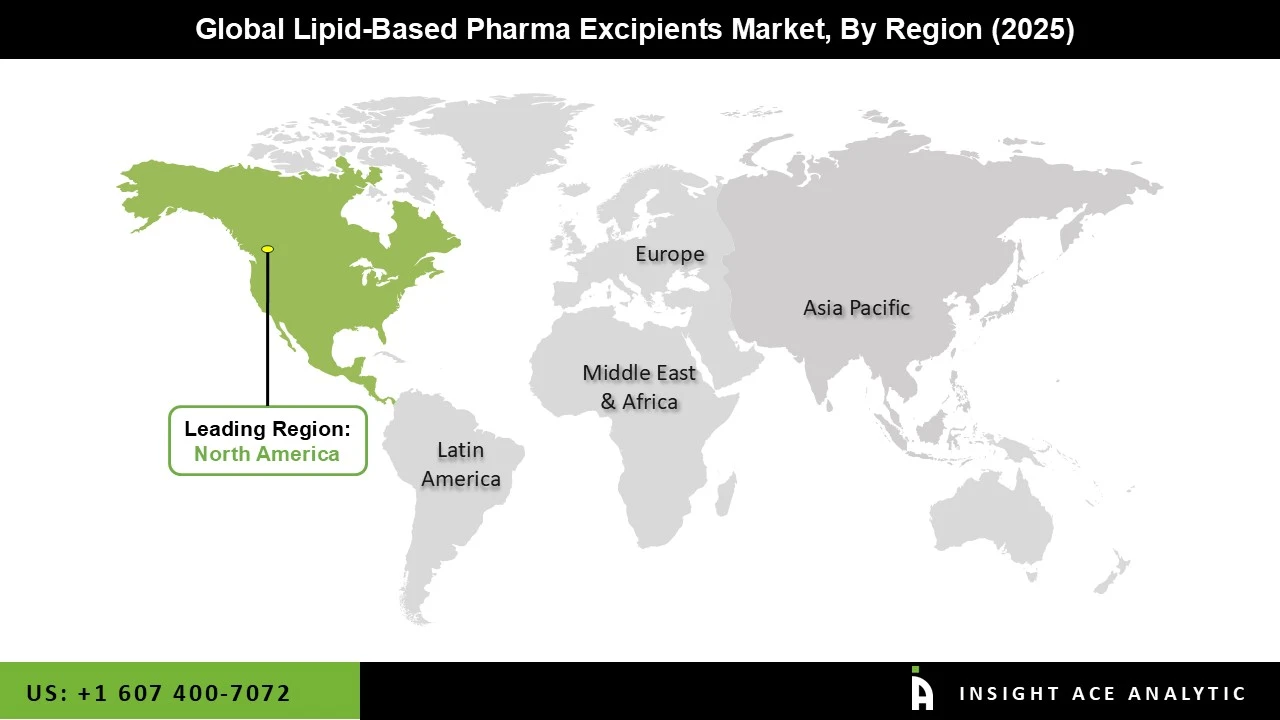
Why North America Led the Lipid-based Pharma Excipients Market?
The Lipid-based Pharma Excipients market was dominated by North America region in 2025. North America has a robust market environment that is well-represented in both huge industrial heavyweights and innovative entrepreneurs. For instance, Evonik formally started building its USD 220 million worldwide pharmaceutical specialty lipid production facility in Lafayette, Indiana, in April 2023. The region's sophisticated pharmaceutical manufacturing infrastructure, robust R&D base, and top international drug manufacturers have fueled the lipid-based pharma excipients market. Additionally, the market is being promoted by the growing tendency towards more individualized treatments, which is impacting the demand for customized excipient solutions. Because of the growing frequency of chronic diseases and the use of personalized medicine, the United States is dominating the North American lipid-based pharma excipients market over the forecast period.
Lipid-based Pharma Excipients Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 1.03 Bn |
| Revenue forecast in 2035 | USD 1.96 Bn |
| Growth Rate CAGR | CAGR of 7.0% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Formulation, Functionality, End-user, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | ABF Ingredients – ABITEC, Gattefosé, AAK, CREMER OLEO, IOI Oleo GMbH, Merck KGaA, BASF, Croda International, Alsiano, Evonik, Kao Corporation, Biopharma PEG Scientific, CD Formulation, CordenPharma, Lipoid, NOF CORPORATION, CLARIANT, and Colorcon |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Lipid-based Pharma Excipients Market by Product Type-
• Fats
• Oils
• Phospholipids
• Waxes
• Others
Lipid-based Pharma Excipients Market by Formulation-
• Injectables
• Enteral
• Topical
Lipid-based Pharma Excipients Market by Functionality-
• Solubilizers & Wetting Agents
• Penetration Enhancers & Solvents
• Structuring Agents & Lubricants
• Emollients
• Emulsifiers & Surfactants
• Others
Lipid-based Pharma Excipients Market by End-user-
• Pharma & Biotech Companies
• Contract Research Organizations (CROs) & Contract Development and Manufacturing Organizations (CDMOs)
• Contract Manufacturing Organizations (CMOs)
• Academic & Research Institutes
Lipid-based Pharma Excipients Market By Region-
North America-
• The US
• Canada
Europe-
• Germany
• The UK
• France
• Italy
• Spain
• Rest of Europe
Asia-Pacific-
• China
• Japan
• India
• South Korea
• South East Asia
• Rest of Asia Pacific
Latin America-
• Brazil
• Argentina
• Mexico
• Rest of Latin America
Middle East & Africa-
• GCC Countries
• South Africa
• Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Lipid based Pharma Excipients Market Size is valued at USD 1.03 Bn in 2025 and is predicted to reach USD 1.96 Bn by the year 2035
Lipid based Pharma Excipients Market is expected to grow at a 7.0% CAGR during the forecast period for 2026 to 2035.
ABF Ingredients – ABITEC, Gattefosé, AAK, CREMER OLEO, IOI Oleo GMbH, Merck KGaA, BASF, Croda International, Alsiano, Evonik, Kao Corporation, Biopharma PEG Scientific, CD Formulation, CordenPharma, Lipoid, NOF CORPORATION, CLARIANT, and Colorcon
Lipid based Pharma Excipients Market is segmented into Product Type, Formulation, Functionality, End-user, and By Region
North America region is leading the Lipid based Pharma Excipients Market