Automated and Closed Cell Therapy Processing Systems Market Size and Revenue Impact Study 2026 to 2035
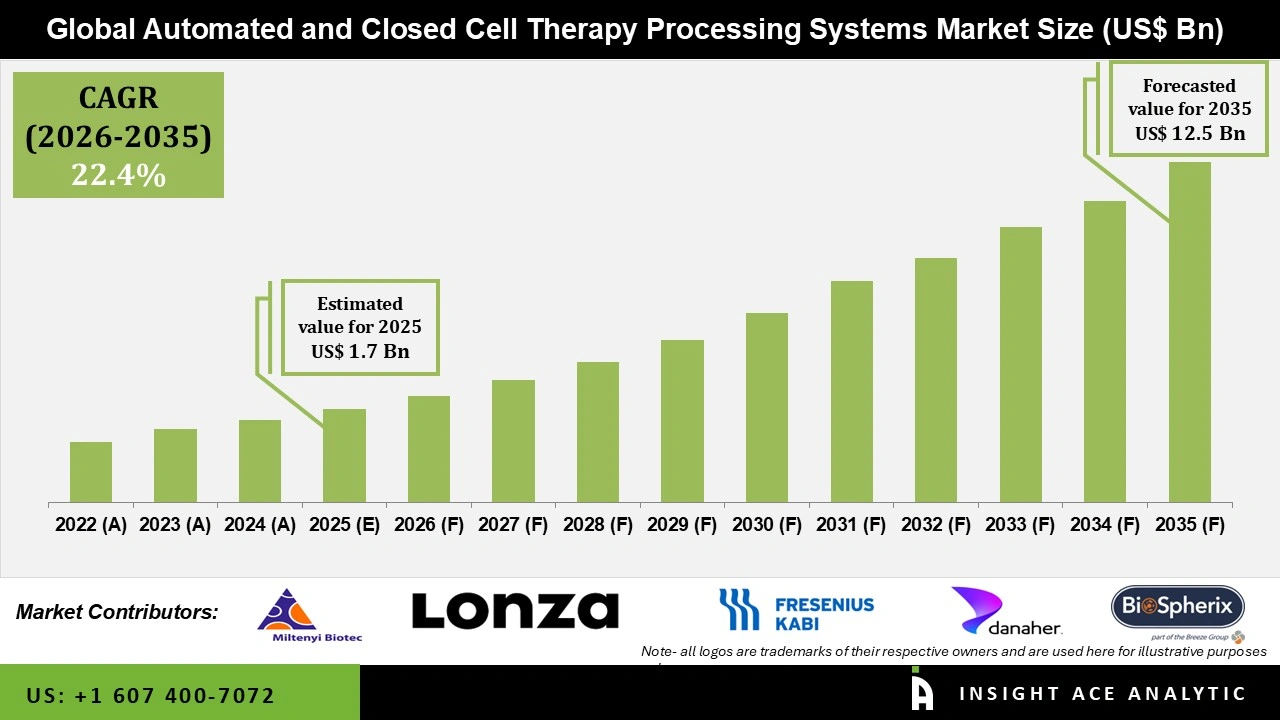
Automated and Closed Cell Therapy Processing Systems Market Size is valued at USD 1.7 Bn in 2025 and is predicted to reach USD 12.5 Bn by the year 2035 at a 22.4% CAGR during the forecast period for 2026 to 2035.
Automated and Closed Cell Therapy Processing Systems Market Size, Share & Trends Analysis Distribution By Type (Stem Cell Therapy and Non Stem Cell Therapy), By Workflow (Separation, Cryopreservation, Expansion, Fill- Finish, Apheresis, and Others), By Scale (Pre-Commercial/ R&D Scale and Commercial Scale), and Segment Forecasts, 2026 to 2035
The goal of an automated and closed cell therapy processing system is to optimize the handling, preparation, and assessment of cellular therapies in a secure environment. This procedure enhances the effectiveness and uniformity of the manufacturing process for cell-based treatments while lowering the possibility of contamination. In the production of cell therapies, the automated and closed cell therapy processing system is essential for maximizing both productivity and safety. The automation reduces the possibility of human error and upholds a high level of quality control, both of which are critical for guaranteeing patient safety. The growing requirement for scalable and contamination-free cell therapy manufacturing solutions, the expanding emphasis on process standardization, and the increasing demand for sophisticated cell-based therapies are the main factors driving the automated and closed cell therapy processing systems market's notable expansion.
The expanding focus on regenerative medicine and the increasing incidence of chronic diseases are important factors driving the automated and closed cell therapy processing systems market growth. These elements are hastening the deployment of automated technologies that guarantee scalable and reliable cell therapy production. Additionally, the market is seeing a rise in R&D spending, which promotes innovation and the launch of next-generation processing systems.
Furthermore, the market for automated and closed cell therapy processing systems is expanding rapidly due to rising demand for personalized medicine and technology improvements. The application of machine learning and artificial intelligence to cell therapy procedures is a significant trend that improves accuracy and productivity. The need to simplify complicated processes and lower human error, which will improve patient outcomes and save expenses, is what is driving this movement.
In addition, the expansion of clinical applications is essential for the automated and closed cell therapy processing systems market growth. The use of cell treatments has expanded beyond a small number of specialized uses. An increasing number of clinical disorders are being investigated for them.
The need for automated and closed cell therapy processing systems is rising along with the number of clinical studies and authorized treatments. These technologies make it possible to produce a variety of cell therapy products in an efficient and scalable manner, meeting a wide range of medical requirements. Additionally, the alliances and cooperation between healthcare providers, biotechnology businesses, and academic institutions are growing. These partnerships enable resource pooling, information exchange, and faster cell therapy technology development.
Competitive Landscape
Which are the Leading Players in Automated and Closed Cell Therapy Processing Systems Market?
• Thermo Fisher Scientific Inc.
• Miltenyi Biotec
• Lonza
• Fresenius Kabi AG
• Terumo Corporation
• Sartorius AG
• Danaher Corporation
• BioSpherix, LLC
• ThermoGenesis Holdings, Inc.
• CELLARES
Market Dynamics
Driver
Growing Demand for Cell Therapy Products
The automated and closed cell therapy processing systems market's primary driver of expansion is the rising need for cell therapy products. Cell treatment is becoming more and more popular as chronic illnesses become more common. The immune cells, stem cells, and CAR T-cells are examples of cell therapy products that are transplanted into patients to replace or repair damaged tissue or cells. These treatments can target any of the hundreds of genes in the body and can reduce or stop such illplasmanesses. Cell therapy development is aided by favorable government funding and growing investments in cell therapy research. The introduction of new products also boosts market expansion. Between 2023 and May 2024, the US Food and Drug Administration approved about ten cell and gene therapy products. Five of them are products used in cell treatment, such as CAR T-cells and stem cells.
Restrain/Challenge
Lack of Raw Material in Automated and Closed-Cell Therapy Processing Systems
Several steps in the overall cell therapy manufacturing process call for starting materials. Raw materials required for good medical practice (GMP) are in short supply due to the growing demand for automated and closed cell therapy processing systems worldwide. The automated and closed-cell therapy processing systems market is experiencing a bottleneck in the supply of plasmids and viral vectors, which are proven GMP-based raw materials needed in the cell therapy process, as a result of the quick rise in cell treatments and the rise in chronic diseases. Additionally, the market's expansion is hampered by the difficulties in acquiring raw materials, including bone marrow, blood, and apheresis. Therefore, the global automated and closed-cell therapy processing systems market is being constrained by the lack of raw materials.
Apheresis Segment is Expected to Drive the Automated and Closed Cell Therapy Processing Systems Market
The apheresis segment held the largest share in the automated and closed cell therapy processing systems market in 2025 because of the growing need for medical treatments that call for the separation of blood components like platelets and plasma. In the context of cell treatments, this procedure is essential because it enables the focused collection of particular cells while eliminating undesirable elements. Healthcare professionals are progressively implementing automated and more efficient apheresis systems as a result of technological improvements to enhance patient outcomes and optimize workflows. Apheresis is now the most popular workflow in this market because of this trend, an increase in chronic illnesses, and an increase in cancer patients in need of cellular therapies.
Pre-Commercial/ R&D Scale Segment is Growing at the Highest Rate in the Automated and Closed Cell Therapy Processing Systems Market
In 2025, the pre-commercial/ R&D scale segment dominated the automated and closed cell therapy processing systems market. This stage is critical for the creation and improvement of cell treatments, where exact control over the production process is vital. Early-stage clinical trials and research greatly benefit from automated and closed systems because they offer controlled settings that reduce human interaction, guaranteeing consistency and avoiding contamination. By preserving cell integrity and guaranteeing excellent results for proof-of-concept research, these methods aid in the improvement of cell treatments. The pre-commercial scale is leading the market due to the emphasis on innovation and research as well as the increasing demand for precision in the early phases of development. However, as therapies progress toward higher production levels, the commercial scale is anticipated to rise significantly.
Why North America Led the Automated and Closed Cell Therapy Processing Systems Market?
The automated and closed cell therapy processing systems market was dominated by North America region in 2025. This supremacy is ascribed to cutting-edge biotech and regulatory support that encourages creativity and makes it easier to develop novel treatments. Significant investment in cell therapy technologies is fueled by the region's strong healthcare system and emphasis on research and development. Notably, agencies like the U.S. Food and Drug Administration (FDA) have expedited approval procedures, accelerating the introduction of novel treatments into the market.
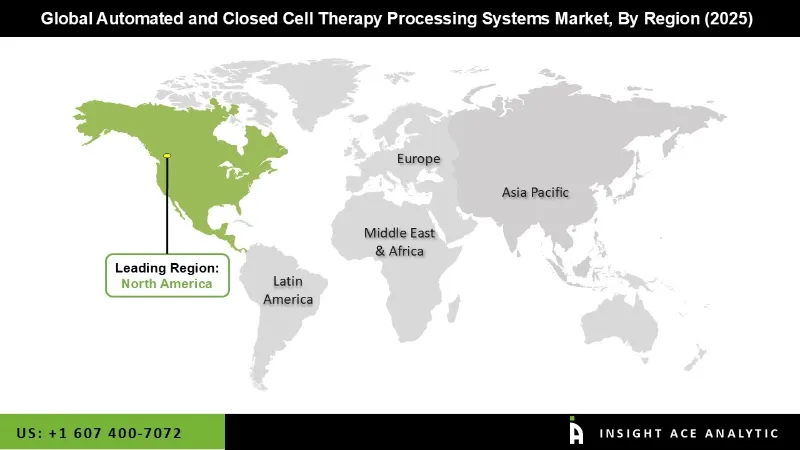
Additionally, the region's economic potential is further enhanced by the expanding emphasis on sustainable healthcare solutions and the growing consumer desire for individualized medication. The market for automated and closed cell therapy processing systems is dominated by the United States, which also serves as the sector's anchor in North America. Rapid developments in cell therapies are made possible by the nation's distinct regulatory environment, which is typified by the FDA's supportive regulations.
Key Development
September 2024: Targeting commercial CAR-T therapy production applications, Miltenyi Biotec introduced the CliniMACS Prodigy T-Pro system, an integrated platform for automated T-cell processing that improves cell recovery rates while reducing manufacturing time by 40%.
May 2024: Astellas Pharma Inc. and YASKAWA ELECTRIC CORPORATION partnered to develop a cutting-edge cell therapy platform that blends pharmaceutical and robotic technology. By streamlining the transition from research to large-scale production, our partnership aims to enhance the stability and quality of manufacturing processes.
April 2024: The Laboratory for Cell and Gene Medicine (LCGM) at Stanford Medicine and Multiply Labs established a collaboration to showcase automation technology's potential for cell therapy production.
January 2023: The acquisition of Xellbio, a business that specializes in automated cell processing technology, was announced by Sartorius AG. The goal of this calculated action is to improve Sartorius' capacity to provide all-inclusive solutions in the rapidly expanding cell and gene therapy markets.
Automated and Closed Cell Therapy Processing Systems Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 1.7 Bn |
| Revenue forecast in 2035 | USD 12.5 Bn |
| Growth Rate CAGR | CAGR of 22.4% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Type, Workflow, Scale, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Thermo Fisher Scientific Inc., Miltenyi Biotec, Lonza, Fresenius Kabi AG, Terumo Corporation, Sartorius AG, Danaher Corporation, BioSpherix, LLC, ThermoGenesis Holdings, Inc., and CELLARES. |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Market Segmentation:
Automated Closed Cell Therapy Processing Systems Market by Type
- Stem Cell Therapy
- Non Stem Cell Therapy
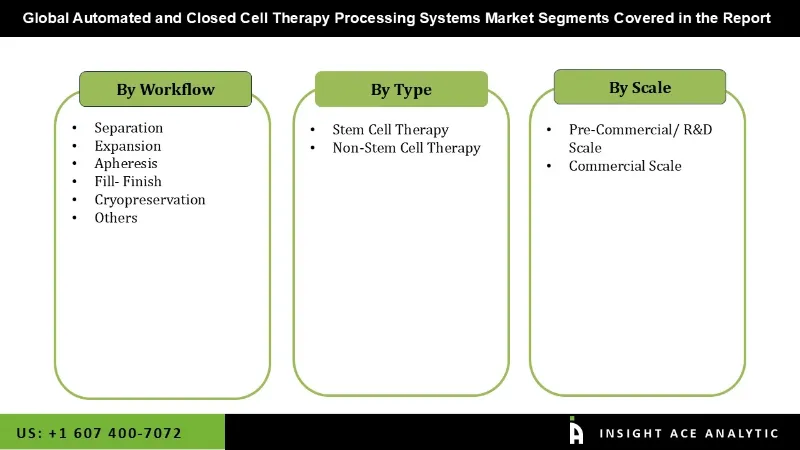
Automated and Closed Cell Therapy Processing Systems Market by Workflow
- Separation
- Fill- Finish
- Expansion
- Apheresis
- Cryopreservation
- Others
Automated and Closed Cell Therapy Processing Systems Market by Scale
- Pre-Commercial/ R&D Scale
- Commercial Scale
Automated and Closed Cell Therapy Processing Systems Market-By Region
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Spain
- Italy
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Automated and Closed Cell Therapy Processing Systems Market Size is valued at USD 1.7 Bn in 2025 and is predicted to reach USD 12.5 Bn by the year 2035.
Automated and Closed Cell Therapy Processing Systems Market is likely to grow at a 22.4% CAGR during the forecast period for 2026 to 2035.
Thermo Fisher Scientific Inc., Miltenyi Biotec, Lonza, Fresenius Kabi AG, Terumo Corporation, Sartorius AG, Danaher Corporation, BioSpherix, LLC, ThermoGenesis Holdings, Inc., and CELLARES.
Type, Workflow, Scale, and By Region are the key segments of the Automated and Closed Cell Therapy Processing Systems Market
North America region is leading the Automated and Closed Cell Therapy Processing Systems Market