2-way Foley Catheter Market Size, Share, Trend, Revenue Report 2026 to 2035
What is 2-way Foley Catheter Market Size?
Global 2-way Foley Catheter Market Size is valued at USD 995.40 Mn in 2025 and is predicted to reach USD 1,813.04 Mn by the year 2035 at a 6.2% CAGR during the forecast period for 2026 to 2035.
2-way Foley Catheter Market Size, Share & Trends Analysis Distribution by Usage (Short-Term Use and Long-Term Use), Material (Latex Foley Catheters and Silicone Foley Catheters), Indication (Urinary Incontinence, Spinal Cord Injury, Enlarged Prostate Gland/BPH, and Others), End-user (Hospitals, Long Term Care Facilities, and Others), and Segment Forecasts, 2026 to 2035

2-way Foley Catheter Market Key Takeaways:
|
A flexible, sterile medical tube called a 2-way Foley catheter is used to continuously drain urine from the bladder. It has two distinct channels, or lumens: one lumen lets urine flow from the bladder into a collecting bag, while the other lumen uses sterile water to inflate a tiny balloon close to the catheter tip. The balloon keeps the catheter firmly in place and stops it from falling out once it has been inflated inside the bladder. Several important factors, including the growing incidence of urological conditions such as bladder cancer and urine incontinence, and the growing demand for minimally invasive surgeries, are driving the 2-way Foley catheter market's expansion.
The growing incidence of urinary illnesses, including bladder obstruction, neurogenic bladder problems, and urine retention, is the main driver of the 2-way Foley catheter market. For patients who are unable to urinate on their own, these catheters are frequently used in hospitals, surgery centers, and long-term care homes to provide continuous urine drainage. Additionally, the need for 2-way Foley catheters during perioperative care is rising due to the increasing number of surgical operations, including urological, gynecological, and general surgeries. Another important aspect driving the 2-way Foley catheter market expansion is the aging population in many nations, since older people are more prone to chronic illnesses that necessitate catheterization and urine incontinence.
In addition, the growing use of 2-way Foley catheters in hospital and home-care settings is a result of rising healthcare costs and better access to healthcare in emerging nations. Furthermore, the growth of the 2-way Foley catheter market is anticipated to be impacted by advances in technology and improvements in catheter material. In order to lower the incidence of catheter-associated urinary tract infections (CAUTIs), which continue to be a major problem in long-term catheterization, manufacturers are concentrating on creating silicone-based and antimicrobial-coated catheters. Higher-quality and safer catheter products are being promoted by hospital infection control procedures and regulatory norms.
Competitive Landscape
Which are the Leading Players in 2-way Foley Catheter Market?
- Medtronic
- Sterimed Group
- Cardinal Health
- Advin Health Care
- AdvaCare Pharma
- B. Braun SE
- ANGIPLAST PRIVATE LIMITED
- Coloplast Corp
- Teleflex Incorporated
- Bactiguard AB
- HEMC (Hospital Equipment Manufacturing Company)
Market Dynamics
Driver
Growing Number of Elderly People and Rising Incidence of Urinary Problems
The rising incidence of urinary diseases and the expanding global elderly population are two of the main factors propelling the 2-way Foley catheter market. Urine disorders, including neurogenic bladder, bladder blockage, urine incontinence, benign prostatic hyperplasia (BPH), and retention, are becoming increasingly prevalent, especially in older people. People are more likely to experience these urological issues as they get older, which frequently necessitates either short-term or long-term catheterization for efficient bladder management. In these situations, a 2-way Foley catheter is frequently utilized because it permits continuous urine drainage while the balloon mechanism keeps the catheter firmly in place inside the bladder. Reliable urine drainage treatments are also becoming more and more necessary due to an increase in long-term care patients, surgical procedures, and hospital admissions. Moreover, the demand for 2-way Foley catheters is anticipated to increase gradually due to the growing number of older people worldwide and the rising prevalence of urological conditions, which is a major market driver.
Restrain/Challenge
Growing Burden of Regulatory Compliance and Risk of CAUTIs
Since extended catheterization can result in infections, longer hospital stays, and greater treatment costs, the danger of catheter-associated urinary tract infections continues to be a major obstacle limiting the 2-way Foley catheter market. Hospitals and gadget makers are under more pressure to comply with healthcare regulators' strict infection control guidelines and reporting obligations. Additionally, managing these dangers necessitates strict adherence to sterilizing guidelines, appropriate catheter insertion procedures, and ongoing staff training, which complicates operations for healthcare professionals. Furthermore, obtaining regulatory approval for novel 2-way Foley catheter materials and antimicrobial coatings can be expensive and time-consuming, which could postpone the release of new products. Moreover, manufacturers attempting to standardize 2-way Foley catheter products worldwide face difficulties due to regional variations in therapeutic recommendations. Inadequate clinical standards continue to limit optimal utilization and increase patient safety concerns due to concerns about prolonged or improper catheter use.
Silicone Foley Catheters Segment is Expected to Drive the 2-way Foley Catheter Market
The silicone foley catheters category held the largest share in the 2-way Foley Catheter market in 2025 driven by its special qualities that lower infection risks and improve patient comfort. Silver ions are pumped into these catheters to prevent microbial development and reduce the incidence of urinary tract infections (CAUTIs) associated with catheter use. The 2-way Foley catheters made of silicone are non-allergenic, perfect for people with sensitive skin, and their flexibility and smoothness make insertion simpler and less uncomfortable. Additionally, they have larger lumens for improved drainage and are more tissue-friendly, which makes them the preferred option for long-term usage and helps the 2-way Foley catheter market expand. Furthermore, they are more tissue-friendly and typically have larger lumens for better drainage, making them a popular choice for long-term usage and contributing to the segment's growth.
Hospitals Segment is Growing at the Highest Rate in the 2-way Foley Catheter Market
In 2025, the hospitals category dominated the 2-way Foley Catheter market because of its large patient volume and widespread use of catheters in critical care, surgery, and elder care. The 2-way Foley catheters are in high demand due to the frequency of urinary problems, especially among hospitalized patients with chronic illnesses or receiving postoperative care. The adoption of cutting-edge catheter designs with features that lower urinary tract infections (UTIs) is another priority for hospitals. Furthermore, the efficient deployment and monitoring of 2-way Foley catheters is ensured by a well-established healthcare infrastructure and the availability of qualified personnel in hospitals, which promotes their use and increases the market value within this category.
Why North America Led the 2-way Foley Catheter Market?
The 2-way Foley Catheter market was dominated by North America region in 2025. The high surgery volumes, sophisticated healthcare infrastructure, urological and maternity care specialists, and strategic partnerships such as HR Pharmaceuticals' commercialization of novel catheter designs that boost the 2-way Foley catheter market presence and enhance treatment efficacy are all factors contributing to the growth of the regional market. This need is supported by the region's advanced healthcare system, higher rates of surgery, and focus on comprehensive urology and maternity care.
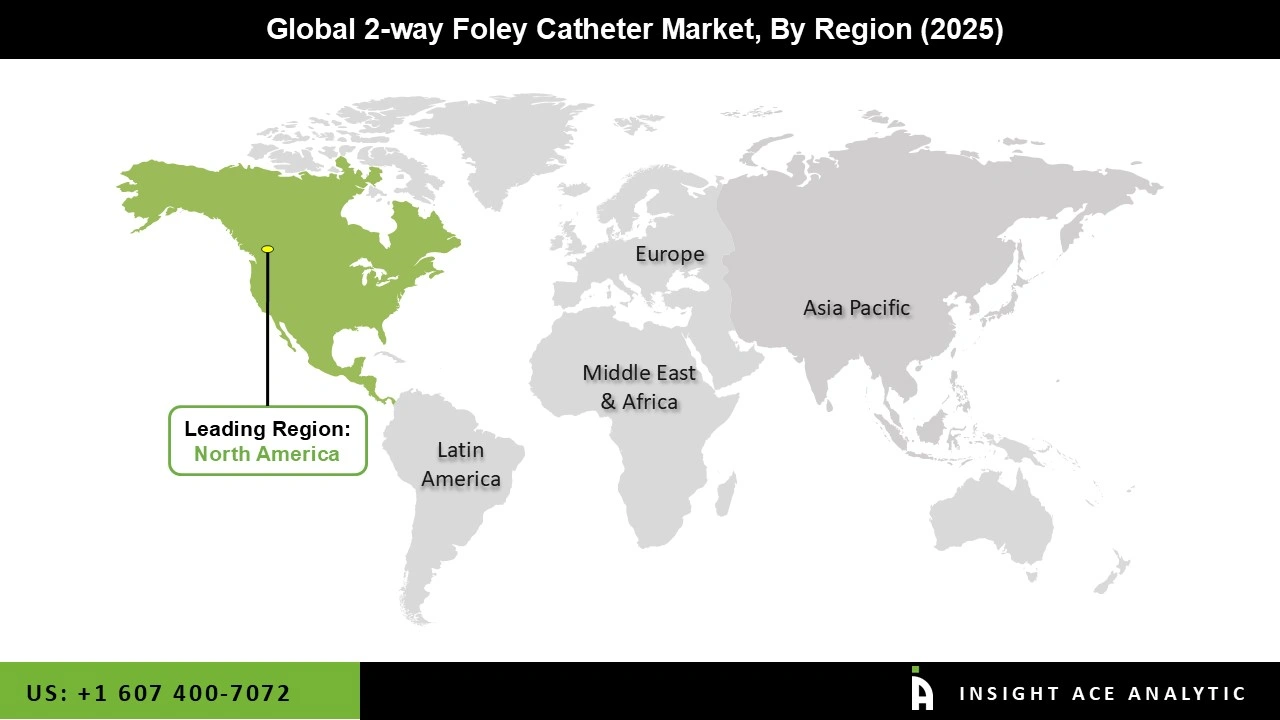
Additionally, North America's dominant position in the 2-way Foley catheter market has been reinforced by the region's high healthcare spending and advantageous reimbursement regulations. Furthermore, advancements in medical technology like the Duette Dual Balloon Catheter and an aging population with chronic illnesses that require long-term catheterization are driving the U.S. 2-way Foley catheter market.
Key Development
In May 2024, The Luja catheter for women has been introduced by Coloplast in four areas, including the US and Europe. This expansion followed the successful launch of a men's version that catered to the unique needs of female patients with urine incontinence in several different parts of the world. The launch's goal was to manage these problems while enhancing women's comfort and secrecy.
2-way Foley Catheter Market Report Scope :
| Report Attribute | Specifications |
| Market size value in 2025 | USD 995.40 Mn |
| Revenue forecast in 2035 | USD 1,813.04 Mn |
| Growth Rate CAGR | CAGR of 6.2% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Mn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026 to 2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Usage, Material, Indication, End-user, Distribution Channel, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Medtronic, Sterimed Group, Cardinal Health, Advin Health Care, AdvaCare Pharma, B. Braun SE, ANGIPLAST PRIVATE LIMITED, Coloplast Corp, Teleflex Incorporated, Bactiguard AB, and HEMC (Hospital Equipment Manufacturing Company) |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of 2-way Foley Catheter Market:
2-way Foley Catheter Market by Usage-
- Short-Term Use
- Long-Term Use
2-way Foley Catheter Market by Material-
- Latex Foley Catheters
- Silicone Foley Catheters
2-way Foley Catheter Market by Indication-
- Urinary Incontinence
- Spinal Cord Injury
- Enlarged Prostate Gland/BPH
- Others
2-way Foley Catheter Market by End-user-
- Hospitals
- Long Term Care Facilities
- Others
2-way Foley Catheter Market by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
2-way Foley Catheter Market Size is valued at USD 995.40 Mn in 2025 and is predicted to reach USD 1,813.04 Mn by the year 2035
The 2-way Foley Catheter Market is expected to grow at a 6.2% CAGR during the forecast period for 2026 to 2035
Medtronic, Sterimed Group, Cardinal Health, Advin Health Care, AdvaCare Pharma, B. Braun SE, ANGIPLAST PRIVATE LIMITED, Coloplast Corp, Teleflex Incorporated, Bactiguard AB, HEMC (Hospital Equipment Manufacturing Company) and others
2-way Foley Catheter Market is segmented into Usage, Material, Indication, End-user, Distribution Channel, Region and other.
North America region is leading the 2-way Foley Catheter Market.