Digital PCR and Real-Time PCR Market Size, Scope, Forecast Report 2026 to 2035
What is Digital PCR and Real-Time PCR Market Size?
Global Digital PCR and Real-Time PCR Market Size is valued at USD 9.61 billion in 2025 and is predicted to reach USD 20.5 billion by the year 2035 at an 8.0% CAGR during the forecast period for 2026 to 2035.
Digital PCR and Real-Time PCR Market Size, Share & Trends Analysis Report By Product (Instruments, Reagents & Consumables, Software & Services), Application (Clinical, Research, Forensic), End User (Hospital, Diagnostic Labs, Pharma, Biotech, CROs), By Region, And Segment Forecasts, 2026 to 2035
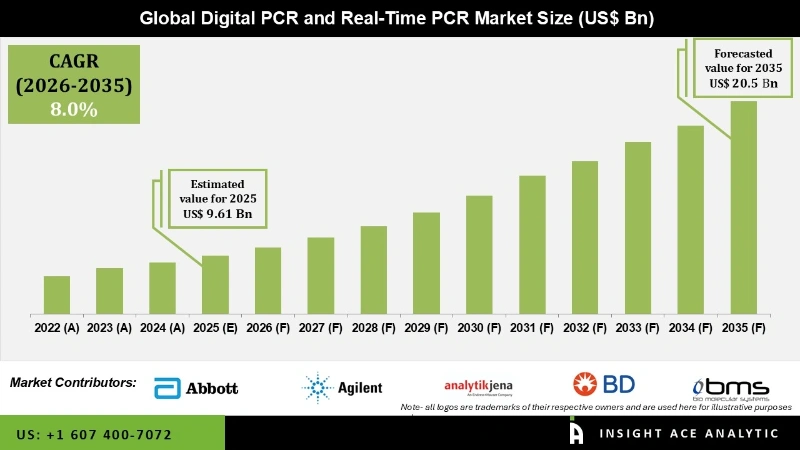
Digital PCR and Real-Time PCR Market Key Takeaways:
|
Key Industry Insights & Findings from the Report:
- This market growth is being attributed to an increasing focus on early disease diagnosis due to the rise of infectious diseases globally.
- A rise in the demand for digital PCR and real-time PCR due to the expansion of customized medicine fuels the market’s demand for these technologies.
- North America dominated the market and accounted for a global revenue share in 2024.
- The shortage of qualified workers and slow updating in emerging economies are expected to impede the market's growth.
Digital PCR is an end-point PCR method for absolute quantification and analyzing minority sequences against a backdrop of similar majority sequences. Digital PCR (dPCR) allows for the exact and sensitive measurement of nucleic acids. Real time PCR is a method for collecting data during the PCR process, integrating amplification and detection into a single phase. Nucleic acid quantification using real-time quantitative PCR is sensitive, specific, and repeatable. It monitors the amplification of a specific DNA molecule throughout the PCR.
The rising prevalence of genetic disorders and major infectious diseases, increased use of biomarker profiling for disease diagnosis, and the successful completion of the Human Genome Project are the primary factors driving demand for technologically advanced PCR tests during the forecast period. Significant technological developments, increased investments, funding, and grants are expected to raise demand for digital PCR in various applications in the next years.
However, the COVID-19 pandemic has significantly impacted the healthcare industry. Digital PCR and real-time PCR have generated considerable interest due to their use in diagnostics and drug discovery. Therefore, industry suppliers are actively developing droplet digital PCR assays for COVID-19 detection. In addition, numerous participants in the industry have developed unique instruments and tests for COVID-19 testing.
Competitive Landscape:
Some of the Digital PCR and Real-Time PCR Market players are:
- Abbott Laboratories
- Agilent Technologies, Inc.
- Analytik Jena AG
- Becton Dickinson and Company
- Bio Molecular Systems
- Biomeme Inc.
- Biomérieux S.A.
- Bioneer Corporation
- Bio-Rad Laboratories, Inc.
- Cole-Parmer Instrument Company Llc
- Danaher Corporation
- Elitech Group Sas
- Endress+Hauser
- Enzo Life Sciences, Inc.
- Eppendorf AG
- Hoffmann-LA Roche Ltd.
- Fluidigm Corporation
- JN Medsys PTE, LTD.
- Merck KGAA
- Meridian Bioscience, Inc.
- Promega Corporation
- Qiagen N.V.
- Quidel Corporation
- Quanta Biosciences Inc.
- Sacace Biotechnologies Srl
- Standard Biotools
- Stilla Technologies
- Takara Bio, Inc.
- Thermo Fisher Scientific, Inc.
Market Segmentation:
The digital PCR and real-time PCR market is segmented on the basis of product & service, application, and end-user. The product & service segment includes QPCR (QPCR reagents and consumables, QPCR instruments, QPCR software and services), DPCR (DPCR reagents and consumables, DPCR instruments, DPCR software and services). DPCR instruments are further segmented into chip-based digital PCR and beaming digital PCR. On the basis of QPCR application, the market is segmented into clinical applications (infectious disease testing, oncology testing, blood screening, transplant diagnostics, other clinical applications), research applications, forensic applications, environmental applications, and other applications. The end-user segment includes Hospitals and diagnostic laboratories, academic & research institutes, pharmaceutical & biotechnology, CROS & CDMOS, forensic laboratories, and other end-users.
Based on dPCR & qPCR end-users, the Hospitals And Diagnostic Laboratories Segment Is Accounted As A Major Contributor In The Digital PCR And Real-Time PCR Market
The hospitals and diagnostic laboratories category is expected to hold a considerable share in the global Digital PCR and Real-Time PCR Market in 2024 owing to the rising availability of qPCR reagents in the market. The use of clinical diagnostics and the continual growth of healthcare infrastructure in emerging markets, and the increasing prevalence and incidence of target diseases such as HIV/AIDS, cancer, malaria, and tuberculosis are likely to drive demand for hospital and diagnostic facilities in the Digital PCR (dPCR) and Real-Time PCR (qPCR) markets. The growing awareness among End-Users about the benefits of qPCR ineffective and speedy illness diagnosis is projected to fuel the growth of the Digital PCR (dPCR) and Real-Time PCR (qPCR) Markets in the coming years.
qPCR & dPCR reagents and consumables segment witness growth at a rapid rate
The QPCR & DPCR reagents and consumables segment is projected to expand at a rapid rate in the global Digital PCR and Real-Time PCR Market. This can be ascribed to the COVID-19 pandemic, increased demand for early illness detection, and the number of prominent companies producing digital PCR consumables and reagents. Furthermore, the widespread usage of consumables and reagents and the growing demand for healthcare, research, and other uses contribute to the segment's substantial share. Furthermore, the category is expected to keep its lead during the forecast period.
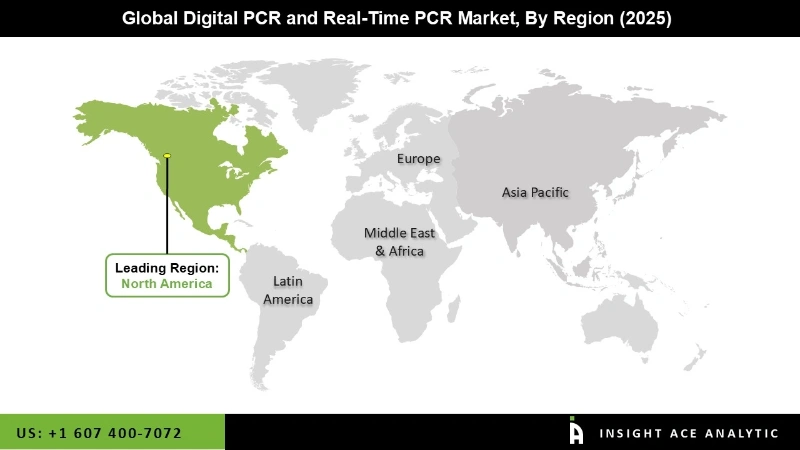
In The Region, The North America Digital PCR And Real-Time PCR Market Holds A Significant Revenue Share
The North America Digital PCR and Real-Time PCR Market is expected to register the highest market share in terms of revenue in the near future, primarily due to the presence of favourable rules and many government efforts aimed at developing healthcare infrastructure, as well as the region's high disease prevalence. Furthermore, the region's strong presence of significant manufacturers and an increasing need for rapid diagnostic tests are projected to be critical factors driving the North American market throughout the projection period.
Due to the significant developments by China and Japan for technological integration of PCR methodologies, as well as the development of healthcare, R&D, and clinical development frameworks by emerging economies such as India and Australia, Asia Pacific is the most lucrative market. Throughout the projection period, the industry will see profitable growth possibilities.
Recent Developments:
- In Oct 2022, BioExcel Diagnostics and Takara Bio USA have formed a collaborative alliance with the intention of creating a qPCR multiplex syndromic infectious disease diagnostic panel. qPCR automation technology and reagents supplied by Takara Bio USA, an entirely owned subsidiary of Takara Bio headquartered in Shiga, Japan, were utilized by the panels to identify antimicrobial resistance genes, bacteria, fungi, and viruses.
- In Aug 2022, Roche announced the introduction of its first digital polymerase chain reaction (PCR) system, the Digital LightCycler System. Designed to accurately quantify trace quantities of specific DNA and RNA targets that are not ordinarily detectable by conventional PCR techniques, this next-generation system detects disease. Clinical researchers are able to partition DNA and RNA extracted from a pre-existing clinical sample into up to one hundred thousand microscopic individual reactions using the Digital LightCycler System. The results of the PCR can then be subjected to an extremely sophisticated data analysis by the system.
Digital PCR and Real-Time PCR Market Report Scope:
| Report Attribute | Specifications |
| Market size value in 2025 | USD 9.61 Billion |
| Revenue forecast in 2035 | USD 20.5 Billion |
| Growth rate CAGR | CAGR of 8.0% from 2026 to 2035 |
| Quantitative units | Representation of revenue in US$ Bn, and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report coverage | The forecast of revenue, the position of the company, the competitive market statistics, growth prospects, and trends |
| Segments covered | Product & Service, Application, And End-User |
| Regional scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; Southeast Asia |
| Competitive Landscape | Danaher Corporation, F. Hoffmann-LA Roche Ltd., Thermo Fisher Scientific Inc., Qiagen N.V., Bio-Rad Laboratories, Inc., Agilent Technologies, Inc., Takara Bio Inc., Becton, Dickson and Company, Abbott Laboratories, Promega Corporation, Merck KGAA, Endress+Hauser, Biomerieux, Meridian Bioscience, Eppendorf AG, Quidel Corporation, Standard Biotools, Enzo Biochem, Inc., Stilla Technologies, JN Medsys PTE, LTD. |
| Customization scope | Free customization report with the procurement of the report, Modifications to the regional and segment scope. Particular Geographic competitive landscape. |
| Pricing and available payment methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of Digital PCR and Real-Time PCR Market-
Digital PCR and Real-Time PCR Market By Product-& Services
- qPCR Product-& Services
- qPCR Reagents and Consumables
- qPCR Instruments
- qPCR Software and Services
- dPCR Product-& Services
- dPCR Reagents and Consumables
- dPCR Instruments
- Chip-based Digital PCR
- Beaming Digital PCR
- dPCR Software and Services)
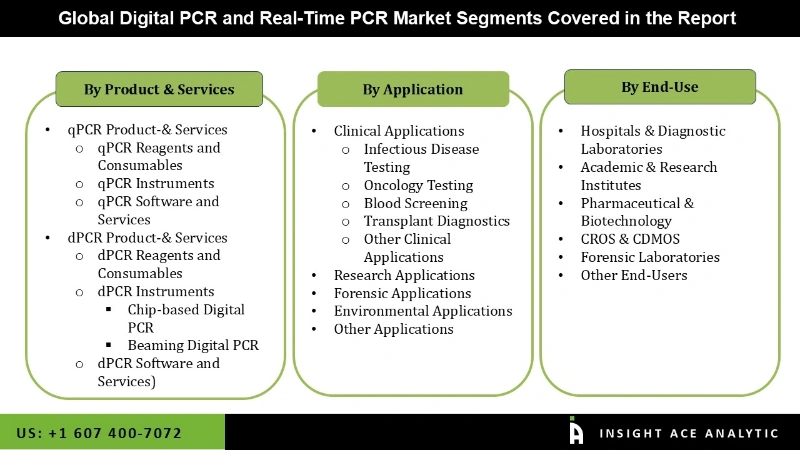
Digital PCR and Real-Time PCR Market By Application-
- Clinical Applications
- Infectious Disease Testing
- Oncology Testing
- Blood Screening
- Transplant Diagnostics
- Other Clinical Applications
- Research Applications
- Forensic Applications
- Environmental Applications
- Other Applications
Digital PCR and Real-Time PCR Market By End-User-
- Hospitals & Diagnostic Laboratories
- Academic & Research Institutes
- Pharmaceutical & Biotechnology
- CROS & CDMOS
- Forensic Laboratories
- Other End-Users
Digital PCR and Real-Time PCR Market By Region-
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Mexico
- Argentina
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
Digital PCR and Real-Time PCR Market Size is valued at USD 9.61 billion in 2025 and is predicted to reach USD 20.5 billion by the year 2035
Digital PCR and Real-Time PCR Market expected to grow at a 8.0% CAGR during the forecast period for 2026-2035.
Danaher Corporation, F. Hoffmann-LA Roche Ltd., Thermo Fisher Scientific Inc., Qiagen N.V., Bio-Rad Laboratories, Inc., Agilent Technologies, Inc.,
Product & Service, Application, and End-User are the key segments of the Digital PCR and Real-Time PCR Market.
North America region is leading the Digital PCR and Real-Time PCR Market.