Site Management Organizations (SMO) Market Size, Share, Trend Report 2026 to 2035
What is Site Management Organisations Market Size?
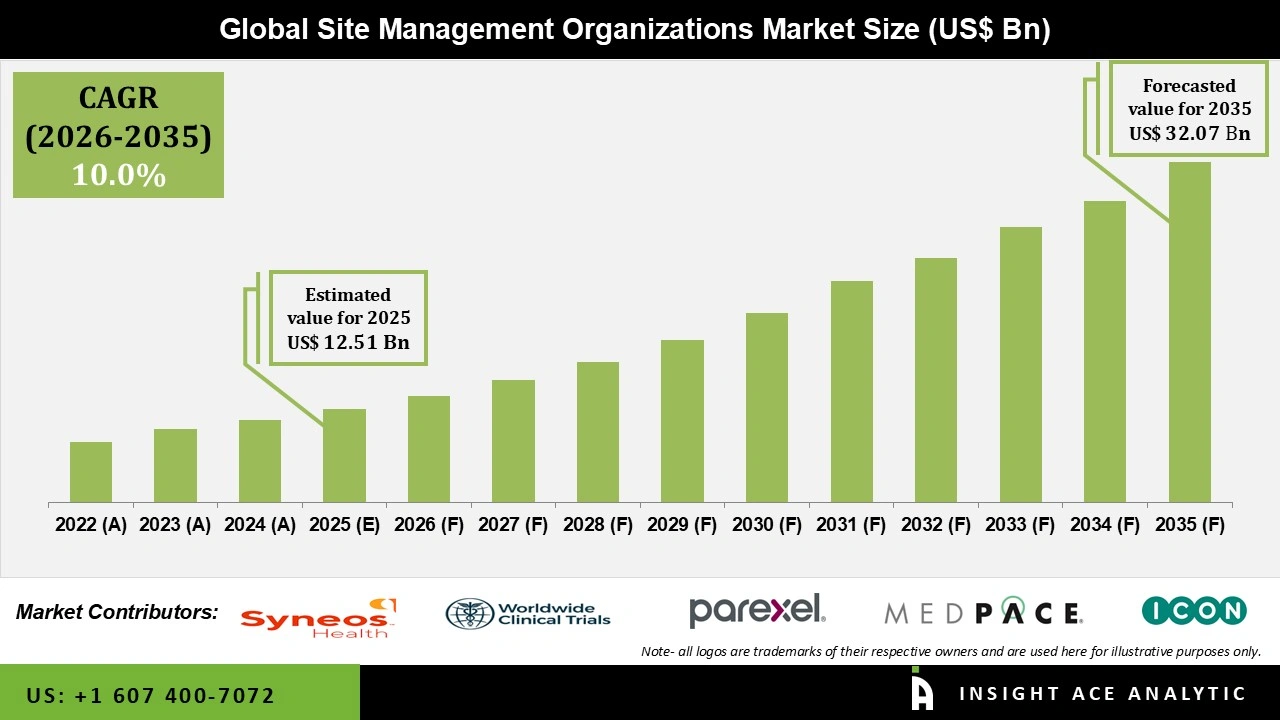
Global Site Management Organisations Market Size is valued at USD 12.51 Bn in 2025 and is predicted to reach USD 32.07 Bn by the year 2035 at a 10.0% CAGR during the forecast period for 2026 to 2035.
Site Management Organizations Market Size, Share & Trends Analysis Distribution By SMO Business Model (Full-Service SMOs, Specialty/Niche SMOs (Therapeutic-Area Focused), Virtual/Decentralized Clinical Trial (DCT) SMOs, Integrated Site Networks (ISNs), CRO-Integrated SMOs), By Service Component (Site Management & Coordination, On-Site Monitoring & Quality Oversight, Project Management & Operations, Patient Recruitment & Retention Services, Regulatory & Ethics Submission Support, Data Management & EDC Coordination, Other Ancillary Services), By Clinical Trial Phase (Phase I, Phase II, Phase III, Phase IV, Investigator-Initiated Trials (IITs)), By Therapeutic Area, By Intervention Type, By End-User / Sponsor Type, By Region and Segment Forecasts, 2026 to 2035
The site management organisations (SMO) has become an important part of the clinical trial ecosystem, mainly because of the growing need for better organisation and efficiency at the site level. As clinical trials expand across multiple countries and become more complex, the role of SMOs has shifted from simple coordination to active management and improvement of site performance. They help maintain consistency across trial sites by standardising processes, improving operational visibility, and ensuring compliance with all regulatory and protocol requirements. This becomes especially important in large, multi-centre trials where maintaining the same level of quality and timelines across all sites can be difficult. With clinical research becoming more global and focused on specialised treatments, SMOs are now seen as strategic partners rather than just support providers.
The growth of the SMO market is being supported by several ongoing changes in the clinical research environment. One of the biggest drivers is the increasing pressure on sponsors to complete trials faster and bring new treatments to market more quickly. At the same time, more clinical studies are being conducted in emerging regions, creating a need for local expertise and infrastructure something SMOs are well positioned to provide. The rise of smaller biotech companies is also contributing to market growth, as these organisations often rely on external partners to efficiently manage trial operations.
In addition, there is a stronger focus on data-driven decision-making in clinical trials, which requires accurate, consistent data collection across all sites. SMOs help meet this need by implementing structured systems and processes, ensuring better data quality and smoother study execution. Even as its importance grows, the SMO market faces several challenges. Managing multiple trial sites across different regions can be complex, especially when each country has its own regulatory requirements. This can sometimes lead to delays or inconsistencies if not handled carefully. Another challenge is the shortage of skilled professionals, as the demand for experienced clinical research staff continues to grow. Integrating diverse technologies and systems can also be difficult, particularly when sponsors, CROs, and sites use different platforms that do not easily connect with one another. In addition, keeping patients engaged throughout the trial remains a challenge, especially in longer or more complex studies. To overcome these issues, there is a need for ongoing investment in training, better technology, and improved processes to ensure consistent performance and long-term growth in the SMO market.
Competitive Landscape
Which are the Leading Players in the Site Management Organizations Market?
- Syneos Health
- Worldwide Clinical Trials
- Parexel International
- Medpace Inc.
- ICON plc
- WCG Clinical
- ClinChoice Inc.
- Flourish Research
- Velocity Clinical Research
- Elligo Health Research
- I'rom Group Co., Ltd.
- CMIC Group
- EPS Corporation
- A2 Healthcare
- Linical Co., Ltd.
- Bellsystem24
- MEDI-CO Plus
- Shin Nippon Biomedical Laboratories (SNBL)
- Tigermed
- Novotech / Acrostar SMO Division
- George Clinical
- Veeda Clinical Research
- JSS Medical Research Asia
- Beijing Aisimo Medical Science
- AusTrials
- ClinChoice
- Sannos
- CROMSOURCE
- Scandinavian CRO
- TFS HealthScience
- Trialbee
- Fidelis Research
- KCR S.A.
- Pharm-Olam
- Veristat
- FOMAT Medical Research
- WCCT Global
- ERG Holding
- Apex Medical Research
- Altasciences
- Synexus (PPD / Thermo Fisher)
- Accelerated Enrollment Solutions (AES)
- Javara Research
- Sarah Cannon Research Institute
- OneOncology
- Headlands Research
- Centricity Research
- MPR Development Group
- Neutra Life Sciences
- Clinedge
Market Dynamics
Driver
Growing Shift Toward Outsourcing Site Management Activities
The main drivers of the site management organisations (SMO) market is the increasing shift toward outsourcing clinical trial site operations. As clinical studies become more complex and time-sensitive, pharmaceutical and biotechnology companies are looking for ways to improve efficiency and reduce internal workload. SMOs help by taking responsibility for critical site-level activities, including patient recruitment, site setup, regulatory documentation, and daily coordination. This allows sponsors to focus more on research and development while ensuring that trials are managed smoothly at the ground level. The need for faster execution, especially in large, multi-country trials, is further encouraging companies to rely on specialised SMO partners who can deliver consistent, scalable support.
Restrain/Challenge
Ongoing Difficulties in Patient Recruitment and Retention
The major challenge in the site management organisations (SMO) market remains patient recruitment and retention. Finding the right patients who meet strict study criteria can take time, and delays in enrolment can slow the entire trial process. Even after recruitment, keeping patients engaged until the end of the study is not always easy, especially in long or demanding trials. Dropouts can impact data quality, increase costs, and extend timelines. While SMOs are adopting digital tools, outreach programs, and patient-friendly approaches to improve participation, maintaining consistent engagement remains a key hurdle that directly affects the success of clinical trials.
Full-Service SMOs Segment is Expected to Drive the Site Management Organisations Market
The full-service SMO segment is expected to play a major role in driving the SMO market's growth. These organisations offer end-to-end solutions, covering everything from site selection and activation to patient management and study close-out. Their ability to provide comprehensive support makes them highly attractive to sponsors conducting large-scale or multi-region clinical trials. Global and national full-service providers bring standardised processes, experienced teams, and established site networks, which help improve trial efficiency and data quality. As clinical trials become more complex and require seamless coordination across multiple locations, the demand for full-service SMOs continues to rise. Their scalability and ability to handle diverse therapeutic areas make them a preferred choice for sponsors.
Patient Recruitment & Retention Services Segment is Expected to Drive the Site Management Organizations Market
Patient recruitment and retention services are emerging as a key growth segment within the SMO market. Successful clinical trials depend heavily on enrolling the right patients and ensuring they remain engaged throughout the study. SMOs play a crucial role by leveraging advanced strategies, including digital outreach, patient databases, and collaborations with healthcare providers and advocacy groups. They also implement retention programs, including patient education, follow-ups, and support services, to improve compliance and reduce dropouts. With increasing competition for patient enrollment and stricter trial requirements, sponsors are relying more on specialised recruitment services. This makes patient recruitment and retention a critical and rapidly growing component of the SMO market.
Why North America Led the Site Management Organisations Market?
North America has established itself as the leading region in the site management organisations (SMO) market, mainly due to its strong and well-developed clinical research ecosystem. The region is home to many pharmaceutical and biotechnology companies that continually invest in research and development, resulting in a high volume of clinical trials. This naturally creates consistent demand for SMO services to manage and streamline site-level operations. Another important factor is the presence of a clear and structured regulatory environment, particularly in the United States.
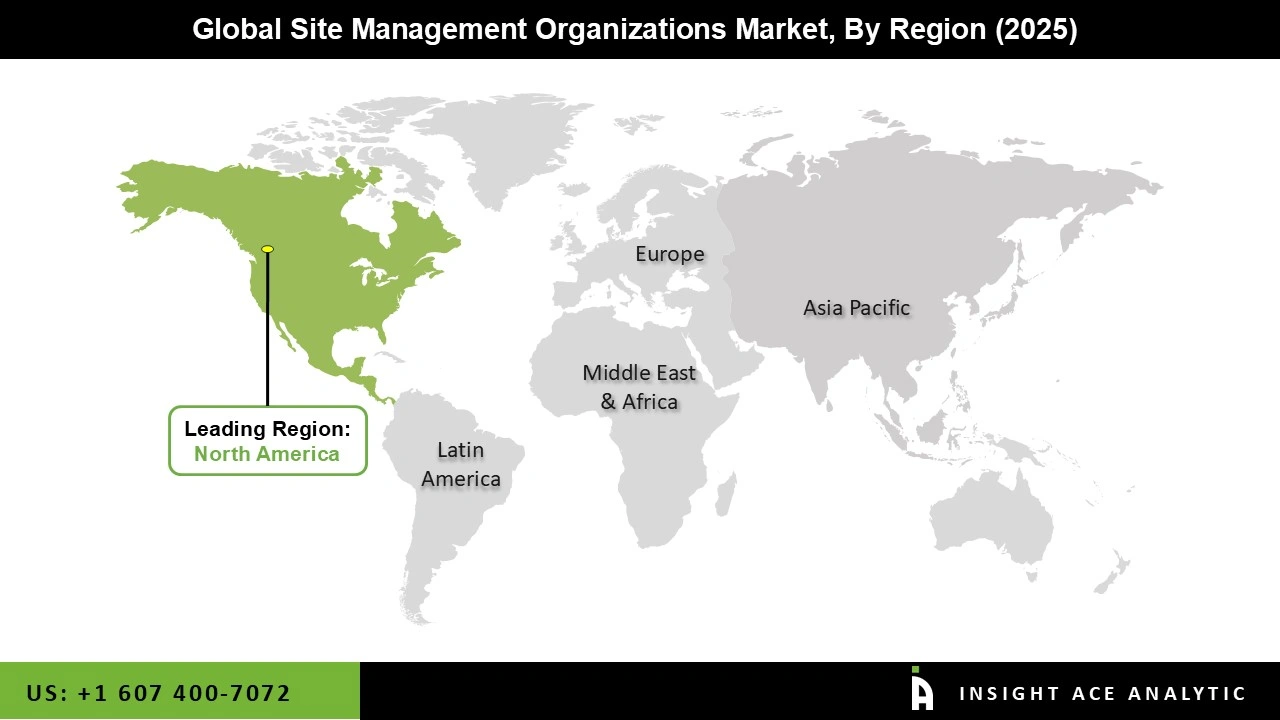
Regulatory bodies such as the U.S. Food and Drug Administration provide well-defined guidelines that help expedite approvals and streamline trial execution. This makes North America an attractive region for sponsors seeking to conduct reliable, high-quality clinical studies. In addition, the region benefits from advanced healthcare infrastructure, experienced investigators, and access to diverse patient populations. These factors improve patient recruitment, data quality, and overall trial efficiency. The growing adoption of decentralised and technology-driven clinical trials is also strengthening the role of SMOs, as companies increasingly rely on specialised partners to manage complex operations. Overall, North America’s leadership in the SMO market is driven by its strong industry presence, supportive regulations, technological advancement, and ability to efficiently manage large-scale clinical trials.
Key Developments
• In January 2025, ICON plc strengthened its global site network by integrating advanced digital tools to support hybrid and decentralised clinical trials.
• In August 2024, Novotech expanded its Pan-Asia SMO services through its Acrostar division, supporting biotech companies with regional clinical trial execution.
Site Management Organisations Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 12.51 Bn |
| Revenue forecast in 2035 | USD 32.07 Bn |
| Growth Rate CAGR | CAGR of 10.0% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2021 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | SMO Business Model, Service Component, Clinical Trial Phase, Therapeutic Area, Intervention Type, End User, and By Region |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Syneos Health, Worldwide Clinical Trials, Parexel International, Medpace Inc., ICON plc, WCG Clinical, ClinChoice Inc., Flourish Research, Velocity Clinical Research, Elligo Health Research, I'rom Group Co., Ltd., CMIC Group, EPS Corporation, A2 Healthcare, Linical Co., Ltd., Bellsystem24, MEDI-CO Plus, Shin Nippon Biomedical Laboratories (SNBL), Tigermed, Novotech / Acrostar SMO Division, George Clinical, Veeda Clinical Research, JSS Medical Research Asia, Beijing Aisimo Medical Science, AusTrials, ClinChoice, Sannos, CROMSOURCE, Scandinavian CRO, TFS HealthScience, Trialbee, Fidelis Research, KCR S.A., Pharm-Olam, Veristat, FOMAT Medical Research, WCCT Global, ERG Holding, Apex Medical Research, Altasciences, Synexus (PPD / Thermo Fisher), Accelerated Enrollment Solutions (AES), Javara Research, Sarah Cannon Research Institute, OneOncology, Headlands Research, Centricity Research, MPR Development Group, Neutra Life Sciences, Clinedge |
| Customization Scope | Free customisation report with the procurement of the report, Modifications to the regional and segment scope. Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customised to your particular study requirements. |
Segmentation of Site Management Organizations (SMO) Market
Site Management Organisations Market, by SMO Business Model-
- Full-Service SMOs
- Multi-region / global full-service providers
- National full-service providers
- Speciality / Niche SMOs (Therapeutic Area Focused)
- Oncology-specialized SMOs
- CNS / neuroscience-specialised SMOs
- Rare disease & orphan indication SMOs
- Cardiovascular & metabolic SMOs (including GLP-1 obesity programs)
- Other therapeutic speciality SMOs
- Virtual / Decentralised Clinical Trial (DCT) SMOs
- Home health & mobile nursing-enabled SMOs
- Fully virtual/siteless trial coordinators
- Integrated Site Networks (ISNs)
- Oncology-focused ISNs (e.g., Sarah Cannon, OneOncology)
- Primary-care embedded ISNs (e.g., Javara, Elligo, Headlands)
- CRO-Integrated SMOs
 Site Management Organisations Market, by Service Component-
Site Management Organisations Market, by Service Component-
- Site Management & Coordination
- Clinical research coordinator (CRC) deployment and supervision
- Site activation, initiation, and close-out management
- Investigator site file (ISF) and essential document management
- On-Site Monitoring & Quality Oversight
- Source data verification (SDV) support
- Protocol deviation tracking and remediation
- Risk-based monitoring (RBM) implementation at sites
- Project Management & Operations
- Multi-site study project coordination
- Sponsor / CRO liaison and reporting
- Timeline, milestone, and budget tracking
- Patient Recruitment & Retention Services
- Feasibility assessment and patient database mapping
- Digital recruitment (social media, patient advocacy partnerships)
- Patient retention programs and compliance support
- Regulatory & Ethics Submission Support
- IRB / IEC submission preparation and maintenance
- Country-level regulatory affairs (FDA, EMA, MHLW/PMDA, NMPA, CDSCO)
- Informed consent form (ICF) localisation and compliance
- Data Management & EDC Coordination
- Other Ancillary Services (Biorepository, Pharmacy, Imaging Coordination)
Site Management Organisations Market, by Clinical Trial Phase -
- Phase I (First-in-Human / Early Clinical Pharmacology)
- Phase II (Proof-of-Concept / Dose-Finding)
- Phase III (Pivotal / Registration)
- Phase IV (Post-Marketing / Real-World Evidence)
- Investigator-Initiated Trials (IITs) & Non-Industry-Sponsored Studies
Site Management Organisations Market, by Therapeutic Area-
- Oncology (Solid Tumours, Haematological Malignancies, Cell & Gene Therapy)
- Central Nervous System (CNS) & Neurology
- Cardiovascular & Metabolic Disorders
- Infectious Diseases & Vaccines
- Autoimmune & Inflammatory Disorders
- Rare & Orphan Diseases
- Ophthalmology
- Women's Health / Obstetrics & Gynecology
- Respiratory Disorders
- Others (Dermatology, Nephrology, Pediatrics, Musculoskeletal)
Site Management Organisations Market, by Intervention Type–
- Drug/Biologic Therapeutics Trials
- Small Molecule Drug Trials
- Biologic & Biosimilar Trials
- Cell & Gene Therapy Trials
- Medical Device Trials
- Diagnostics & Digital Therapeutics Trials
- Surgical Procedure Studies
- Behavioral & Observational Studies
Site Management Organisations Market, by End User / Sponsor Type–
- Large Pharmaceutical Companies
- Emerging Biotech & Mid-Size Pharma
- Contract Research Organisations (CROs)
- Medical Device Manufacturers
- Academic Medical Centers & Government/Public Sponsors
- Digital Health & Diagnostics Companies
Site Management Organizations Market, by Region-
- North America-
- The US
- Canada
- Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
- Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East and Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Global Site Management Organizations (SMO) Market is expected to grow at an 10.0% CAGR during the forecast period for 2026 to 2035
Syneos Health, Worldwide Clinical Trials, Parexel International, Medpace Inc., ICON plc, WCG Clinical, ClinChoice Inc., Flourish Research, Velocity Clinical Research, Elligo Health Research, I\'rom Group Co., Ltd., CMIC Group, EPS Corporation, A2 Healthcare, Linical Co., Ltd., Bellsystem24, MEDI-CO Plus, Shin Nippon Biomedical Laboratories (SNBL), Tigermed, Novotech / Acrostar SMO Division, George Clinical, Veeda Clinical Research, JSS Medical Research Asia, Beijing Aisimo Medical Science, AusTrials, ClinChoice, Sannos, CROMSOURCE, Scandinavian CRO, TFS HealthScience, Trialbee, Fidelis Research, KCR S.A., Pharm-Olam, Veristat, FOMAT Medical Research, WCCT Global, ERG Holding, Apex Medical Research, Altasciences, Synexus (PPD / Thermo Fisher), Accelerated Enrollment Solutions (AES), Javara Research, Sarah Cannon Research Institute, OneOncology, Headlands Research, Centricity Research, MPR Development Group, Neutra Life Sciences, Clinedge and Others.
Site Management Organizations (SMO) Market is segmented into SMO Business Model, Service Component, Clinical Trial Phase, Therapeutic Area, Intervention Type, End User and Other.
North American region is leading the Site Management Organizations (SMO) Market.
Site Management Organisations Market Size is valued at USD 12.51 Bn in 2025 and is predicted to reach USD 32.07 Bn by the year 2035