GMP Grade IVT Enzymes for Therapeutic RNA Market Size, Scope, Forecast Report 2026 to 2035
What is GMP-Grade IVT Enzymes for Therapeutic RNA Market Size?
Global GMP-Grade IVT Enzymes for Therapeutic RNA Market Size is valued at USD 394.84 Mn in 2025 and is predicted to reach USD 1,059.53 Mn by the year 2035 at a 10.5% CAGR during the forecast period for 2026 to 2035.
GMP-Grade IVT Enzymes for Therapeutic RNA Market, Share & Trends Analysis Report, By Enzyme Type (IVT Workflow) (RNA Polymerases, Capping Enzymes, Tailing Enzymes - Poly(A) Polymerase, Template Generation Enzymes, Cleanup / Yield / Protection, Circular RNA Enzymes), By RNA Modality (mRNA, saRNA/replicons, circRNA, gRNAs/crRNAs (CRISPR), Other therapeutic RNAs), By Application / Use Stage (GMP Drug Substance Manufacturing, GMP Clinical Supply & Process Development (Phase I–III), Diagnostics/IVD, Translational Research with GMP continuity), By End User, By Region, and Segment Forecasts, 2026 to 2035
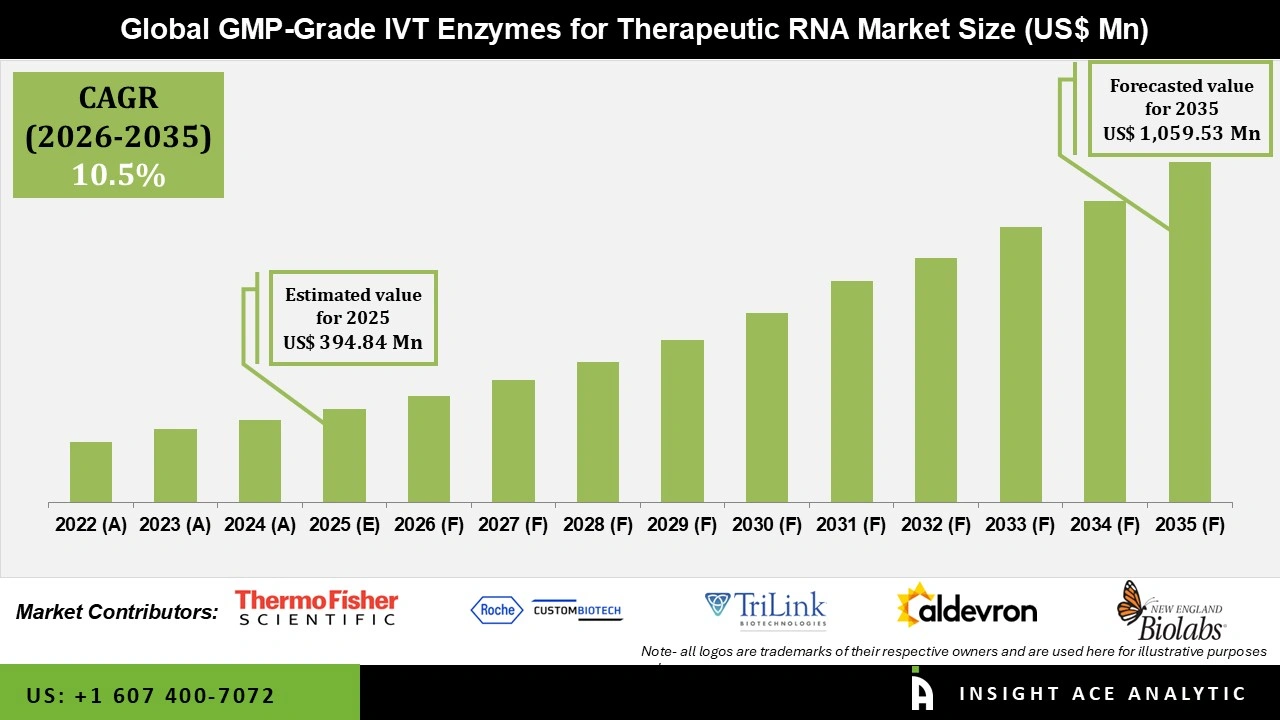
GMP-Grade IVT Enzymes for Therapeutic RNA Market Key Takeaways:
|
GMP-grade IVT (In Vitro Transcription) enzymes are essential for producing therapeutic RNA including mRNA used in vaccines, Gene therapies, and other RNA-based treatments. Manufactured under strict Good Manufacturing Practice (GMP) standards, these high-purity enzymes ensure consistent, high-quality RNA synthesis suitable for research, preclinical, and clinical applications.
RNA is transcribed from a DNA template containing a T7 promoter, enabling high-yield production commonly used in large-scale manufacturing of mRNA vaccines (such as COVID-19 vaccines), gene-editing tools like CRISPR, and protein replacement therapies. Key GMP-grade IVT enzymes include T7 RNA polymerase, poly(A) polymerase, and capping enzymes such as Vaccinia Capping Enzyme, all of which contribute to generating stable, translation-ready mRNA. Beyond vaccines, these enzymes also support regenerative medicine by enabling the creation of mRNA that drives tissue repair and cellular reprogramming.
Enzymes that enable high-yield, scalable IVT (In Vitro Transcription) processes for producing mRNA at gram-to-kilogram scales are vital to the GMP drug substance manufacturing (commercial) segment. This demand was clearly demonstrated during the COVID-19 pandemic, when billions of vaccine doses required large-scale mRNA synthesis. Among these, GMP-grade T7 RNA polymerase plays a central role engineered for superior activity and stability, it allows manufacturers to consistently meet industrial-scale requirements. Expanded GMP production infrastructure and the growing capabilities of contract manufacturing organizations (CMOs) have further increased the availability of these RNA polymerases, driving the expansion of commercial mRNA production. As the mRNA therapeutics market continues to grow, demand for GMP-grade IVT enzymes rises correspondingly, with RNA polymerases maintaining the largest market share due to their pivotal role in transcription. Continuous advancements in enzyme engineering are now enhancing yield, purity, and cost efficiency, solidifying their importance in large-scale mRNA drug substance manufacturing.
Competitive Landscape
Some of the Major Key Players in the GMP-Grade IVT Enzymes for Therapeutic RNA Market is:
· New England Biolabs (NEB)
· Thermo Fisher Scientific
· Roche CustomBiotech
· Merck KGaA (MilliporeSigma)
· Aldevron (Danaher/Cytiva)
· TriLink BioTechnologies (Maravai)
· Kactus Bio
· Yeasen Biotech
· Takara Bio
· Canvax Biotech
· LGC Biosearch Technologies
· Novoprotein
· Jena Bioscience
· Baseclick GmbH
· Tinzyme
· Promega Corporation
· Kaneka Eurogentec
· BOC Sciences
· Creative Biogene
· HONGENE
Market Segmentation
The GMP-grade IVT enzymes for the therapeutic RNA market are segmented into enzyme type (IVT workflow), RNA modality, application/use stage, and end user. Based on the enzyme type, the market is segmented into RNA polymerases, capping enzymes, tailing enzymes - poly(A) polymerase, template generation enzymes, cleanup/yield/protection, and circular RNA enzymes. Based on the RNA modality, the market is divided into mRNA, saRNA/replicons, circRNA, gRNAs/crRNAs (CRISPR), and other therapeutic RNAs. Based on the application/use stage, the market is divided into GMP drug substance manufacturing, GMP clinical supply & process development (phase I–III), diagnostics/IVD, and translational research with GMP continuity. Based on the end-user, the market is divided into biopharma/biotech sponsors (in-house manufacturing), CDMOs/CMOs, diagnostics companies, and academic/government translational centers.
The mRNA Segment is Expected to Have the Highest Growth Rate During the Forecast Period
mRNA is being widely explored for applications such as protein replacement therapies and cancer immunotherapies, including personalized neoantigen vaccines. To ensure safety, purity, and consistency, all mRNA Therapeutics intended for human use must be produced under rigorous Good Manufacturing Practice (GMP) standards, following guidelines like ICH Q7 and ISO 13485. GMP-grade IVT enzymes such as T7 RNA polymerase, RNase inhibitors, and capping enzymes serve as critical raw materials that meet these regulatory requirements. As more mRNA candidates advance into clinical development, the demand for GMP-grade enzymes has surged; however, limited availability of high-quality raw materials and the high cost of key enzymes like T7 RNA polymerase and DNase I have created supply challenges. The inherent flexibility of the mRNA platform allowing rapid sequence modifications without altering the core production process makes it ideal for fast-response vaccines and personalized therapies, further amplifying the demand for GMP-grade IVT enzymes to ensure reliable, high-quality mRNA synthesis across diverse therapeutic areas.
The RNA Polymerases Segment Dominates the Market
RNA polymerases are key enzymes in the synthesis of mRNA used for a range of applications, including CRISPR/Cas9 systems that encode Cas proteins and protein replacement therapies targeting conditions such as hemophilia and cystic fibrosis. The expanding pipeline of RNA-based therapeutics is driving strong demand for high-quality, GMP-grade RNA polymerases to ensure a consistent supply for both clinical and commercial-scale production. Within the therapeutic RNA ecosystem, the RNA polymerase segment plays a crucial role in overcoming supply chain bottlenecks by providing reliable, high-purity enzymes. Ongoing advances in RNA polymerase engineering aimed at improving transcription efficiency and minimizing unwanted double-stranded RNA byproducts are further strengthening supplier competitiveness. Meanwhile, emerging technologies such as microfluidics and continuous IVT systems are enhancing the scalability and productivity of RNA synthesis, fueling the growing demand for GMP-grade RNA polymerases to support next-generation RNA therapies.
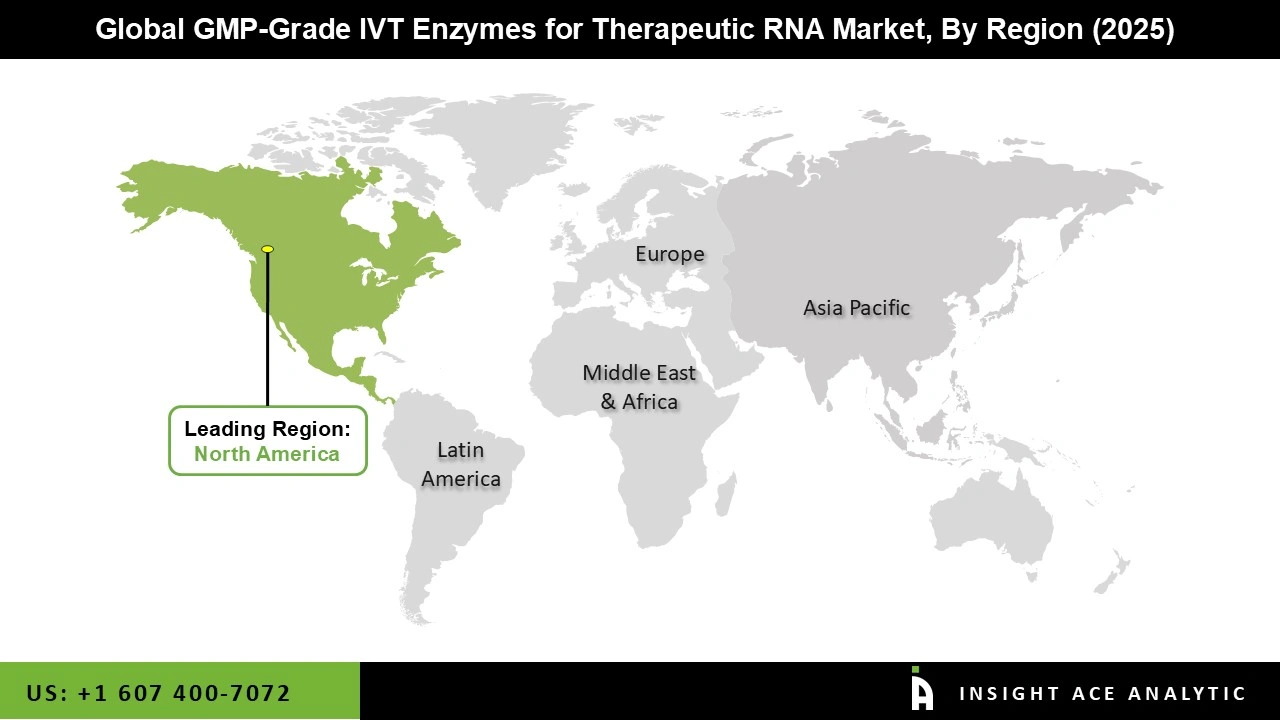
North America Has the Largest Market Share During the Forecast Period.
North America dominates the market for GMP-grade IVT enzymes for therapeutic RNA due to significant investments from private investors and pharmaceutical companies that are speeding up the development of mRNA technology, as well as significant financing from government agencies like the NIH and BARDA. This financial assistance directly supports the need for high-quality GMP-grade enzymes essential for both clinical and commercial-scale RNA synthesis.
In May 2024, U.S.-based Aldevron joined forces with Acuitas Therapeutics to enhance mRNA services through lipid nanoparticle (LNP) encapsulation, further solidifying this leadership. The partnership combines state-of-the-art LNP formulation with GMP-grade RNA polymerases, such as Aldevron's Codex® HiCap T7 RNA Polymerase, to create an integrated solution that expedites the manufacturing of mRNA therapeutics and further solidifies North America's leadership in providing end-to-end mRNA platforms.
Recent Developments:
· In April 2024, TriLink BioTechnologies (TriLink), a Maravai LifeSciences company declared its new cGMP mRNA production facility's grand opening. Using TriLink's strong mRNA production capabilities, the 32,000-square-foot facility was built especially for mRNA manufacture to assist late-phase drug researchers from Phase 2 to commercialization. As developers swarm to capitalize on the promising technique for an expanding range of applications, the milestone opening is anticipated to contribute to the advancement of mRNA-based therapy. The facility, which is situated in San Diego's Sorrento Valley, has separate Grade C cleanroom suites for the production of mRNA, a capacity increase from 1g to >100g per batch, extensive in-house analytical capabilities, and laboratory space for on-site quality control testing.
· In Mar 2023, Creative Biogene was committed to improving the newest medical technologies, such as vaccines, gene editing, cell treatments, and immunotechnology. In order to support research in the areas of preclinical drug discovery, industrial synthetic application, biomedical development, and fundamental life sciences research, Creative Biogene delivers knowledge to deliver products of the highest quality consistently on time. To further research and project development across multiple domains, Creative Biogene has announced the launch of its GMP-grade mRNA synthesis services.
GMP-Grade IVT Enzymes for Therapeutic RNA Market Report Scope:
| Report Attribute | Specifications |
| Market Size Value In 2025 | USD 394.84 Mn |
| Revenue Forecast In 2035 | USD 1,059.53 Mn |
| Growth Rate CAGR | CAGR of 10.5% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Mn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Enzyme Type (IVT Workflow), RNA Modality, Application / Use Stage, End User |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; South East Asia |
| Competitive Landscape | New England Biolabs (NEB), Thermo Fisher Scientific, Roche CustomBiotech, Merck KGaA (MilliporeSigma), Aldevron (Danaher/Cytiva), TriLink BioTechnologies (Maravai), Kactus Bio, Yeasen Biotech, Takara Bio, Canvax Biotech, LGC Biosearch Technologies, Novoprotein, Jena Bioscience, Baseclick GmbH, Tinzyme, Promega Corporation, Kaneka Eurogentec, BOC Sciences, Creative Biogene, HONGENE |
| Customization Scope | Free customization report with the procurement of the report and modifications to the regional and segment scope. Particular Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of the GMP-Grade IVT Enzymes for Therapeutic RNA Market :
GMP-Grade IVT Enzymes for Therapeutic RNA Market, By Enzyme Type (IVT Workflow)-
· RNA Polymerases
o T7 RNA Polymerase (dominant)
o SP6 RNA Polymerase
o T3 RNA Polymerase
· Capping Enzymes
o Vaccinia Capping Enzyme (Cap-0)
o mRNA Cap 2′-O-Methyltransferase (Cap-1)
o Alternative Viral Capping Enzymes (Faustovirus,etc)
· Tailing Enzymes - Poly(A) Polymerase
· Template Generation Enzymes
o Restriction Endonucleases
o High-fidelity DNA Polymerases
· Cleanup / Yield / Protection
o DNase I / dsDNase (RNase-free)
o Inorganic pyrophosphatase
o RNase inhibitor
· Circular RNA Enzymes
o T4 RNA Ligase I/II
o RNase R
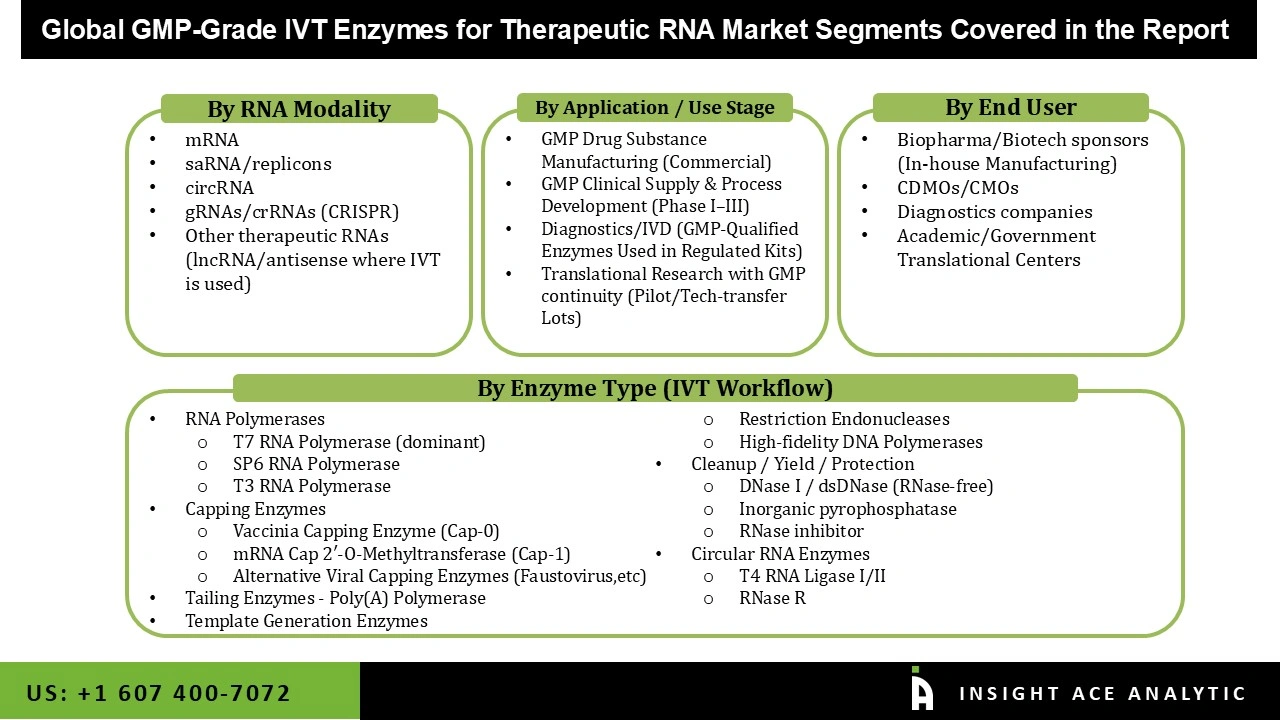
GMP-Grade IVT Enzymes for Therapeutic RNA Market, By RNA Modality-
· mRNA
· saRNA/replicons
· circRNA
· gRNAs/crRNAs (CRISPR)
· Other therapeutic RNAs (lncRNA/antisense where IVT is used)
GMP-Grade IVT Enzymes for Therapeutic RNA Market, By Application / Use Stage-
· GMP Drug Substance Manufacturing (Commercial)
· GMP Clinical Supply & Process Development (Phase I–III)
· Diagnostics/IVD (GMP-Qualified Enzymes Used in Regulated Kits)
· Translational Research with GMP continuity (Pilot/Tech-transfer Lots)
GMP-Grade IVT Enzymes for Therapeutic RNA Market, By End User-
· Biopharma/Biotech sponsors (In-house Manufacturing)
· CDMOs/CMOs
· Diagnostics companies
· Academic/Government Translational Centers
GMP-Grade IVT Enzymes for Therapeutic RNA Market, By Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East and Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
GMP-Grade IVT Enzymes for Therapeutic RNA Market Size is valued at USD 394.84 Mn in 2025 and is predicted to reach USD 1,059.53 Mn by the year 2035
GMP-Grade IVT Enzymes for Therapeutic RNA Market is expected to grow at a 10.5% CAGR during the forecast period for 2026 to 2035
New England Biolabs (NEB), Thermo Fisher Scientific, Roche CustomBiotech, Merck KGaA (MilliporeSigma), Aldevron (Danaher/Cytiva), TriLink BioTechnologies (Maravai), Kactus Bio, Yeasen Biotech, Takara Bio, Canvax Biotech, LGC Biosearch Technologies, Novoprotein, Jena Bioscience, Baseclick GmbH, Tinzyme, Promega Corporation, Kaneka Eurogentec, BOC Sciences, Creative Biogene, HONGENE and Others
The GMP-grade IVT enzymes for the therapeutic RNA market are segmented into enzyme type (IVT workflow), RNA modality, application/use stage, and end user.
North America region is leading the GMP-Grade IVT Enzymes for Therapeutic RNA Market.