De identified Health Data Market Size, Share and Trends Analysis 2026 to 2035
What is De identified Health Data Market Size?
De identified Health Data Market Size is valued at USD 8.77 Bn in 2025 and is predicted to reach USD 21.47 Bn by the year 2035 at an 9.5% CAGR during the forecast period for 2026 to 2035.
De-identified Health Data Market, Share & Trends Analysis Report, By Type of Data (Clinical, Genomic and Others), By End-use (Pharmaceutical Companies, Biotechnology Firms and Others), By Application, By Region, and Segment Forecasts, 2026 to 2035
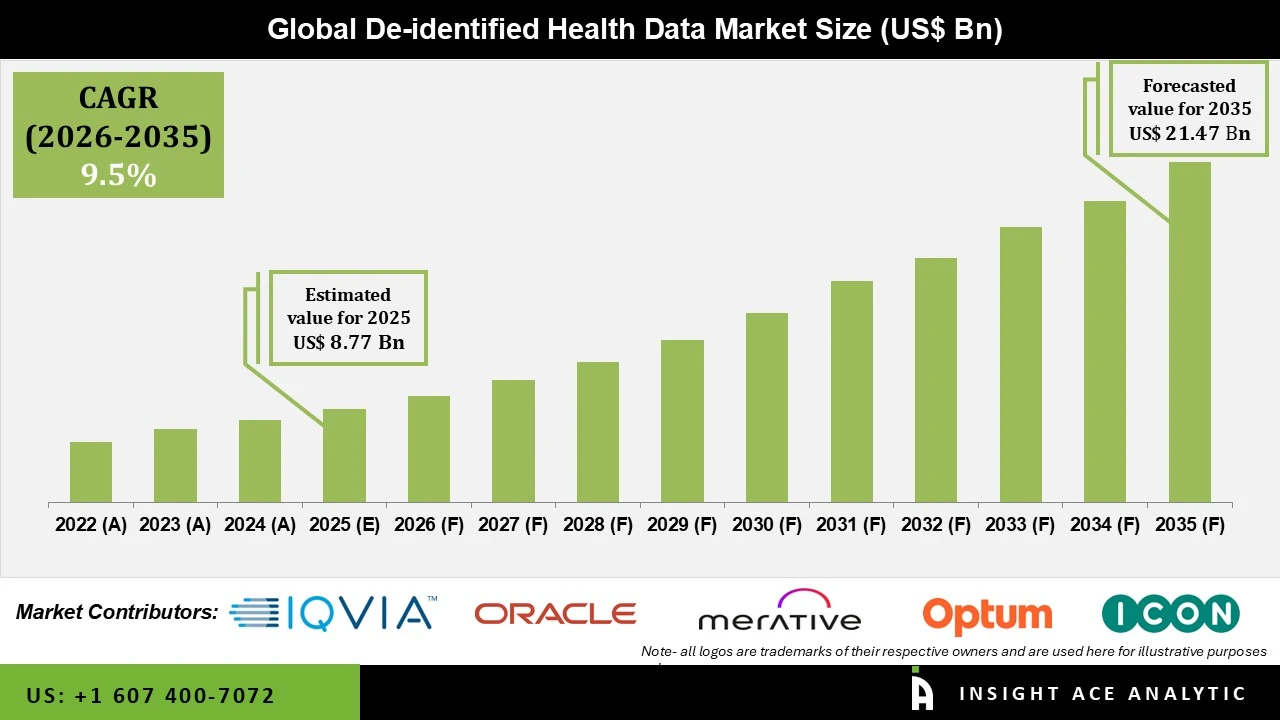
De identified Health Data Market Key Takeaways:
|
De-identification is an organizational method used to eliminate personal information from data that is gathered, utilized, stored, and shared with other organizations. Rather than being a single strategy, it encompasses a set of approaches, algorithms, and tools applied to various types of data with differing levels of effectiveness. More aggressive de-identification algorithms typically provide stronger privacy protection, though they can reduce the dataset's overall usefulness. For enterprises, government agencies, and other organizations that seek to make data accessible to external parties, de-identification is especially crucial. It safeguards individuals' privacy by preventing the unauthorized disclosure of their personal health information. This allows de-identified data to be used for diverse research purposes without compromising patient confidentiality and facilitates the sharing of health data for collaborative research and analysis.
Section 164.514(a) of the HIPAA Privacy Rule provides the standard for de-identification of protected health information, stating that health information is not considered individually identifiable if it does not identify an individual and if the covered entity has no reasonable basis to believe it could be used to identify one. Similarly, the General Data Protection Regulation (GDPR) in the European Union sets strict standards for data protection, including the processing of personal data, which can include de-identified health data. Both regulatory frameworks, HIPAA and GDPR, impose stringent requirements for managing personal health information. De-identification serves as a key method for organizations to comply with these regulations while still making health data available for research and collaboration. As healthcare systems become increasingly interconnected, the need for collaboration between institutions, researchers, and pharmaceutical companies grows. De-identified data facilitates the exchange of health information across organizations, supporting joint research, drug development, and clinical trials without compromising patient confidentiality.
Competitive Landscape
Some of the Major Key Players in the De-identified Health Data Market are
- IQVIA
- Oracle
- Merative
- Optum, Inc.
- ICON plc
- Veradigm LLC
- IBM
- Flatiron Health
- Premier, Inc.
- Shaip
- Komodo Health, Inc.
- Evidation Health, Inc.
- Medidata
- Clarify Health Solutions
- Satori Cyber Ltd.
- Kitware
- BioData Consortium
- Akrivia Health
- iMerit
Market Segmentation
The de-identified health data market is segmented based on the type of data, end-use, application. Based on the type of data, the market is divided into clinical, genomic, patient demographics, prescription data, claims data, behavioral data, wearable and sensor data, survey and patient-reported data, imaging data, laboratory data, hospital and provider data, social determinants of health (SDOH) data, pharmacogenomic data, biometric data, operational and financial data, epidemiological data, healthcare utilization data, others. Based on the end-use, the market is divided pharmaceutical companies, biotechnology firms, medical device manufacturers, healthcare providers, insurance companies/ healthcare payers, research institutions, government agencies and others. Based on the application, the market is divided into clinical research and trials, public health, precision medicine, health economics and outcomes research (HEOR), population health management, drug discovery and development, healthcare quality improvement, insurance underwriting and risk assessment, market access and commercial strategy, business intelligence and operational efficiency, telemedicine and remote monitoring, patient engagement and support programs, others.
The Clinical Data Segment is Expected to Have the Highest Growth Rate During the Forecast Period
Based on the type of data, the market is divided into clinical, genomic, patient demographics prescription data, claims data, behavioral data, wearable and sensor data, survey and patient-reported data, imaging data, laboratory data, hospital and provider data, social determinants of health (SDOH) data, pharmacogenomic data, biometric data, operational and financial data, epidemiological data, healthcare utilization data, others. Among these, the clinical data segment is expected to have the highest growth rate during the forecast period. Clinical data includes a wide range of information such as medical histories, diagnoses, treatments, procedures, outcomes, and lab results. This data provides a detailed view of patient health, making it highly valuable for research, healthcare optimization, and decision-making. Clinical data is essential for advanced healthcare analytics, machine learning, and AI-driven models aimed at improving diagnostics, predicting health outcomes, and optimizing healthcare delivery. The use of this data fuels advancements in personalized medicine and value-based care.
The Clinical Research and Trials Segment Dominates the Market
Based on the application, the market is divided into clinical research and trials, public health, precision medicine, health economics and outcomes research (HEOR), population health management, drug discovery and development, healthcare quality improvement, insurance underwriting and risk assessment, market access and commercial strategy, business intelligence and operational efficiency, telemedicine and remote monitoring, patient engagement and support programs, others. Among these, the clinical research and trials segment dominates the market. Clinical research and trials require access to vast amounts of health data to generate statistically significant results. De-identified health data provides researchers with large, diverse patient datasets while protecting individual privacy. This is essential for understanding disease progression, treatment effectiveness, and potential side effects across different populations. Pharmaceutical and biotech companies are major consumers of de-identified health data, as they are constantly conducting research for drug discovery and clinical trials. The continuous demand for de-identified data by these industries ensures that the clinical research and trials segment remains dominant.
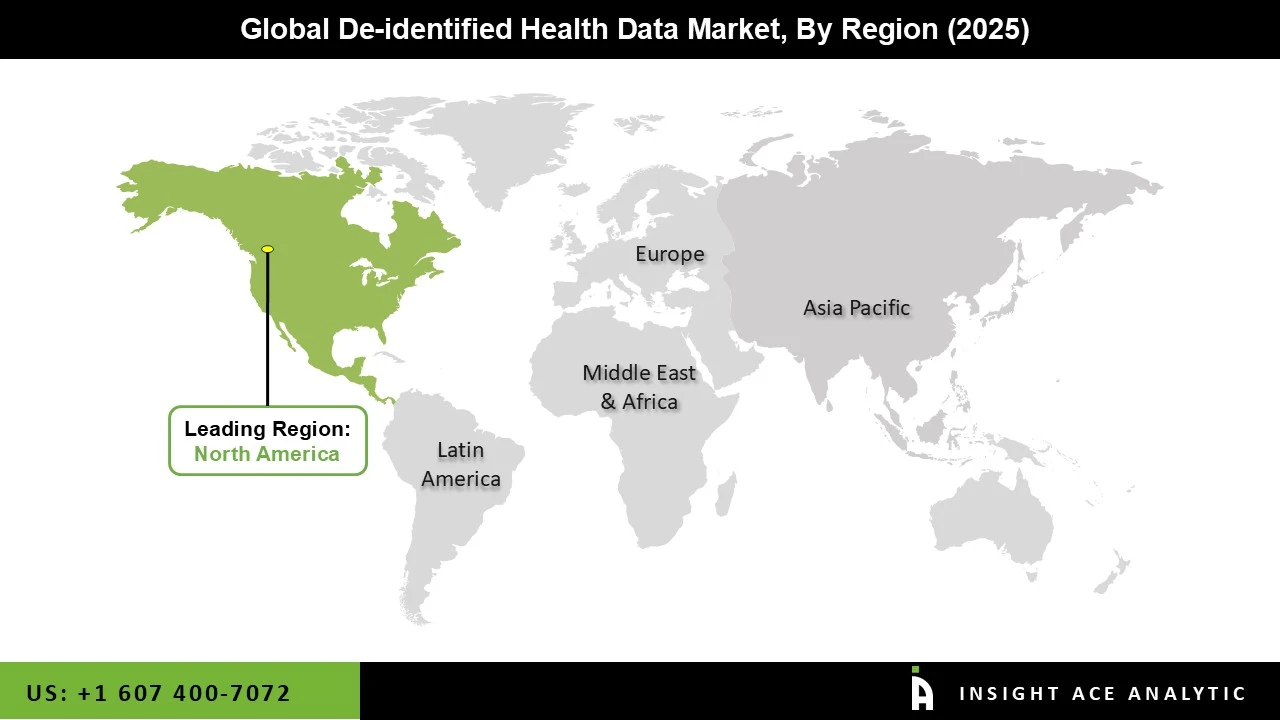
North America has the Largest Market Share During the Forecast Period.
North America leads in the development and adoption of advanced healthcare analytics, machine learning, and AI technologies. These innovations require large volumes of health data to train algorithms and develop predictive models. De-identified health data is critical to feeding these technologies, driving further demand in the region. The rise of precision medicine in North America, especially through initiatives like the All of Us Research Program in the U.S., requires vast amounts of diverse and de-identified health data to understand genetic, environmental, and lifestyle factors that influence health. North America's leadership in precision medicine drives the demand for de-identified datasets to enable tailored treatments and interventions.
Recent Developments:
- In December 2020, DeNA Life Science Inc and IQVIA collaboration on the Genome Wide Study Platform, a platform for pharmaceutical companies, in December 2020 with the goal of building a database of statistical genetic information* about disease and pertinent analysis findings of survey data on disease, physical predispositions, lifestyle choices, etc., and using that data for drug utility & safety validation and drug discovery research.
- In Oct 2018, IQVIA, declared that it will collaborate with Genomics England to create a platform that combines clinical and de-identified genomics data; the nature of the partnership was not disclosed.
De-identified Health Data Market Report Scope
| Report Attribute | Specifications |
| Market Size Value In 2025 | USD 8.77 Bn |
| Revenue Forecast In 2035 | USD 21.47 Bn |
| Growth Rate CAGR | CAGR of 9.5% from 2026 to 2035 |
| Quantitative Units | Representation of revenue in US$ Bn and CAGR from 2026 to 2035 |
| Historic Year | 2022 to 2025 |
| Forecast Year | 2026-2035 |
| Report Coverage | The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
| Segments Covered | By Type Of Data, By End-Use, By Application |
| Regional Scope | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Country Scope | U.S.; Canada; U.K.; Germany; China; India; Japan; Brazil; Mexico; The UK; France; Italy; Spain; China; Japan; India; South Korea; Southeast Asia; South Korea; South East Asia |
| Competitive Landscape | IQVIA, Oracle (Cerner Corporation), Merative (Truven Health Analytics), Optum, Inc. (UnitedHealth Group), ICON plc, Veradigm LLC (Formerly known as Allscripts), IBM, Flatiron Health (F. Hoffmann-La Roche Ltd), Premier, Inc., Shaip, Komodo Health, Inc., Evidation Health, Inc., Medidata, Clarify Health Solutions, Satori Cyber Ltd., Kitware, BioData Consortium, Akrivia Health, iMerit |
| Customization Scope | Free customization report with the procurement of the report and modifications to the regional and segment scope. Particular Geographic competitive landscape. |
| Pricing and Available Payment Methods | Explore pricing alternatives that are customized to your particular study requirements. |
Segmentation of De-identified Health Data Market
Global De-identified Health Data Market- By End Use
- Pharmaceutical Companies
- Biotechnology Firms
- Medical Device Manufacturers
- Healthcare Providers
- Insurance Companies/ Healthcare Payers
- Research Institutions
- Government Agencies
- Others
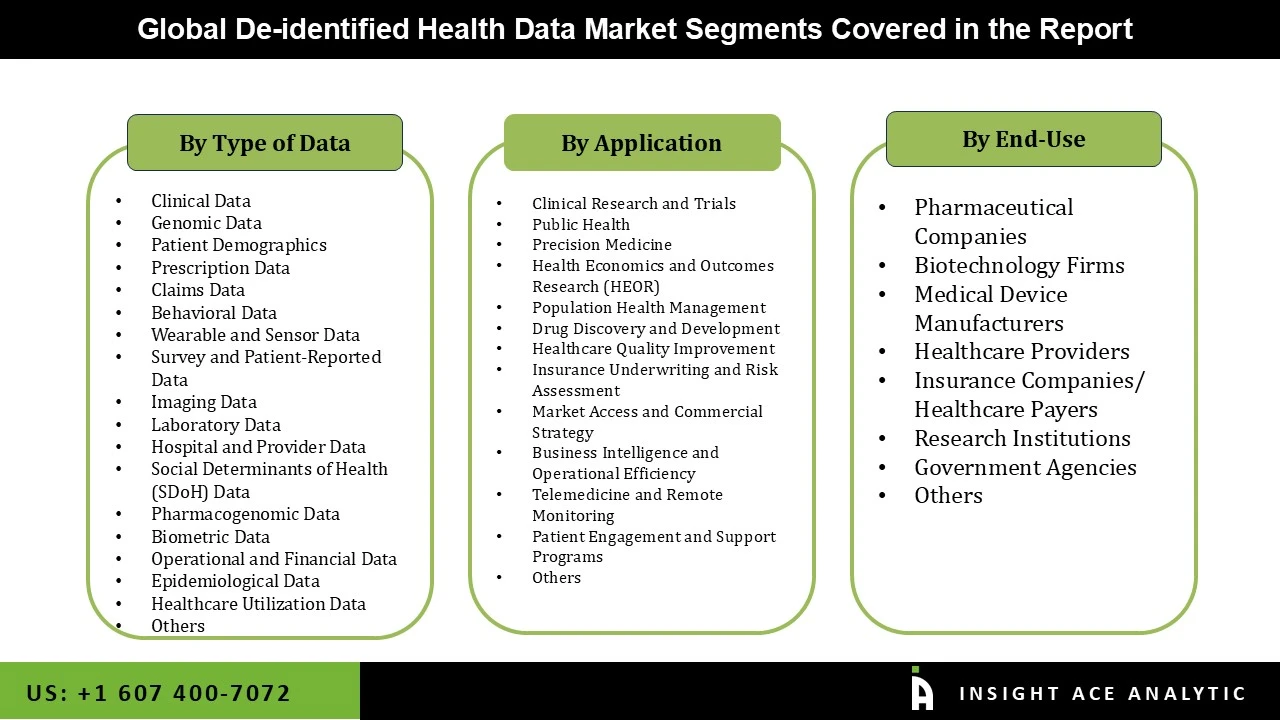
Global De-identified Health Data Market – By Application
- Clinical Research and Trials
- Public Health
- Precision Medicine
- Health Economics and Outcomes Research (HEOR)
- Population Health Management
- Drug Discovery and Development
- Healthcare Quality Improvement
- Insurance Underwriting and Risk Assessment
- Market Access and Commercial Strategy
- Business Intelligence and Operational Efficiency
- Telemedicine and Remote Monitoring
- Patient Engagement and Support Programs
- Others
Global De-identified Health Data Market – Type of Data
- Clinical Data
- Genomic Data
- Patient Demographics
- Prescription Data
- Claims Data
- Behavioral Data
- Wearable and Sensor Data
- Survey and Patient-Reported Data
- Imaging Data
- Laboratory Data
- Hospital and Provider Data
- Social Determinants of Health (SDoH) Data
- Pharmacogenomic Data
- Biometric Data
- Operational and Financial Data
- Epidemiological Data
- Healthcare Utilization Data
- Others
Global De-identified Health Data Market – By Region
North America-
- The US
- Canada
- Mexico
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of the Middle East
Research Design and Approach
This study employed a multi-step, mixed-method research approach that integrates:
- Secondary research
- Primary research
- Data triangulation
- Hybrid top-down and bottom-up modelling
- Forecasting and scenario analysis
This approach ensures a balanced and validated understanding of both macro- and micro-level market factors influencing the market.
Secondary Research
Secondary research for this study involved the collection, review, and analysis of publicly available and paid data sources to build the initial fact base, understand historical market behaviour, identify data gaps, and refine the hypotheses for primary research.
Sources Consulted
Secondary data for the market study was gathered from multiple credible sources, including:
- Government databases, regulatory bodies, and public institutions
- International organizations (WHO, OECD, IMF, World Bank, etc.)
- Commercial and paid databases
- Industry associations, trade publications, and technical journals
- Company annual reports, investor presentations, press releases, and SEC filings
- Academic research papers, patents, and scientific literature
- Previous market research publications and syndicated reports
These sources were used to compile historical data, market volumes/prices, industry trends, technological developments, and competitive insights.

Primary Research
Primary research was conducted to validate secondary data, understand real-time market dynamics, capture price points and adoption trends, and verify the assumptions used in the market modelling.
Stakeholders Interviewed
Primary interviews for this study involved:
- Manufacturers and suppliers in the market value chain
- Distributors, channel partners, and integrators
- End-users / customers (e.g., hospitals, labs, enterprises, consumers, etc., depending on the market)
- Industry experts, technology specialists, consultants, and regulatory professionals
- Senior executives (CEOs, CTOs, VPs, Directors) and product managers
Interview Process
Interviews were conducted via:
- Structured and semi-structured questionnaires
- Telephonic and video interactions
- Email correspondences
- Expert consultation sessions
Primary insights were incorporated into demand modelling, pricing analysis, technology evaluation, and market share estimation.
Data Processing, Normalization, and Validation
All collected data were processed and normalized to ensure consistency and comparability across regions and time frames.
The data validation process included:
- Standardization of units (currency conversions, volume units, inflation adjustments)
- Cross-verification of data points across multiple secondary sources
- Normalization of inconsistent datasets
- Identification and resolution of data gaps
- Outlier detection and removal through algorithmic and manual checks
- Plausibility and coherence checks across segments and geographies
This ensured that the dataset used for modelling was clean, robust, and reliable.
Market Size Estimation and Data Triangulation
Bottom-Up Approach
The bottom-up approach involved aggregating segment-level data, such as:
- Company revenues
- Product-level sales
- Installed base/usage volumes
- Adoption and penetration rates
- Pricing analysis
This method was primarily used when detailed micro-level market data were available.

Top-Down Approach
The top-down approach used macro-level indicators:
- Parent market benchmarks
- Global/regional industry trends
- Economic indicators (GDP, demographics, spending patterns)
- Penetration and usage ratios
This approach was used for segments where granular data were limited or inconsistent.
Hybrid Triangulation Approach
To ensure accuracy, a triangulated hybrid model was used. This included:
- Reconciling top-down and bottom-up estimates
- Cross-checking revenues, volumes, and pricing assumptions
- Incorporating expert insights to validate segment splits and adoption rates
This multi-angle validation yielded the final market size.
Forecasting Framework and Scenario Modelling
Market forecasts were developed using a combination of time-series modelling, adoption curve analysis, and driver-based forecasting tools.
Forecasting Methods
- Time-series modelling
- S-curve and diffusion models (for emerging technologies)
- Driver-based forecasting (GDP, disposable income, adoption rates, regulatory changes)
- Price elasticity models
- Market maturity and lifecycle-based projections
Scenario Analysis
Given inherent uncertainties, three scenarios were constructed:
- Base-Case Scenario: Expected trajectory under current conditions
- Optimistic Scenario: High adoption, favourable regulation, strong economic tailwinds
- Conservative Scenario: Slow adoption, regulatory delays, economic constraints
Sensitivity testing was conducted on key variables, including pricing, demand elasticity, and regional adoption.
Request Customization
Add countries, segments, company profiles, or extend forecast — free 10% customization with purchase.
Customize This Report →Enquire Before Buying
Speak with our analyst team about scope, methodology, pricing, or deliverable formats.
Enquire Now →Frequently Asked Questions
De-identified Health Data Market Size is valued at USD 8.77 Bn in 2025 and is predicted to reach USD 21.47 Bn by the year 2035
The De-identified Health Data Market is expected to grow at an 9.5% CAGR during the forecast period for 2026 to 2035.
IQVIA, Oracle (Cerner Corporation), Merative (Truven Health Analytics), Optum, Inc. (UnitedHealth Group), ICON plc, Veradigm LLC (Formerly known as Al
The De-identified Health Data Market has three segments Type Of Data, End-Use, and Application.
North American region is leading the De-identified Health Data Market?