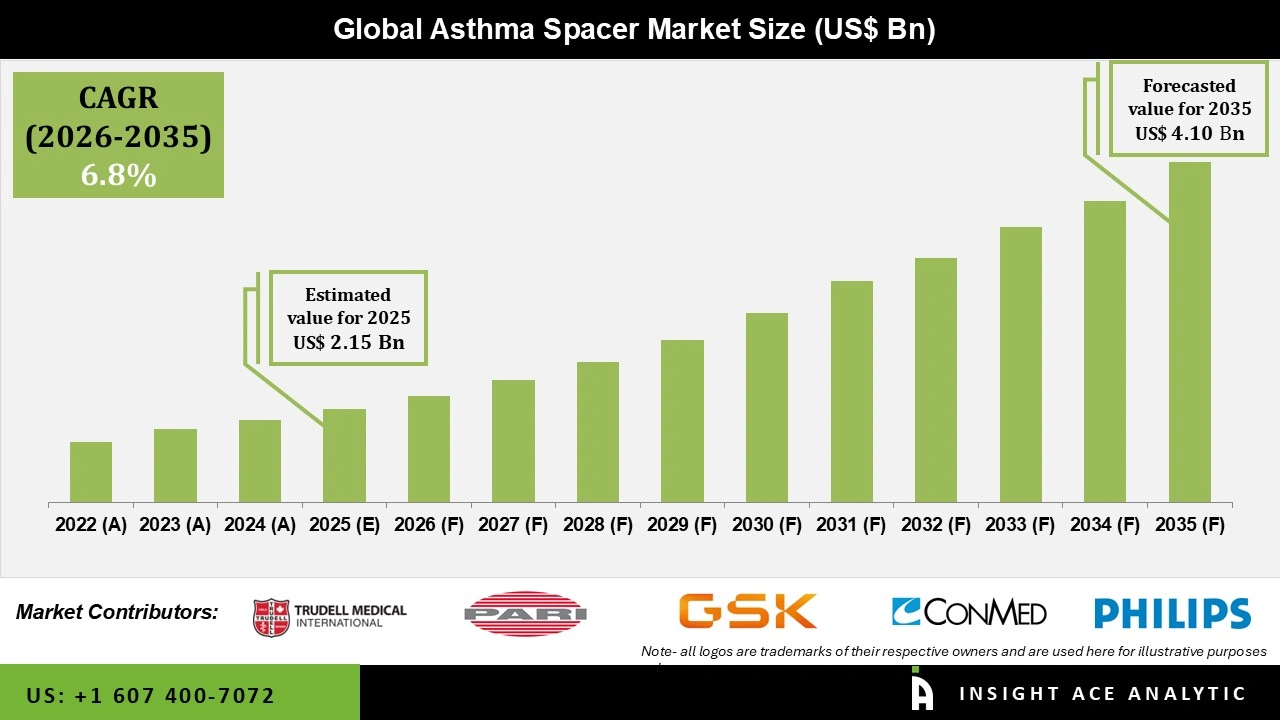
Global Asthma Spacers Market Size is valued at USD 2.15 Billion in 2025 and is predicted to reach USD 4.10 Billion by the year 2035 at a 6.8% CAGR during the forecast period for 2026 to 2035.
Asthma Spacers Market Size, Share & Trends Analysis Report By Brand (Space, Chamber Plus, A2A Spacer, Flo-Tone CR, Able Spacer, AeroChamber, Volumatic, Vortex, OptiChamber, Others), By Application, By Region, And Segment Forecasts, 2026 to 2035
Asthma Spacers Market Key Takeaways:
|
Spacers are a plastic tubes specifically designed to act as an interface between the pressurized metered-dose inhalers and patients. Spacers are also known as valved holding chanmber (VHC). The Spacers attaches to a pressurized metered-dose inhaler at one end and to the other end has mouthpiece to breathe through. The use of a spacer decreases the risk of drug underdose, minimizes oropharyngeal deposition and improves the amount of medication reaching to lungs. Spacers make pressurized metered dose inhalers (pMDI) comfortable to use by decreasing or eliminating the requirement for proper coordination between actuation and inhalation. The spacer helps proper delivery of drug by reducing the cold Freon effect. However, the benefits of spacers are offered at the cost of making pMDI system significantly large and less convenient. In general, spacers are categorized into Reverse-flow devices, Holding chambers, and Simple tube extensions to the actuator mouthpiece.
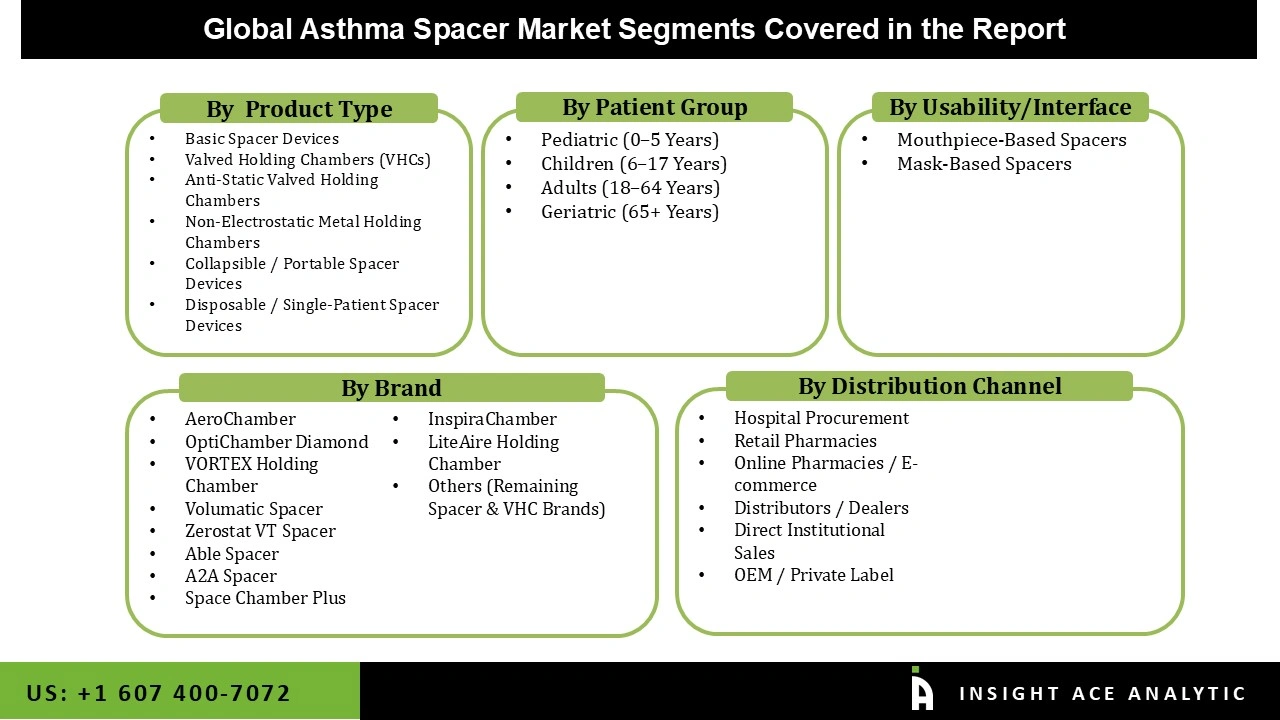
The asthma spacers market is segmented on the basis of product type, patient group, usability / interface, brand, and distribution channel. By product type, the market comprises Basic Spacer Devices, Valved Holding Chambers (VHCs), Anti-Static Valved Holding Chambers, Non-Electrostatic Metal Holding Chambers, Collapsible / Portable Spacer Devices, and Disposable / Single-Patient Spacer Devices. According to patient group, the market is categorized into Pediatric (0–5 Years), Children (6–17 Years), Adults (18–64 Years), and Geriatric (65+ Years). By usability / interface, the market includes Mouthpiece-Based Spacers and Mask-Based Spacers, catering to different patient needs and age-specific requirements. The brand segment consists of established names such as AeroChamber, OptiChamber Diamond, VORTEX Holding Chamber, Volumatic Spacer, Zerostat VT Spacer, Able Spacer, A2A Spacer, Space Chamber Plus, InspiraChamber, LiteAire Holding Chamber, along with other remaining spacer and VHC brands. By distribution channel, the market is segmented into Hospital Procurement, Retail Pharmacies, Online Pharmacies / E-commerce, Distributors / Dealers, Direct Institutional Sales, and OEM / Private Label, reflecting the diverse pathways through which these respiratory devices reach healthcare facilities and end users.
Valved Holding Chambers represent the most clinically preferred and commercially dominant product segment within the asthma spacers market, owing to their superior drug delivery efficiency compared to basic spacer devices. Unlike simple tube-style spacers, VHCs incorporate a one-way valve mechanism that holds the aerosolized medication in suspension momentarily, allowing patients to inhale at their own pace without requiring precise breath-actuation coordination — a critical advantage for patients with limited respiratory capacity or coordination difficulties.
This functional superiority has made VHCs the standard of care recommended by major respiratory health organizations including the Global Initiative for Asthma (GINA) and the American Academy of Allergy, Asthma and Immunology. Growing diagnosis rates of asthma globally, increasing physician preference for VHCs in both hospital and home-care settings, and the rising adoption of inhaled corticosteroid therapies are collectively reinforcing the segment's market leadership. Manufacturers are also investing in mask-equipped VHC variants compatible with multiple inhaler brands, further broadening the addressable patient population and sustaining segment dominance.
The pediatric segment spanning infants and toddlers aged zero to five years holds a disproportionately significant share of the asthma spacers market, reflecting the high prevalence of early-onset asthma and wheeze in this age group as well as the clinical necessity of spacer-assisted inhalation therapy in young children. Children in this cohort lack the pulmonary coordination required to operate pressurized metered-dose inhalers independently, making spacer devices with soft face masks an indispensable component of any inhaled therapy regimen.
Pediatric respiratory conditions including viral-induced wheeze, allergic asthma, and reactive airway disease are among the most common reasons for emergency department visits in children under five globally, generating consistent and high-volume demand for age-appropriate spacer devices. Heightened parental awareness of long-term asthma management, expanding newborn and infant healthcare infrastructure in emerging markets, and the introduction of lightweight, child-friendly spacer designs with minimal dead space are all contributing to the sustained prominence of this segment across both developed and developing healthcare systems.
| Report Attribute | Specifications |
| Market Size Value In 2025 | USD 2.15 Billion |
| Revenue Forecast In 2035 | USD 4.10 Billion |
| Growth Rate CAGR | CAGR of 6.8% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Billion and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | Product Type, Patient Group, Usability / Interface, Brand, Distribution Channel |
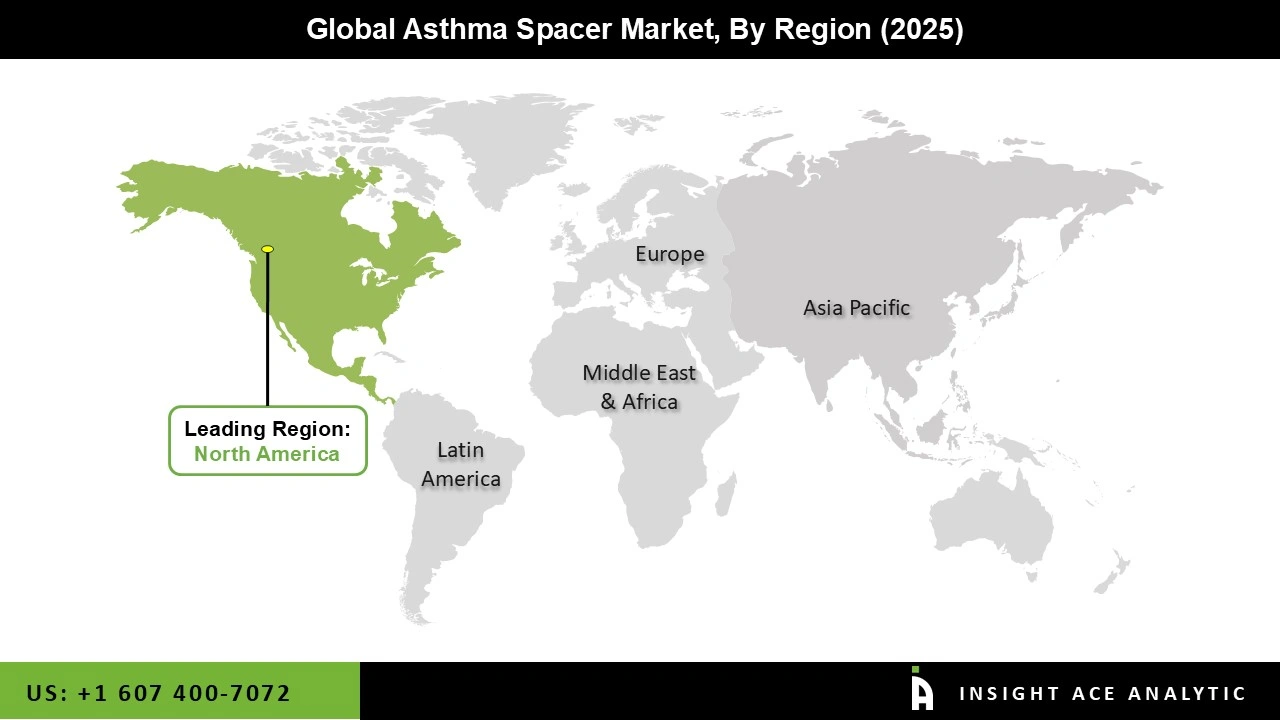
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico ; France; Italy; Spain; South Korea; South East Asia |
| Competitive Landscape | Trudell Medical International, Monaghan Medical Corporation, Koninklijke Philips N.V. (Philips Respironics), PARI Respiratory Equipment GmbH, GlaxoSmithKline plc, Cipla Limited, Lupin Limited, Clement Clarke International / Flexicare, Medical Developments International Ltd., Teleflex Incorporated, Rossmax International Ltd., Haag-Streit Group, Laboratoire ProtecSom, Air Liquide Healthcare, GaleMed Corporation, SunMed / Vyaire Medical, Armstrong Medical Ltd., DTF Medical, Visiomed Group, AGPMED and Others |
| Customization Scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Particular Geographic competitive landscape. |
| Pricing And Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
This study employed a multi-step, mixed-method research approach that integrates:
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Secondary data for the market study was gathered from multiple credible sources, including:
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Primary interviews for this study involved:
Interviews were conducted via:
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
This ensured that the dataset used for modelling was clean, robust, and reliable.
The bottom-up approach involved aggregating segment-level data, such as:
This method was primarily used when detailed micro-level market data were available.

The top-down approach used macro-level indicators:
This approach was used for segments where granular data were limited or inconsistent.
To ensure accuracy, a triangulated hybrid model was used. This included:
This multi-angle validation yielded the final market size.
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Given inherent uncertainties, three scenarios were constructed:
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.